VDAC2
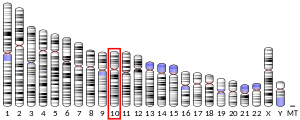
Voltage-dependent anion-selective channel protein 2 is a protein that in humans is encoded by the VDAC2 gene on chromosome 10.[4][5] This protein is a voltage-dependent anion channel and shares high structural homology with the other VDAC isoforms.[6][7][8] VDACs are generally involved in the regulation of cell metabolism, mitochondrial apoptosis, and spermatogenesis.[9][10][11][12] Additionally, VDAC2 participates in cardiac contractions and pulmonary circulation, which implicate it in cardiopulmonary diseases.[9][10] VDAC2 also mediates immune response to infectious bursal disease (IBD).[10]
Structure
The three VDAC isoforms in human are highly conserved, particularly with respect to their 3D structure. VDACs form a wide β-barrel structure, inside of which the N-terminal resides to partially close the pore. The sequence of the VDAC2 isoform contains an abundance of cysteines, which allow for the formation of disulfide bridges and, ultimately, affect the flexibility of the β-barrel. VDACs also contain a mitochondrial targeting sequence for the protein's translocation to the outer mitochondrial membrane.[13] In particular, VDAC2 possesses an N-terminal longer by 11 residues compared to the other two isoforms.[8]
Function
VDAC2 belongs to the mitochondrial porin family and is expected to share similar biological functions to the other VDAC isoforms. VDACs generally are involved in cellular energy metabolism by transporting ATP and other small ions and metabolites across the outer mitochondrial membrane.[9][10] In mammalian cardiomyocytes, VDAC2 promotes mitochondrial transport of calcium ions in order to power cardiac contractions.[9]
In addition, VDACs form part of the mitochondrial permeability transition pore (MPTP) and, thus, facilitate cytochrome C release, leading to apoptosis.[9][14] VDACs have also been observed to interact with pro- or antiapoptotic proteins, such as Bcl-2 family proteins and kinases, and so may contribute to apoptosis independently from the MPTP.[10][12][14] VDAC2 in particular has demonstrated a protective effect in cells undergoing mitochondrial apoptosis, and may even confer protection during aging.[15][16]
Furthermore, VDAcs have been linked to spermatogenesis, sperm maturation, motility, and fertilization.[12] Though all VDAC isoforms are ubiquitously expressed, VDAC2 is majorly found in the sperm outer dense fiber (ODF), where it is hypothesized to promote proper assembly and maintenance of sperm flagella.[17][18] It also localizes to the acrosomal membrane of the sperm, where it putatively mediates calcium ion transmembrane transport.[19]
Clinical significance
The VDAC2 protein belongs to a group of mitochondrial membrane channels involved in translocation of adenine nucleotides through the outer membrane. These channels may also function as a mitochondrial binding site for hexokinase and glycerol kinase. The VDAC is an important constituent in apoptotic signaling and oxidative stress, most notably as part of the mitochondrial death pathway and cardiac myocyte apoptosis signaling.[20] Programmed cell death is a distinct genetic and biochemical pathway essential to metazoans. An intact death pathway is required for successful embryonic development and the maintenance of normal tissue homeostasis. Apoptosis has proven to be tightly interwoven with other essential cell pathways. The identification of critical control points in the cell death pathway has yielded fundamental insights for basic biology, as well as provided rational targets for new therapeutics a normal embryologic processes, or during cell injury (such as ischemia-reperfusion injury during heart attacks and strokes) or during developments and processes in cancer, an apoptotic cell undergoes structural changes including cell shrinkage, plasma membrane blebbing, nuclear condensation, and fragmentation of the DNA and nucleus. This is followed by fragmentation into apoptotic bodies that are quickly removed by phagocytes, thereby preventing an inflammatory response.[21] It is a mode of cell death defined by characteristic morphological, biochemical and molecular changes. It was first described as a "shrinkage necrosis", and then this term was replaced by apoptosis to emphasize its role opposite mitosis in tissue kinetics. In later stages of apoptosis the entire cell becomes fragmented, forming a number of plasma membrane-bounded apoptotic bodies which contain nuclear and or cytoplasmic elements. The ultrastructural appearance of necrosis is quite different, the main features being mitochondrial swelling, plasma membrane breakdown and cellular disintegration. Apoptosis occurs in many physiological and pathological processes. It plays an important role during embryonal development as programmed cell death and accompanies a variety of normal involutional processes in which it serves as a mechanism to remove "unwanted" cells.
The VDAC2 protein has been implicated in cardioprotection against ischemia-reperfusion injury, such as during ischemic preconditioning of the heart.[22] Although a large burst of reactive oxygen species (ROS) is known to lead to cell damage, a moderate release of ROS from the mitochondria, which occurs during nonlethal short episodes of ischemia, can play a significant triggering role in the signal transduction pathways of ischemic preconditioning leading to reduction of cell damage. It has even been observed that during this release of reactive oxygen species, VDAC2 plays an important role in the mitochondrial cell death pathway transduction hereby regulating apoptotic signaling and cell death.
The VDAC2 protein has been linked persistent pulmonary hypertension of the newborn (PPHN), which causes a large majority of neonatal morbidity and mortality, due to its role as a major regulator of endothelium-dependent nitric oxide synthase (eNOS) in the pulmonary endothelium. eNOS has been attributed with regulating NOS activity in response to physiological stimuli, which is vital to maintain NO production for proper blood circulation to the lungs. As a result, VDAC2 is significantly involved in pulmonary circulation and may become a therapeutic target for treating diseases such as pulmonary hypertension,[10]
VDAC2 may also serve an immune function, as it has been hypothesized to detect and induce apoptosis in cells infected by the IBD virus. IBD, the equivalent HIV in birds, can compromise their immune systems and even cause fatal injury to the lymphoid organ, Studies of this process indicate that VDAC2 interacts with the viral protein V5 to mediate cell death.[12]
Interactions
VDAC2 has been shown to interact with:
See also
References
- GRCh38: Ensembl release 89: ENSG00000165637 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Blachly-Dyson E, Baldini A, Litt M, McCabe ER, Forte M (Mar 1994). "Human genes encoding the voltage-dependent anion channel (VDAC) of the outer mitochondrial membrane: mapping and identification of two new isoforms". Genomics. 20 (1): 62–7. doi:10.1006/geno.1994.1127. PMID 7517385.
- Messina A, Oliva M, Rosato C, Huizing M, Ruitenbeek W, van den Heuvel LP, Forte M, Rocchi M, De Pinto V (Feb 1999). "Mapping of the human Voltage-Dependent Anion Channel isoforms 1 and 2 reconsidered". Biochemical and Biophysical Research Communications. 255 (3): 707–10. doi:10.1006/bbrc.1998.0136. PMID 10049775.
- Mao M, Fu G, Wu JS, Zhang QH, Zhou J, Kan LX, Huang QH, He KL, Gu BW, Han ZG, Shen Y, Gu J, Yu YP, Xu SH, Wang YX, Chen SJ, Chen Z (Jul 1998). "Identification of genes expressed in human CD34(+) hematopoietic stem/progenitor cells by expressed sequence tags and efficient full-length cDNA cloning". Proceedings of the National Academy of Sciences of the United States of America. 95 (14): 8175–80. doi:10.1073/pnas.95.14.8175. PMC 20949. PMID 9653160.
- Rahmani Z, Maunoury C, Siddiqui A (1998). "Isolation of a novel human voltage-dependent anion channel gene". European Journal of Human Genetics. 6 (4): 337–40. doi:10.1038/sj.ejhg.5200198. PMID 9781040.
- Amodeo GF, Scorciapino MA, Messina A, De Pinto V, Ceccarelli M (2014). "Charged residues distribution modulates selectivity of the open state of human isoforms of the voltage dependent anion-selective channel". PLOS ONE. 9 (8): e103879. doi:10.1371/journal.pone.0103879. PMC 4146382. PMID 25084457.
- Subedi KP, Kim JC, Kang M, Son MJ, Kim YS, Woo SH (Feb 2011). "Voltage-dependent anion channel 2 modulates resting Ca²+ sparks, but not action potential-induced Ca²+ signaling in cardiac myocytes". Cell Calcium. 49 (2): 136–43. doi:10.1016/j.ceca.2010.12.004. PMID 21241999.
- Alvira CM, Umesh A, Husted C, Ying L, Hou Y, Lyu SC, Nowak J, Cornfield DN (Nov 2012). "Voltage-dependent anion channel-2 interaction with nitric oxide synthase enhances pulmonary artery endothelial cell nitric oxide production". American Journal of Respiratory Cell and Molecular Biology. 47 (5): 669–78. doi:10.1165/rcmb.2011-0436OC. PMC 3547107. PMID 22842492.
- Cheng EH, Sheiko TV, Fisher JK, Craigen WJ, Korsmeyer SJ (Jul 2003). "VDAC2 inhibits BAK activation and mitochondrial apoptosis". Science. 301 (5632): 513–7. doi:10.1126/science.1083995. PMID 12881569.
- Li Z, Wang Y, Xue Y, Li X, Cao H, Zheng SJ (Feb 2012). "Critical role for voltage-dependent anion channel 2 in infectious bursal disease virus-induced apoptosis in host cells via interaction with VP5". Journal of Virology. 86 (3): 1328–38. doi:10.1128/JVI.06104-11. PMC 3264341. PMID 22114330.
- De Pinto V, Messina A, Lane DJ, Lawen A (May 2010). "Voltage-dependent anion-selective channel (VDAC) in the plasma membrane". FEBS Letters. 584 (9): 1793–9. doi:10.1016/j.febslet.2010.02.049. PMID 20184885.
- Lee MJ, Kim JY, Suk K, Park JH (May 2004). "Identification of the hypoxia-inducible factor 1 alpha-responsive HGTD-P gene as a mediator in the mitochondrial apoptotic pathway". Molecular and Cellular Biology. 24 (9): 3918–27. doi:10.1128/mcb.24.9.3918-3927.2004. PMC 387743. PMID 15082785.
- De Pinto V, Guarino F, Guarnera A, Messina A, Reina S, Tomasello FM, Palermo V, Mazzoni C (2010). "Characterization of human VDAC isoforms: a peculiar function for VDAC3?". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1797 (6–7): 1268–75. doi:10.1016/j.bbabio.2010.01.031. PMID 20138821.
- Reina S, Palermo V, Guarnera A, Guarino F, Messina A, Mazzoni C, De Pinto V (Jul 2010). "Swapping of the N-terminus of VDAC1 with VDAC3 restores full activity of the channel and confers anti-aging features to the cell". FEBS Letters. 584 (13): 2837–44. doi:10.1016/j.febslet.2010.04.066. PMID 20434446.
- Majumder S, Slabodnick M, Pike A, Marquardt J, Fisk HA (Oct 2012). "VDAC3 regulates centriole assembly by targeting Mps1 to centrosomes". Cell Cycle. 11 (19): 3666–78. doi:10.4161/cc.21927. PMC 3478317. PMID 22935710.
- Majumder S, Fisk HA (Mar 2013). "VDAC3 and Mps1 negatively regulate ciliogenesis". Cell Cycle. 12 (5): 849–58. doi:10.4161/cc.23824. PMC 3610733. PMID 23388454.
- Liu B, Wang P, Wang Z, Zhang W (9 February 2011). "The use of anti-VDAC2 antibody for the combined assessment of human sperm acrosome integrity and ionophore A23187-induced acrosome reaction". PLOS ONE. 6 (2): e16985. doi:10.1371/journal.pone.0016985. PMC 3036732. PMID 21347391.
- Danial NN, Korsmeyer SJ (Jan 2004). "Cell death: critical control points". Cell. 116 (2): 205–19. doi:10.1016/S0092-8674(04)00046-7. PMID 14744432.
- Kerr JF, Wyllie AH, Currie AR (Aug 1972). "Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics". British Journal of Cancer. 26 (4): 239–57. doi:10.1038/bjc.1972.33. PMC 2008650. PMID 4561027.
- Liem DA, Honda HM, Zhang J, Woo D, Ping P (Dec 2007). "Past and present course of cardioprotection against ischemia-reperfusion injury". Journal of Applied Physiology. 103 (6): 2129–36. doi:10.1152/japplphysiol.00383.2007. PMID 17673563.
- Sun Y, Vashisht AA, Tchieu J, Wohlschlegel JA, Dreier L (Nov 2012). "Voltage-dependent anion channels (VDACs) recruit Parkin to defective mitochondria to promote mitochondrial autophagy". The Journal of Biological Chemistry. 287 (48): 40652–60. doi:10.1074/jbc.M112.419721. PMC 3504778. PMID 23060438.
Further reading
- Blachly-Dyson E, Zambronicz EB, Yu WH, Adams V, McCabe ER, Adelman J, Colombini M, Forte M (Jan 1993). "Cloning and functional expression in yeast of two human isoforms of the outer mitochondrial membrane channel, the voltage-dependent anion channel". The Journal of Biological Chemistry. 268 (3): 1835–41. PMID 8420959.
- Valis K, Neubauerova J, Man P, Pompach P, Vohradsky J, Kovar J (Apr 2008). "VDAC2 and aldolase A identified as membrane proteins of K562 cells with increased expression under iron deprivation". Molecular and Cellular Biochemistry. 311 (1–2): 225–31. doi:10.1007/s11010-008-9712-x. PMID 18278581.
- Mannella CA (1998). "Conformational changes in the mitochondrial channel protein, VDAC, and their functional implications". Journal of Structural Biology. 121 (2): 207–18. doi:10.1006/jsbi.1997.3954. PMID 9615439.
- Bogenhagen DF, Rousseau D, Burke S (Feb 2008). "The layered structure of human mitochondrial DNA nucleoids". The Journal of Biological Chemistry. 283 (6): 3665–75. doi:10.1074/jbc.M708444200. PMID 18063578.
- Chandra D, Choy G, Daniel PT, Tang DG (May 2005). "Bax-dependent regulation of Bak by voltage-dependent anion channel 2". The Journal of Biological Chemistry. 280 (19): 19051–61. doi:10.1074/jbc.M501391200. PMID 15757910.
- Olsen JV, Blagoev B, Gnad F, Macek B, Kumar C, Mortensen P, Mann M (Nov 2006). "Global, in vivo, and site-specific phosphorylation dynamics in signaling networks". Cell. 127 (3): 635–48. doi:10.1016/j.cell.2006.09.026. PMID 17081983.
- Yoo BC, Fountoulakis M, Cairns N, Lubec G (Jan 2001). "Changes of voltage-dependent anion-selective channel proteins VDAC1 and VDAC2 brain levels in patients with Alzheimer's disease and Down syndrome". Electrophoresis. 22 (1): 172–9. doi:10.1002/1522-2683(200101)22:1<172::AID-ELPS172>3.0.CO;2-P. PMID 11197169.
- Ahmed M, Forsberg J, Bergsten P (2005). "Protein profiling of human pancreatic islets by two-dimensional gel electrophoresis and mass spectrometry". Journal of Proteome Research. 4 (3): 931–40. doi:10.1021/pr050024a. PMID 15952740.
- Ha H, Hajek P, Bedwell DM, Burrows PD (Jun 1993). "A mitochondrial porin cDNA predicts the existence of multiple human porins". The Journal of Biological Chemistry. 268 (16): 12143–9. PMID 7685033.
- Yu WH, Wolfgang W, Forte M (Jun 1995). "Subcellular localization of human voltage-dependent anion channel isoforms". The Journal of Biological Chemistry. 270 (23): 13998–4006. doi:10.1074/jbc.270.23.13998. PMID 7539795.
- Hinsch KD, De Pinto V, Aires VA, Schneider X, Messina A, Hinsch E (Apr 2004). "Voltage-dependent anion-selective channels VDAC2 and VDAC3 are abundant proteins in bovine outer dense fibers, a cytoskeletal component of the sperm flagellum". The Journal of Biological Chemistry. 279 (15): 15281–8. doi:10.1074/jbc.M313433200. PMID 14739283.
- Decker WK, Bowles KR, Schatte EC, Towbin JA, Craigen WJ (Oct 1999). "Revised fine mapping of the human voltage-dependent anion channel loci by radiation hybrid analysis". Mammalian Genome. 10 (10): 1041–2. doi:10.1007/s003359901158. PMID 10501981.
- Andersen JS, Lam YW, Leung AK, Ong SE, Lyon CE, Lamond AI, Mann M (Jan 2005). "Nucleolar proteome dynamics". Nature. 433 (7021): 77–83. doi:10.1038/nature03207. PMID 15635413.
- Bernhard OK, Cunningham AL, Sheil MM (Apr 2004). "Analysis of proteins copurifying with the CD4/lck complex using one-dimensional polyacrylamide gel electrophoresis and mass spectrometry: comparison with affinity-tag based protein detection and evaluation of different solubilization methods". Journal of the American Society for Mass Spectrometry. 15 (4): 558–67. doi:10.1016/j.jasms.2003.12.006. PMID 15047060.
- Ewing RM, Chu P, Elisma F, Li H, Taylor P, Climie S, McBroom-Cerajewski L, Robinson MD, O'Connor L, Li M, Taylor R, Dharsee M, Ho Y, Heilbut A, Moore L, Zhang S, Ornatsky O, Bukhman YV, Ethier M, Sheng Y, Vasilescu J, Abu-Farha M, Lambert JP, Duewel HS, Stewart II, Kuehl B, Hogue K, Colwill K, Gladwish K, Muskat B, Kinach R, Adams SL, Moran MF, Morin GB, Topaloglou T, Figeys D (2007). "Large-scale mapping of human protein-protein interactions by mass spectrometry". Molecular Systems Biology. 3 (1): 89. doi:10.1038/msb4100134. PMC 1847948. PMID 17353931.
External links
- VDAC2+protein,+human at the US National Library of Medicine Medical Subject Headings (MeSH)