Zicronapine
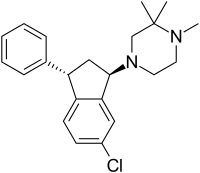
Zicronapine (/zaɪˈkrɒnəpiːn/ zye-KRON-ə-peen, previously known as Lu 31-130) is an atypical antipsychotic medication[1] formerly under development by H. Lundbeck A/S. In phase II studies zicronapine showed statistically significant separation from placebo and convincing efficacy and safety data when compared to olanzapine.[2]
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C22H27ClN2 |
| Molar mass | 354.92 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Zicronapine exhibits monoaminergic activity and has a multi-receptorial profile. In vitro and in vivo it has shown potent antagonistic effects at dopamine D1, D2 and serotonin 5HT2A receptors.[3]
In 2014 Lundbeck removed zicronapine from its development portfolio in favor of pursuing the more promising antipsychotic Lu AF35700 (a prodrug of Lu AF356152).[4]
References
- Citrome L (November 2013). "A review of the pharmacology, efficacy and tolerability of recently approved and upcoming oral antipsychotics: an evidence-based medicine approach". CNS Drugs. 27 (11): 879–911. doi:10.1007/s40263-013-0105-7. PMID 24062193. S2CID 23867019.
- "The clinical phase III programme commenced on zicronapine". January 20, 2011. Retrieved 6 February 2014.
- "Zicronapine shows significant positive data in clinical phase II in the treatment of patients with schizophrenia - planning for continued clinical work". December 18, 2009. Retrieved 6 February 2014.
- "Performance in 2014 positions Lundbeck well for 2015 and beyond" (PDF). February 5, 2015. Retrieved 18 June 2015.
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.