Soil
Soil is a mixture of organic matter, minerals, gases, liquids, and organisms that together support life. Earth's body of soil, called the pedosphere, has four important functions:
- as a medium for plant growth
- as a means of water storage, supply and purification
- as a modifier of Earth's atmosphere
- as a habitat for organisms
| Look up soil in Wiktionary, the free dictionary. |
All of these functions, in their turn, modify the soil and its properties.
The pedosphere interfaces with the lithosphere, the hydrosphere, the atmosphere, and the biosphere.[1] The term pedolith, used commonly to refer to the soil, translates to ground stone in the sense "fundamental stone."[2] Soil consists of a solid phase of minerals and organic matter (the soil matrix), as well as a porous phase that holds gases (the soil atmosphere) and water (the soil solution).[3][4][5] Accordingly, soil scientists can envisage soils as a three-state system of solids, liquids, and gases.[6]
Soil is a product of several factors: the influence of climate, relief (elevation, orientation, and slope of terrain), organisms, and the soil's parent materials (original minerals) interacting over time.[7] It continually undergoes development by way of numerous physical, chemical and biological processes, which include weathering with associated erosion. Given its complexity and strong internal connectedness, soil ecologists regard soil as an ecosystem.[8]
Most soils have a dry bulk density (density of soil taking into account voids when dry) between 1.1 and 1.6 g/cm3, while the soil particle density is much higher, in the range of 2.6 to 2.7 g/cm3.[9] Little of the soil of planet Earth is older than the Pleistocene and none is older than the Cenozoic,[10] although fossilized soils are preserved from as far back as the Archean.[11]
Soil science has two basic branches of study: edaphology and pedology. Edaphology studies the influence of soils on living things.[12] Pedology focuses on the formation, description (morphology), and classification of soils in their natural environment.[13] In engineering terms, soil is included in the broader concept of regolith, which also includes other loose material that lies above the bedrock, as can be found on the Moon and on other celestial objects as well.[14] Soil is also commonly referred to as earth or dirt; some scientific definitions distinguish dirt from soil by restricting the former term specifically to displaced soil.
Functions
Soil is a major component of the Earth's ecosystem. The world's ecosystems are impacted in far-reaching ways by the processes carried out in the soil, from ozone depletion and global warming to rainforest destruction and water pollution. With respect to Earth's carbon cycle, soil is an important carbon reservoir, and it is potentially one of the most reactive to human disturbance[15] and climate change.[16] As the planet warms, it has been predicted that soils will add carbon dioxide to the atmosphere due to increased biological activity at higher temperatures, a positive feedback (amplification).[17] This prediction has, however, been questioned on consideration of more recent knowledge on soil carbon turnover.[18]
Soil acts as an engineering medium, a habitat for soil organisms, a recycling system for nutrients and organic wastes, a regulator of water quality, a modifier of atmospheric composition, and a medium for plant growth, making it a critically important provider of ecosystem services.[19] Since soil has a tremendous range of available niches and habitats, it contains most of the Earth's genetic diversity. A gram of soil can contain billions of organisms, belonging to thousands of species, mostly microbial and largely still unexplored.[20][21] Soil has a mean prokaryotic density of roughly 108 organisms per gram,[22] whereas the ocean has no more than 107 prokaryotic organisms per milliliter (gram) of seawater.[23] Organic carbon held in soil is eventually returned to the atmosphere through the process of respiration carried out by heterotrophic organisms, but a substantial part is retained in the soil in the form of soil organic matter; tillage usually increases the rate of soil respiration, leading to the depletion of soil organic matter.[24] Since plant roots need oxygen, ventilation is an important characteristic of soil. This ventilation can be accomplished via networks of interconnected soil pores, which also absorb and hold rainwater making it readily available for uptake by plants. Since plants require a nearly continuous supply of water, but most regions receive sporadic rainfall, the water-holding capacity of soils is vital for plant survival.[25]
Soils can effectively remove impurities,[26] kill disease agents,[27] and degrade contaminants, this latter property being called natural attenuation.[28] Typically, soils maintain a net absorption of oxygen and methane and undergo a net release of carbon dioxide and nitrous oxide.[29] Soils offer plants physical support, air, water, temperature moderation, nutrients, and protection from toxins.[30] Soils provide readily available nutrients to plants and animals by converting dead organic matter into various nutrient forms.[31]
Composition
Components of a loam soil by percent volume
A typical soil is about 50% solids (45% mineral and 5% organic matter), and 50% voids (or pores) of which half is occupied by water and half by gas.[32] The percent soil mineral and organic content can be treated as a constant (in the short term), while the percent soil water and gas content is considered highly variable whereby a rise in one is simultaneously balanced by a reduction in the other.[33] The pore space allows for the infiltration and movement of air and water, both of which are critical for life existing in soil.[34] Compaction, a common problem with soils, reduces this space, preventing air and water from reaching plant roots and soil organisms.[35]
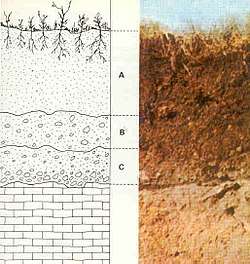
Given sufficient time, an undifferentiated soil will evolve a soil profile which consists of two or more layers, referred to as soil horizons. These differ in one or more properties such as in their texture, structure, density, porosity, consistency, temperature, color, and reactivity.[10] The horizons differ greatly in thickness and generally lack sharp boundaries; their development is dependent on the type of parent material, the processes that modify those parent materials, and the soil-forming factors that influence those processes. The biological influences on soil properties are strongest near the surface, while the geochemical influences on soil properties increase with depth. Mature soil profiles typically include three basic master horizons: A, B, and C. The solum normally includes the A and B horizons. The living component of the soil is largely confined to the solum, and is generally more prominent in the A horizon.[36]
The soil texture is determined by the relative proportions of the individual particles of sand, silt, and clay that make up the soil. The interaction of the individual mineral particles with organic matter, water, gases via biotic and abiotic processes causes those particles to flocculate (stick together) to form aggregates or peds.[37] Where these aggregates can be identified, a soil can be said to be developed, and can be described further in terms of color, porosity, consistency, reaction (acidity), etc.
Water is a critical agent in soil development due to its involvement in the dissolution, precipitation, erosion, transport, and deposition of the materials of which a soil is composed.[38] The mixture of water and dissolved or suspended materials that occupy the soil pore space is called the soil solution. Since soil water is never pure water, but contains hundreds of dissolved organic and mineral substances, it may be more accurately called the soil solution. Water is central to the dissolution, precipitation and leaching of minerals from the soil profile. Finally, water affects the type of vegetation that grows in a soil, which in turn affects the development of the soil, a complex feedback which is exemplified in the dynamics of banded vegetation patterns in semi-arid regions.[39]
Soils supply plants with nutrients, most of which are held in place by particles of clay and organic matter (colloids)[40] The nutrients may be adsorbed on clay mineral surfaces, bound within clay minerals (absorbed), or bound within organic compounds as part of the living organisms or dead soil organic matter. These bound nutrients interact with soil water to buffer the soil solution composition (attenuate changes in the soil solution) as soils wet up or dry out, as plants take up nutrients, as salts are leached, or as acids or alkalis are added.[41][42]
Plant nutrient availability is affected by soil pH, which is a measure of the hydrogen ion activity in the soil solution. Soil pH is a function of many soil forming factors, and is generally lower (more acid) where weathering is more advanced.[43]
Most plant nutrients, with the exception of nitrogen, originate from the minerals that make up the soil parent material. Some nitrogen originates from rain as dilute nitric acid and ammonia,[44] but most of the nitrogen is available in soils as a result of nitrogen fixation by bacteria. Once in the soil-plant system, most nutrients are recycled through living organisms, plant and microbial residues (soil organic matter), mineral-bound forms, and the soil solution. Both living microorganisms and soil organic matter are of critical importance to this recycling, and thereby to soil formation and soil fertility.[45] Microbial activity in soils may release nutrients from minerals or organic matter for use by plants and other microorganisms, sequester (incorporate) them into living cells, or cause their loss from the soil by volatilisation (loss to the atmosphere as gases) or leaching.
Formation
Soil formation, or pedogenesis, is the combined effect of physical, chemical, biological and anthropogenic processes working on soil parent material. Soil is said to be formed when organic matter has accumulated and colloids are washed downward, leaving deposits of clay, humus, iron oxide, carbonate, and gypsum, producing a distinct layer called the B horizon. This is a somewhat arbitrary definition as mixtures of sand, silt, clay and humus will support biological and agricultural activity before that time. These constituents are moved from one level to another by water and animal activity. As a result, layers (horizons) form in the soil profile. The alteration and movement of materials within a soil causes the formation of distinctive soil horizons. However, more recent definitions of soil embrace soils without any organic matter, such as those regoliths that formed on Mars[46] and analogous conditions in planet Earth deserts.[47]
An example of the development of a soil would begin with the weathering of lava flow bedrock, which would produce the purely mineral-based parent material from which the soil texture forms. Soil development would proceed most rapidly from bare rock of recent flows in a warm climate, under heavy and frequent rainfall. Under such conditions, plants (in a first stage nitrogen-fixing lichens and cyanobacteria then epilithic higher plants) become established very quickly on basaltic lava, even though there is very little organic material. The plants are supported by the porous rock as it is filled with nutrient-bearing water that carries minerals dissolved from the rocks. Crevasses and pockets, local topography of the rocks, would hold fine materials and harbour plant roots. The developing plant roots are associated with mineral-weathering mycorrhizal fungi[48] that assist in breaking up the porous lava, and by these means organic matter and a finer mineral soil accumulate with time. Such initial stages of soil development have been described on volcanoes,[49] inselbergs,[50] and glacial moraines.[51]
How soil formation proceeds is influenced by at least five classic factors that are intertwined in the evolution of a soil. They are: parent material, climate, topography (relief), organisms, and time.[52] When reordered to climate, relief, organisms, parent material, and time, they form the acronym CROPT.[53]
Physical properties
The physical properties of soils, in order of decreasing importance for ecosystem services such as crop production, are texture, structure, bulk density, porosity, consistency, temperature, colour and resistivity.[54] Soil texture is determined by the relative proportion of the three kinds of soil mineral particles, called soil separates: sand, silt, and clay. At the next larger scale, soil structures called peds or more commonly soil aggregates are created from the soil separates when iron oxides, carbonates, clay, silica and humus, coat particles and cause them to adhere into larger, relatively stable secondary structures.[55] Soil bulk density, when determined at standardized moisture conditions, is an estimate of soil compaction.[56] Soil porosity consists of the void part of the soil volume and is occupied by gases or water. Soil consistency is the ability of soil materials to stick together. Soil temperature and colour are self-defining. Resistivity refers to the resistance to conduction of electric currents and affects the rate of corrosion of metal and concrete structures which are buried in soil.[57] These properties vary through the depth of a soil profile, i.e. through soil horizons. Most of these properties determine the aeration of the soil and the ability of water to infiltrate and to be held within the soil.[58]
Soil moisture
Soil moisture refers to the water content of the soil. It can be expressed in terms of volumes or weights. Soil moisture measurement can be based on in situ probes or remote sensing methods.
Water that enters a field is removed from a field by runoff, drainage, evaporation or transpiration.[59] Runoff is the water that flows on the surface to the edge of the field; drainage is the water that flows through the soil downward or toward the edge of the field underground; evaporative water loss from a field is that part of the water that evaporates into the atmosphere directly from the field's surface; transpiration is the loss of water from the field by its evaporation from the plant itself.
Water affects soil formation, structure, stability and erosion but is of primary concern with respect to plant growth.[60] Water is essential to plants for four reasons:
- It constitutes 80%-95% of the plant's protoplasm.
- It is essential for photosynthesis.
- It is the solvent in which nutrients are carried to, into and throughout the plant.
- It provides the turgidity by which the plant keeps itself in proper position.[61]
In addition, water alters the soil profile by dissolving and re-depositing minerals, often at lower levels.[62] In a loam soil, solids constitute half the volume, gas one-quarter of the volume, and water one-quarter of the volume[32] of which only half will be available to most plants, with a strong variation according to matric potential.[63]
A flooded field will drain the gravitational water under the influence of gravity until water's adhesive and cohesive forces resist further drainage at which point it is said to have reached field capacity.[64] At that point, plants must apply suction[64][65] to draw water from a soil. The water that plants may draw from the soil is called the available water.[64][66] Once the available water is used up the remaining moisture is called unavailable water as the plant cannot produce sufficient suction to draw that water in. At 15 bar suction, wilting point, seeds will not germinate,[67][64][68] plants begin to wilt and then die. Water moves in soil under the influence of gravity, osmosis and capillarity.[69] When water enters the soil, it displaces air from interconnected macropores by buoyancy, and breaks aggregates into which air is entrapped, a process called slaking.[70]
The rate at which a soil can absorb water depends on the soil and its other conditions. As a plant grows, its roots remove water from the largest pores (macropores) first. Soon the larger pores hold only air, and the remaining water is found only in the intermediate- and smallest-sized pores (micropores). The water in the smallest pores is so strongly held to particle surfaces that plant roots cannot pull it away. Consequently, not all soil water is available to plants, with a strong dependence on texture.[71] When saturated, the soil may lose nutrients as the water drains.[72] Water moves in a draining field under the influence of pressure where the soil is locally saturated and by capillarity pull to drier parts of the soil.[73] Most plant water needs are supplied from the suction caused by evaporation from plant leaves (transpiration) and a lower fraction is supplied by suction created by osmotic pressure differences between the plant interior and the soil solution.[74][75] Plant roots must seek out water and grow preferentially in moister soil microsites,[76] but some parts of the root system are also able to remoisten dry parts of the soil.[77] Insufficient water will damage the yield of a crop.[78] Most of the available water is used in transpiration to pull nutrients into the plant.[79]
Soil water is also important for climate modeling and numerical weather prediction. Global Climate Observing System specified soil water as one of the 50 Essential Climate Variables (ECVs).[80] Soil water can be measured in situ with soil moisture sensor or can be estimated from satellite data and hydrological models. Each method exhibits pros and cons, and hence, the integration of different techniques may decrease the drawbacks of a single given method.[81]
Water retention
Water is retained in a soil when the adhesive force of attraction that water's hydrogen atoms have for the oxygen of soil particles is stronger than the cohesive forces that water's hydrogen feels for other water oxygen atoms.[82] When a field is flooded, the soil pore space is completely filled by water. The field will drain under the force of gravity until it reaches what is called field capacity, at which point the smallest pores are filled with water and the largest with water and gases.[83] The total amount of water held when field capacity is reached is a function of the specific surface area of the soil particles.[84] As a result, high clay and high organic soils have higher field capacities.[85] The potential energy of water per unit volume relative to pure water in reference conditions is called water potential. Total water potential is a sum of matric potential which results from capillary action, osmotic potential for saline soil, and gravitational potential when dealing with vertical direction of water movement. Water potential in soil usually has negative values, and therefore it is also expressed in suction, which is defined as the minus of water potential. Suction has a positive value and can be regarded as the total force required to pull or push water out of soil. Water potential or suction is expressed in units of kPa (103 pascal), bar (100 kPa), or cm H2O (approximately 0.098 kPa). Common logarithm of suction in cm H2O is called pF.[86] Therefore, pF 3 = 1000 cm = 98 kPa = 0.98 bar.
The forces with which water is held in soils determine its availability to plants. Forces of adhesion hold water strongly to mineral and humus surfaces and less strongly to itself by cohesive forces. A plant's root may penetrate a very small volume of water that is adhering to soil and be initially able to draw in water that is only lightly held by the cohesive forces. But as the droplet is drawn down, the forces of adhesion of the water for the soil particles produce increasingly higher suction, finally up to 1500 kPa (pF = 4.2).[87] At 1500 kPa suction, the soil water amount is called wilting point. At that suction the plant cannot sustain its water needs as water is still being lost from the plant by transpiration, the plant's turgidity is lost, and it wilts, although stomatal closure may decrease transpiration and thus may retard wilting below the wilting point, in particular under adaptation or acclimatization to drought.[88] The next level, called air-dry, occurs at 100,000 kPa suction (pF = 6). Finally the oven dry condition is reached at 1,000,000 kPa suction (pF = 7). All water below wilting point is called unavailable water.[89]
When the soil moisture content is optimal for plant growth, the water in the large and intermediate size pores can move about in the soil and be easily used by plants.[71] The amount of water remaining in a soil drained to field capacity and the amount that is available are functions of the soil type. Sandy soil will retain very little water, while clay will hold the maximum amount.[85] The available water for the silt loam might be 20% whereas for the sand it might be only 6% by volume, as shown in this table.
| Soil Texture | Wilting Point | Field Capacity | Available water |
|---|---|---|---|
| Sand | 3.3 | 9.1 | 5.8 |
| Sandy loam | 9.5 | 20.7 | 11.2 |
| Loam | 11.7 | 27.0 | 15.3 |
| Silt loam | 13.3 | 33.0 | 19.7 |
| Clay loam | 19.7 | 31.8 | 12.1 |
| Clay | 27.2 | 39.6 | 12.4 |
The above are average values for the soil textures.
Water flow
Water moves through soil due to the force of gravity, osmosis and capillarity. At zero to 33 kPa suction (field capacity), water is pushed through soil from the point of its application under the force of gravity and the pressure gradient created by the pressure of the water; this is called saturated flow. At higher suction, water movement is pulled by capillarity from wetter toward drier soil. This is caused by water's adhesion to soil solids, and is called unsaturated flow.[91][92]
Water infiltration and movement in soil is controlled by six factors:
- Soil texture
- Soil structure. Fine-textured soils with granular structure are most favourable to infiltration of water.
- The amount of organic matter. Coarse matter is best and if on the surface helps prevent the destruction of soil structure and the creation of crusts.
- Depth of soil to impervious layers such as hardpans or bedrock
- The amount of water already in the soil
- Soil temperature. Warm soils take in water faster while frozen soils may not be able to absorb depending on the type of freezing.[93]
Water infiltration rates range from 0.25 cm per hour for high clay soils to 2.5 cm per hour for sand and well stabilized and aggregated soil structures.[94] Water flows through the ground unevenly, in the form of so-called "gravity fingers", because of the surface tension between water particles.[95][96]
Tree roots, whether living or dead, create preferential channels for rainwater flow through soil,[97] magnifying infiltration rates of water up to 27 times.[98]
Flooding temporarily increases soil permeability in river beds, helping to recharge aquifers.[99]
Water applied to a soil is pushed by pressure gradients from the point of its application where it is saturated locally, to less saturated areas, such as the vadose zone.[100][101] Once soil is completely wetted, any more water will move downward, or percolate out of the range of plant roots, carrying with it clay, humus, nutrients, primarily cations, and various contaminants, including pesticides, pollutants, viruses and bacteria, potentially causing groundwater contamination.[102][103] In order of decreasing solubility, the leached nutrients are:
- Calcium
- Magnesium, Sulfur, Potassium; depending upon soil composition
- Nitrogen; usually little, unless nitrate fertiliser was applied recently
- Phosphorus; very little as its forms in soil are of low solubility.[104]
In the United States percolation water due to rainfall ranges from almost zero centimeters just east of the Rocky Mountains to fifty or more centimeters per day in the Appalachian Mountains and the north coast of the Gulf of Mexico.[105]
Water is pulled by capillary action due to the adhesion force of water to the soil solids, producing a suction gradient from wet towards drier soil[106] and from macropores to micropores. The so-called Richards equation allows calculation of the time rate of change of moisture content in soils due to the movement of water in unsaturated soils.[107] Interestingly, this equation attributed to Richards was originally published by Richardson in 1922.[108] The Soil Moisture Velocity Equation,[109] which can be solved using the finite water-content vadose zone flow method,[110][111] describes the velocity of flowing water through an unsaturated soil in the vertical direction. The numerical solution of the Richardson/Richards equation allows calculation of unsaturated water flow and solute transport using software such as Hydrus,[112] by giving soil hydraulic parameters of hydraulic functions (water retention function and unsaturated hydraulic conductivity function) and initial and boundary conditions. Preferential flow occurs along interconnected macropores, crevices, root and worm channels, which drain water under gravity.[113][114] Many models based on soil physics now allow for some representation of preferential flow as a dual continuum, dual porosity or dual permeability options, but these have generally been "bolted on" to the Richards solution without any rigorous physical underpinning.[115]
Water uptake by plants
Of equal importance to the storage and movement of water in soil is the means by which plants acquire it and their nutrients. Most soil water is taken up by plants as passive absorption caused by the pulling force of water evaporating (transpiring) from the long column of water (xylem sap flow) that leads from the plant's roots to its leaves, according to the cohesion-tension theory.[116] The upward movement of water and solutes (hydraulic lift) is regulated in the roots by the endodermis[117] and in the plant foliage by stomatal conductance,[118] and can be interrupted in root and shoot xylem vessels by cavitation, also called xylem embolism.[119] In addition, the high concentration of salts within plant roots creates an osmotic pressure gradient that pushes soil water into the roots.[120] Osmotic absorption becomes more important during times of low water transpiration caused by lower temperatures (for example at night) or high humidity, and the reverse occurs under high temperature or low humidity. It is these process that cause guttation and wilting, respectively.[121][122]
Root extension is vital for plant survival. A study of a single winter rye plant grown for four months in one cubic foot (0.0283 cubic meters) of loam soil showed that the plant developed 13,800,000 roots, a total of 620 km in length with 237 square meters in surface area; and 14 billion hair roots of 10,620 km total length and 400 square meters total area; for a total surface area of 638 square meters. The total surface area of the loam soil was estimated to be 52,000 square meters.[123] In other words, the roots were in contact with only 1.2% of the soil. However, root extension should be viewed as a dynamic process, allowing new roots to explore a new volume of soil each day, increasing dramatically the total volume of soil explored over a given growth period, and thus the volume of water taken up by the root system over this period.[124] Root architecture, i.e. the spatial configuration of the root system, plays a prominent role in the adaptation of plants to soil water and nutrient availabiity, and thus in plant productivity.[125]
Roots must seek out water as the unsaturated flow of water in soil can move only at a rate of up to 2.5 cm per day; as a result they are constantly dying and growing as they seek out high concentrations of soil moisture.[126] Insufficient soil moisture, to the point of causing wilting, will cause permanent damage and crop yields will suffer. When grain sorghum was exposed to soil suction as low as 1300 kPa during the seed head emergence through bloom and seed set stages of growth, its production was reduced by 34%.[127]
Consumptive use and water use efficiency
Only a small fraction (0.1% to 1%) of the water used by a plant is held within the plant. The majority is ultimately lost via transpiration, while evaporation from the soil surface is also substantial, the transpiration:evaporation ratio varying according to vegetation type and climate, peaking in tropical rainforests and dipping in steppes and deserts.[128] Transpiration plus evaporative soil moisture loss is called evapotranspiration. Evapotranspiration plus water held in the plant totals to consumptive use, which is nearly identical to evapotranspiration.[127][129]
The total water used in an agricultural field includes surface runoff, drainage and consumptive use. The use of loose mulches will reduce evaporative losses for a period after a field is irrigated, but in the end the total evaporative loss (plant plus soil) will approach that of an uncovered soil, while more water is immediately available for plant growth.[130] Water use efficiency is measured by the transpiration ratio, which is the ratio of the total water transpired by a plant to the dry weight of the harvested plant. Transpiration ratios for crops range from 300 to 700. For example, alfalfa may have a transpiration ratio of 500 and as a result 500 kilograms of water will produce one kilogram of dry alfalfa.[131]
Soil gas
The atmosphere of soil, or soil gas, is very different from the atmosphere above. The consumption of oxygen by microbes and plant roots, and their release of carbon dioxide, decrease oxygen and increase carbon dioxide concentration. Atmospheric CO2 concentration is 0.04%, but in the soil pore space it may range from 10 to 100 times that level, thus potentially contributing to the inhibition of root respiration.[132] Calcareous soils regulate CO2 concentration by carbonate buffering, contrary to acid soils in which all CO2 respired accumulates in the soil pore system.[133] At extreme levels CO2 is toxic.[134] This suggests a possible negative feedback control of soil CO2 concentration through its inhibitory effects on root and microbial respiration (also called 'soil respiration').[135] In addition, the soil voids are saturated with water vapour, at least until the point of maximal hygroscopicity, beyond which a vapour-pressure deficit occurs in the soil pore space.[34] Adequate porosity is necessary, not just to allow the penetration of water, but also to allow gases to diffuse in and out. Movement of gases is by diffusion from high concentrations to lower, the diffusion coefficient decreasing with soil compaction.[136] Oxygen from above atmosphere diffuses in the soil where it is consumed and levels of carbon dioxide in excess of above atmosphere diffuse out with other gases (including greenhouse gases) as well as water.[137] Soil texture and structure strongly affect soil porosity and gas diffusion. It is the total pore space (porosity) of soil, not the pore size, and the degree of pore interconnection (or conversely pore sealing), together with water content, air turbulence and temperature, that determine the rate of diffusion of gases into and out of soil.[138][137] Platy soil structure and soil compaction (low porosity) impede gas flow, and a deficiency of oxygen may encourage anaerobic bacteria to reduce (strip oxygen) from nitrate NO3 to the gases N2, N2O, and NO, which are then lost to the atmosphere, thereby depleting the soil of nitrogen.[139] Aerated soil is also a net sink of methane CH4[140] but a net producer of methane (a strong heat-absorbing greenhouse gas) when soils are depleted of oxygen and subject to elevated temperatures.[141]
Soil atmosphere is also the seat of emissions of volatiles other than carbon and nitrogen oxides from various soil organisms, e.g. roots,[142] bacteria,[143] fungi,[144] animals.[145] These volatiles are used as chemical cues, making soil atmosphere the seat of interaction networks[146][147] playing a decisive role in the stability, dynamics and evolution of soil ecosystems.[148] Biogenic soil volatile organic compounds are exchanged with the aboveground atmosphere, in which they are just 1–2 orders of magnitude lower than those from aboveground vegetation.[149]
We humans can get some idea of the soil atmosphere through the well-known 'after-the-rain' scent, when infiltering rainwater flushes out the whole soil atmosphere after a drought period, or when soil is excavated,[150] a bulk property attributed in a reductionist manner to particular biochemical compounds such as petrichor or geosmin.
Solid phase (soil matrix)
Soil particles can be classified by their chemical composition (mineralogy) as well as their size. The particle size distribution of a soil, its texture, determines many of the properties of that soil, in particular hydraulic conductivity and water potential,[151] but the mineralogy of those particles can strongly modify those properties. The mineralogy of the finest soil particles, clay, is especially important.[152]
Chemistry
The chemistry of a soil determines its ability to supply available plant nutrients and affects its physical properties and the health of its living population. In addition, a soil's chemistry also determines its corrosivity, stability, and ability to absorb pollutants and to filter water. It is the surface chemistry of mineral and organic colloids that determines soil's chemical properties.[153] A colloid is a small, insoluble particle ranging in size from 1 nanometer to 1 micrometer, thus small enough to remain suspended by Brownian motion in a fluid medium without settling.[154] Most soils contain organic colloidal particles called humus as well as the inorganic colloidal particles of clays. The very high specific surface area of colloids and their net electrical charges give soil its ability to hold and release ions. Negatively charged sites on colloids attract and release cations in what is referred to as cation exchange. Cation-exchange capacity (CEC) is the amount of exchangeable cations per unit weight of dry soil and is expressed in terms of milliequivalents of positively charged ions per 100 grams of soil (or centimoles of positive charge per kilogram of soil; cmolc/kg). Similarly, positively charged sites on colloids can attract and release anions in the soil giving the soil anion exchange capacity (AEC).
Cation and anion exchange
The cation exchange, that takes place between colloids and soil water, buffers (moderates) soil pH, alters soil structure, and purifies percolating water by adsorbing cations of all types, both useful and harmful.
The negative or positive charges on colloid particles make them able to hold cations or anions, respectively, to their surfaces. The charges result from four sources.[155]
- Isomorphous substitution occurs in clay during its formation, when lower-valence cations substitute for higher-valence cations in the crystal structure.[156] Substitutions in the outermost layers are more effective than for the innermost layers, as the electric charge strength drops off as the square of the distance. The net result is oxygen atoms with net negative charge and the ability to attract cations.
- Edge-of-clay oxygen atoms are not in balance ionically as the tetrahedral and octahedral structures are incomplete.[157]
- Hydroxyls may substitute for oxygens of the silica layers, a process called hydroxylation. When the hydrogens of the clay hydroxyls are ionised into solution, they leave the oxygen with a negative charge (anionic clays).[158]
- Hydrogens of humus hydroxyl groups may also be ionised into solution, leaving, similarly to clay, an oxygen with a negative charge.[159]
Cations held to the negatively charged colloids resist being washed downward by water and out of reach of plants' roots, thereby preserving the fertility of soils in areas of moderate rainfall and low temperatures.[160][161]
There is a hierarchy in the process of cation exchange on colloids, as they differ in the strength of adsorption by the colloid and hence their ability to replace one another (ion exchange). If present in equal amounts in the soil water solution:
Al3+ replaces H+ replaces Ca2+ replaces Mg2+ replaces K+ same as NH4+ replaces Na+[162]
If one cation is added in large amounts, it may replace the others by the sheer force of its numbers. This is called law of mass action. This is largely what occurs with the addition of cationic fertilisers (potash, lime).[163]
As the soil solution becomes more acidic (low pH, meaning an abundance of H+, the other cations more weakly bound to colloids are pushed into solution as hydrogen ions occupy exchange sites (protonation). A low pH may cause hydrogen of hydroxyl groups to be pulled into solution, leaving charged sites on the colloid available to be occupied by other cations. This ionisation of hydroxyl groups on the surface of soil colloids creates what is described as pH-dependent surface charges.[164] Unlike permanent charges developed by isomorphous substitution, pH-dependent charges are variable and increase with increasing pH.[42] Freed cations can be made available to plants but are also prone to be leached from the soil, possibly making the soil less fertile.[165] Plants are able to excrete H+ into the soil through the synthesis of organic acids and by that means, change the pH of the soil near the root and push cations off the colloids, thus making those available to the plant.[166]
Cation exchange capacity (CEC)
Cation exchange capacity should be thought of as the soil's ability to remove cations from the soil water solution and sequester those to be exchanged later as the plant roots release hydrogen ions to the solution. CEC is the amount of exchangeable hydrogen cation (H+) that will combine with 100 grams dry weight of soil and whose measure is one milliequivalents per 100 grams of soil (1 meq/100 g). Hydrogen ions have a single charge and one-thousandth of a gram of hydrogen ions per 100 grams dry soil gives a measure of one milliequivalent of hydrogen ion. Calcium, with an atomic weight 40 times that of hydrogen and with a valence of two, converts to (40/2) x 1 milliequivalent = 20 milliequivalents of hydrogen ion per 100 grams of dry soil or 20 meq/100 g.[167] The modern measure of CEC is expressed as centimoles of positive charge per kilogram (cmol/kg) of oven-dry soil.
Most of the soil's CEC occurs on clay and humus colloids, and the lack of those in hot, humid, wet climates, due to leaching and decomposition, respectively, explains the apparent sterility of tropical soils.[168] Live plant roots also have some CEC, linked to their specific surface area.[169]
| Soil | State | CEC meq/100 g |
|---|---|---|
| Charlotte fine sand | Florida | 1.0 |
| Ruston fine sandy loam | Texas | 1.9 |
| Glouchester loam | New Jersey | 11.9 |
| Grundy silt loam | Illinois | 26.3 |
| Gleason clay loam | California | 31.6 |
| Susquehanna clay loam | Alabama | 34.3 |
| Davie mucky fine sand | Florida | 100.8 |
| Sands | ------ | 1–5 |
| Fine sandy loams | ------ | 5–10 |
| Loams and silt loams | ----- | 5–15 |
| Clay loams | ----- | 15–30 |
| Clays | ----- | over 30 |
| Sesquioxides | ----- | 0–3 |
| Kaolinite | ----- | 3–15 |
| Illite | ----- | 25–40 |
| Montmorillonite | ----- | 60–100 |
| Vermiculite (similar to illite) | ----- | 80–150 |
| Humus | ----- | 100–300 |
Anion exchange capacity (AEC)
Anion exchange capacity should be thought of as the soil's ability to remove anions (e.g. nitrate, phosphate) from the soil water solution and sequester those for later exchange as the plant roots release carbonate anions to the soil water solution. Those colloids which have low CEC tend to have some AEC. Amorphous and sesquioxide clays have the highest AEC,[171] followed by the iron oxides. Levels of AEC are much lower than for CEC, because of the generally higher rate of positively (versus negatively) charged surfaces on soil colloids, to the exception of variable-charge soils.[172] Phosphates tend to be held at anion exchange sites.[173]
Iron and aluminum hydroxide clays are able to exchange their hydroxide anions (OH−) for other anions.[174] The order reflecting the strength of anion adhesion is as follows:
- H2PO4− replaces SO42− replaces NO3− replaces Cl−
The amount of exchangeable anions is of a magnitude of tenths to a few milliequivalents per 100 g dry soil.[170] As pH rises, there are relatively more hydroxyls, which will displace anions from the colloids and force them into solution and out of storage; hence AEC decreases with increasing pH (alkalinity).[175]
Reactivity (pH)
Soil reactivity is expressed in terms of pH and is a measure of the acidity or alkalinity of the soil. More precisely, it is a measure of hydrogen ion concentration in an aqueous solution and ranges in values from 0 to 14 (acidic to basic) but practically speaking for soils, pH ranges from 3.5 to 9.5, as pH values beyond those extremes are toxic to life forms.[176]
At 25 °C an aqueous solution that has a pH of 3.5 has 10−3.5 moles H+ (hydrogen ions) per litre of solution (and also 10−10.5 mole/litre OH−). A pH of 7, defined as neutral, has 10−7 moles of hydrogen ions per litre of solution and also 10−7 moles of OH− per litre; since the two concentrations are equal, they are said to neutralise each other. A pH of 9.5 has 10−9.5 moles hydrogen ions per litre of solution (and also 10−2.5 mole per litre OH−). A pH of 3.5 has one million times more hydrogen ions per litre than a solution with pH of 9.5 (9.5–3.5 = 6 or 106) and is more acidic.[177]
The effect of pH on a soil is to remove from the soil or to make available certain ions. Soils with high acidity tend to have toxic amounts of aluminium and manganese.[178] As a result of a trade-off between toxicity and requirement most nutrients are better available to plants at moderate pH,[179] although most minerals are more soluble in acid soils. Soil organisms are hindered by high acidity, and most agricultural crops do best with mineral soils of pH 6.5 and organic soils of pH 5.5.[180] Given that at low pH toxic metals (e.g. cadmium, zinc, lead) are positively charged as cations and organic pollutants are in non-ionic form, thus both made more available to organisms,[181][182] it has been suggested that plants, animals and microbes commonly living in acid soils are pre-adapted to every kind of pollution, whether of natural or human origin.[183]
In high rainfall areas, soils tend to acidity as the basic cations are forced off the soil colloids by the mass action of hydrogen ions from the rain against those attached to the colloids. High rainfall rates can then wash the nutrients out, leaving the soil inhabited only by those organisms which are particularly efficient to uptake nutrients in very acid conditions, like in tropical rainforests.[184] Once the colloids are saturated with H+, the addition of any more hydrogen ions or aluminum hydroxyl cations drives the pH even lower (more acidic) as the soil has been left with no buffering capacity.[185] In areas of extreme rainfall and high temperatures, the clay and humus may be washed out, further reducing the buffering capacity of the soil.[186] In low rainfall areas, unleached calcium pushes pH to 8.5 and with the addition of exchangeable sodium, soils may reach pH 10.[187] Beyond a pH of 9, plant growth is reduced.[188] High pH results in low micro-nutrient mobility, but water-soluble chelates of those nutrients can correct the deficit.[189] Sodium can be reduced by the addition of gypsum (calcium sulphate) as calcium adheres to clay more tightly than does sodium causing sodium to be pushed into the soil water solution where it can be washed out by an abundance of water.[190][191]
Base saturation percentage
There are acid-forming cations (e.g. hydrogen, aluminium, iron) and there are base-forming cations (e.g. calcium, magnesium, sodium). The fraction of the negatively-charged soil colloid exchange sites (CEC) that are occupied by base-forming cations is called base saturation. If a soil has a CEC of 20 meq and 5 meq are aluminium and hydrogen cations (acid-forming), the remainder of positions on the colloids (20-5 = 15 meq) are assumed occupied by base-forming cations, so that the base saturation is 15/20 x 100% = 75% (the compliment 25% is assumed acid-forming cations or protons). Base saturation is almost in direct proportion to pH (it increases with increasing pH).[192] It is of use in calculating the amount of lime needed to neutralise an acid soil (lime requirement). The amount of lime needed to neutralize a soil must take account of the amount of acid forming ions on the colloids (exchangeable acidity), not just those in the soil water solution (free acidity).[193] The addition of enough lime to neutralize the soil water solution will be insufficient to change the pH, as the acid forming cations stored on the soil colloids will tend to restore the original pH condition as they are pushed off those colloids by the calcium of the added lime.[194]
Buffering
The resistance of soil to change in pH, as a result of the addition of acid or basic material, is a measure of the buffering capacity of a soil and (for a particular soil type) increases as the CEC increases. Hence, pure sand has almost no buffering ability, while soils high in colloids (whether mineral or organic) have high buffering capacity.[195] Buffering occurs by cation exchange and neutralisation. However, colloids are not the only regulators of soil pH. The role of carbonates should be underlined, too.[196] More generally, according to pH levels, several buffer systems take precedence over each other, from calcium carbonate buffer range to iron buffer range.[197]
The addition of a small amount of highly basic aqueous ammonia to a soil will cause the ammonium to displace hydrogen ions from the colloids, and the end product is water and colloidally fixed ammonium, but little permanent change overall in soil pH.
The addition of a small amount of lime, Ca(OH)2, will displace hydrogen ions from the soil colloids, causing the fixation of calcium to colloids and the evolution of CO2 and water, with little permanent change in soil pH.
The above are examples of the buffering of soil pH. The general principal is that an increase in a particular cation in the soil water solution will cause that cation to be fixed to colloids (buffered) and a decrease in solution of that cation will cause it to be withdrawn from the colloid and moved into solution (buffered). The degree of buffering is often related to the CEC of the soil; the greater the CEC, the greater the buffering capacity of the soil.[198]
Nutrients
Seventeen elements or nutrients are essential for plant growth and reproduction. They are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), potassium (K), sulfur (S), calcium (Ca), magnesium (Mg), iron (Fe), boron (B), manganese (Mn), copper (Cu), zinc (Zn), molybdenum (Mo), nickel (Ni) and chlorine (Cl).[199][200][201] Nutrients required for plants to complete their life cycle are considered essential nutrients. Nutrients that enhance the growth of plants but are not necessary to complete the plant's life cycle are considered non-essential. With the exception of carbon, hydrogen and oxygen, which are supplied by carbon dioxide and water, and nitrogen, provided through nitrogen fixation,[201] the nutrients derive originally from the mineral component of the soil. The Law of the Minimum expresses that when the available form of a nutrient is not in enough proportion in the soil solution, then other nutrients cannot be taken up at an optimum rate by a plant.[202] A particular nutrient ratio of the soil solution is thus mandatory for optimizing plant growth, a value which might differ from nutrient ratios calculated from plant composition.[203]
Plant uptake of nutrients can only proceed when they are present in a plant-available form. In most situations, nutrients are absorbed in an ionic form from (or together with) soil water. Although minerals are the origin of most nutrients, and the bulk of most nutrient elements in the soil is held in crystalline form within primary and secondary minerals, they weather too slowly to support rapid plant growth. For example, the application of finely ground minerals, feldspar and apatite, to soil seldom provides the necessary amounts of potassium and phosphorus at a rate sufficient for good plant growth, as most of the nutrients remain bound in the crystals of those minerals.[204]
The nutrients adsorbed onto the surfaces of clay colloids and soil organic matter provide a more accessible reservoir of many plant nutrients (e.g. K, Ca, Mg, P, Zn). As plants absorb the nutrients from the soil water, the soluble pool is replenished from the surface-bound pool. The decomposition of soil organic matter by microorganisms is another mechanism whereby the soluble pool of nutrients is replenished – this is important for the supply of plant-available N, S, P, and B from soil.[205]
Gram for gram, the capacity of humus to hold nutrients and water is far greater than that of clay minerals, most of the soil cation exchange capacity arising from charged carboxylic groups on organic matter.[206] However, despite the great capacity of humus to retain water once water-soaked, its high hydrophobicity decreases its wettability.[207] All in all, small amounts of humus may remarkably increase the soil's capacity to promote plant growth.[208][205]
| Element | Symbol | Ion or molecule |
|---|---|---|
| Carbon | C | CO2 (mostly through leaves) |
| Hydrogen | H | H+, HOH (water) |
| Oxygen | O | O2−, OH −, CO32−, SO42−, CO2 |
| Phosphorus | P | H2PO4 −, HPO42− (phosphates) |
| Potassium | K | K+ |
| Nitrogen | N | NH4+, NO3 − (ammonium, nitrate) |
| Sulfur | S | SO42− |
| Calcium | Ca | Ca2+ |
| Iron | Fe | Fe2+, Fe3+ (ferrous, ferric) |
| Magnesium | Mg | Mg2+ |
| Boron | B | H3BO3, H2BO3 −, B(OH)4 − |
| Manganese | Mn | Mn2+ |
| Copper | Cu | Cu2+ |
| Zinc | Zn | Zn2+ |
| Molybdenum | Mo | MoO42− (molybdate) |
| Chlorine | Cl | Cl − (chloride) |
Uptake processes
Nutrients in the soil are taken up by the plant through its roots, and in particular its root hairs. To be taken up by a plant, a nutrient element must be located near the root surface; however, the supply of nutrients in contact with the root is rapidly depleted within a distance of ca. 2 mm.[210] There are three basic mechanisms whereby nutrient ions dissolved in the soil solution are brought into contact with plant roots:
- Mass flow of water
- Diffusion within water
- Interception by root growth
All three mechanisms operate simultaneously, but one mechanism or another may be most important for a particular nutrient.[211] For example, in the case of calcium, which is generally plentiful in the soil solution, except when aluminium over competes calcium on cation exchange sites in very acid soils (pH less than 4),[212] mass flow alone can usually bring sufficient amounts to the root surface. However, in the case of phosphorus, diffusion is needed to supplement mass flow. For the most part, nutrient ions must travel some distance in the soil solution to reach the root surface. This movement can take place by mass flow, as when dissolved nutrients are carried along with the soil water flowing toward a root that is actively drawing water from the soil. In this type of movement, the nutrient ions are somewhat analogous to leaves floating down a stream. In addition, nutrient ions continually move by diffusion from areas of greater concentration toward the nutrient-depleted areas of lower concentration around the root surface. That process is due to random motion, also called Brownian motion, of molecules within a gradient of decreasing concentration.[213] By this means, plants can continue to take up nutrients even at night, when water is only slowly absorbed into the roots as transpiration has almost stopped following stomatal closure. Finally, root interception comes into play as roots continually grow into new, undepleted soil. By this way roots are also able to absorb nanomaterials such as nanoparticulate organic matter.[214]
| Nutrient | Approximate percentage supplied by: | ||
|---|---|---|---|
| Mass flow | Root interception | Diffusion | |
| Nitrogen | 98.8 | 1.2 | 0 |
| Phosphorus | 6.3 | 2.8 | 90.9 |
| Potassium | 20.0 | 2.3 | 77.7 |
| Calcium | 71.4 | 28.6 | 0 |
| Sulfur | 95.0 | 5.0 | 0 |
| Molybdenum | 95.2 | 4.8 | 0 |
In the above table, phosphorus and potassium nutrients move more by diffusion than they do by mass flow in the soil water solution, as they are rapidly taken up by the roots creating a concentration of almost zero near the roots (the plants cannot transpire enough water to draw more of those nutrients near the roots). The very steep concentration gradient is of greater influence in the movement of those ions than is the movement of those by mass flow.[216] The movement by mass flow requires the transpiration of water from the plant causing water and solution ions to also move toward the roots.[217] Movement by root interception is slowest as the plants must extend their roots.[218]
Plants move ions out of their roots in an effort to move nutrients in from the soil, an exchange process which occurs in the root apoplast.[219] Hydrogen H+ is exchanged for other cations, and carbonate (HCO3−) and hydroxide (OH−) anions are exchanged for nutrient anions.[220] As plant roots remove nutrients from the soil water solution, they are replenished as other ions move off of clay and humus (by ion exchange or desorption), are added from the weathering of soil minerals, and are released by the decomposition of soil organic matter. However, the rate at which plant roots remove nutrients may not cope with the rate at which they are replenished in the soil solution, stemming in nutrient limitation to plant growth.[221] Plants derive a large proportion of their anion nutrients from decomposing organic matter, which typically holds about 95 percent of the soil nitrogen, 5 to 60 percent of the soil phosphorus and about 80 percent of the soil sulfur. Where crops are produced, the replenishment of nutrients in the soil must usually be augmented by the addition of fertilizer or organic matter.[215]
Because nutrient uptake is an active metabolic process, conditions that inhibit root metabolism may also inhibit nutrient uptake.[222] Examples of such conditions include waterlogging or soil compaction resulting in poor soil aeration, excessively high or low soil temperatures, and above-ground conditions that result in low translocation of sugars to plant roots.[223]
Carbon

Plants obtain their carbon from atmospheric carbon dioxide through photosynthetic carboxylation, to which must be added the uptake of dissolved carbon from the soil solution[224] and carbon transfer through mycorrhizal networks.[225] About 45% of a plant's dry mass is carbon; plant residues typically have a carbon to nitrogen ratio (C/N) of between 13:1 and 100:1. As the soil organic material is digested by micro-organisms and saprophagous soil fauna, the C/N decreases as the carbonaceous material is metabolized and carbon dioxide (CO2) is released as a byproduct which then finds its way out of the soil and into the atmosphere. Nitrogen turnover (mostly involved in protein turnover) is lesser than that of carbon (mostly involved in respiration) in the living, then dead matter of decomposers, which are always richer in nitrogen than plant litter, and so it builds up in the soil.[226] Normal CO2 concentration in the atmosphere is 0.03%, this can be the factor limiting plant growth. In a field of maize on a still day during high light conditions in the growing season, the CO2 concentration drops very low, but under such conditions the crop could use up to 20 times the normal concentration. The respiration of CO2 by soil micro-organisms decomposing soil organic matter and the CO2 respired by roots contribute an important amount of CO2 to the photosynthesising plants, to which must be added the CO2 respired by aboveground plant tissues.[227] Root-respired CO2 can be accumulated overnight within hollow stems of plants, to be further used for photosynthesis during the day.[228] Within the soil, CO2 concentration is 10 to 100 times that of atmospheric levels but may rise to toxic levels if the soil porosity is low or if diffusion is impeded by flooding.[229][199][230]
Nitrogen
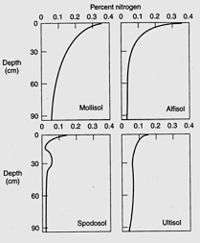
Nitrogen is the most critical element obtained by plants from the soil, to the exception of moist tropical forests where phosphorus is the limiting soil nutrient,[231] and nitrogen deficiency often limits plant growth.[232] Plants can use the nitrogen as either the ammonium cation (NH4+) or the anion nitrate (NO3−). Plants are commonly classified as ammonium or nitrate plants according to their preferential nitrogen nutrition.[233] Usually, most of the nitrogen in soil is bound within organic compounds that make up the soil organic matter, and must be mineralized to the ammonium or nitrate form before it can be taken up by most plants. However, symbiosis with mycorrhizal fungi allow plants to get access to the organic nitrogen pool where and when mineral forms of nitrogen are poorly available.[234] The total nitrogen content depends largely on the soil organic matter content, which in turn depends on texture, climate, vegetation, topography, age and soil management.[235] Soil nitrogen typically decreases by 0.2 to 0.3% for every temperature increase by 10 °C. Usually, grassland soils contain more soil nitrogen than forest soils, because of a higher turnover rate of grassland organic matter.[236] Cultivation decreases soil nitrogen by exposing soil organic matter to decomposition by microorganisms,[237] most losses being caused by denitrification,[238] and soils under no-tillage maintain more soil nitrogen than tilled soils.[239]
Some micro-organisms are able to metabolise organic matter and release ammonium in a process called mineralisation. Others, called nitrifiers, take free ammonium or nitrite as an intermediary step in the process of nitrification, and oxidise it to nitrate. Nitrogen-fixing bacteria are capable of metabolising N2 into the form of ammonia or related nitrogenous compounds in a process called nitrogen fixation. Both ammonium and nitrate can be immobilized by their incorporation into microbial living cells, where it is temporarily sequestered in the form of amino acids and proteins. Nitrate may be lost from the soil to the atmosphere when bacteria metabolise it to the gases NH3, N2 and N2O, a process called denitrification. Nitrogen may also be leached from the vadose zone if in the form of nitrate, acting as a pollutant if it reaches the water table or flows over land, more especially in agricultural soils under high use of nutrient fertilizers.[240] Ammonium may also be sequestered in 2:1 clay minerals.[241] A small amount of nitrogen is added to soil by rainfall, to the exception of wide areas of North America and West Europe where the excess use of nitrogen fertilizers and manure has caused atmospheric pollution by ammonia emission, stemming in soil acidification and eutrophication of soils and aquatic ecosystems.[242][243][205][244][245][246]
Gains
In the process of mineralisation, microbes feed on organic matter, releasing ammonia (NH3), ammonium (NH4+), nitrate (NO3−) and other nutrients. As long as the carbon to nitrogen ratio (C/N) of fresh residues in the soil is above 30:1, nitrogen will be in short supply for the nitrogen-rich microbal biomass (nitrogen deficiency), and other bacteria will uptake ammonium and to a lesser extent nitrate and incorporate them into their cells in the immobilization process.[247] In that form the nitrogen is said to be immobilised. Later, when such bacteria die, they too are mineralised and some of the nitrogen is released as ammonium and nitrate. Predation of bacteria by soil fauna, in particular protozoa and nematodes, play a decisive role in the return of immobilized nitrogen to mineral forms.[248] If the C/N of fresh residues is less than 15, mineral nitrogen is freed to the soil and directly available to plants.[249] Bacteria may on average add 25 pounds (11 kg) nitrogen per acre, and in an unfertilised field, this is the most important source of usable nitrogen. In a soil with 5% organic matter perhaps 2 to 5% of that is released to the soil by such decomposition. It occurs fastest in warm, moist, well aerated soil.[250] The mineralisation of 3% of the organic material of a soil that is 4% organic matter overall, would release 120 pounds (54 kg) of nitrogen as ammonium per acre.[251]
| Organic Material | C:N Ratio |
|---|---|
| Alfalfa | 13 |
| Bacteria | 4 |
| Clover, green sweet | 16 |
| Clover, mature sweet | 23 |
| Fungi | 9 |
| Forest litter | 30 |
| Humus in warm cultivated soils | 11 |
| Legume-grass hay | 25 |
| Legumes (alfalfa or clover), mature | 20 |
| Manure, cow | 18 |
| Manure, horse | 16–45 |
| Manure, human | 10 |
| Oat straw | 80 |
| Straw, cornstalks | 90 |
| Sawdust | 250 |
In nitrogen fixation, rhizobium bacteria convert N2 to ammonia (NH3), which is rapidly converted to amino acids, parts of which are used by the rhizobia for the synthesis of their own biomass proteins, while other parts are transported to the xylem of the host plant.[253] Rhizobia share a symbiotic relationship with host plants, since rhizobia supply the host with nitrogen and the host provides rhizobia with other nutrients and a safe environment. It is estimated that such symbiotic bacteria in the root nodules of legumes add 45 to 250 pounds of nitrogen per acre per year, which may be sufficient for the crop. Other, free-living nitrogen-fixing diazotroph bacteria and archaea live independently in the soil and release mineral forms of nitrogen when their dead bodies are converted by way of mineralization.[254]
Some amount of usable nitrogen is fixed by lightning as nitric oxide (NO) and nitrogen dioxide (NO2−).[255] Nitrogen dioxide is soluble in water to form nitric acid (HNO3) dissociating in H+ and NO3−. Ammonia, NH3, previously emitted from the soil, may fall with precipitation as nitric acid at a rate of about five pounds nitrogen per acre per year.[256]
Sequestration
When bacteria feed on soluble forms of nitrogen (ammonium and nitrate), they temporarily sequester that nitrogen in their bodies in a process called immobilization. At a later time when those bacteria die, their nitrogen may be released as ammonium by the process of mineralization, sped up by predatory fauna.[257]
Protein material is easily broken down, but the rate of its decomposition is slowed by its attachment to the crystalline structure of clay and when trapped between the clay layers[258] or attached to rough clay surfaces.[259] The layers are small enough that bacteria cannot enter.[260] Some organisms can exude extracellular enzymes that can act on the sequestered proteins. However, those enzymes too may be trapped on the clay crystals, resulting in a complex interaction between proteins, microbial enzymes and mineral surfaces.[261]
Ammonium fixation occurs mainly between the layers of 2:1 type clay minerals such as illite, vermiculite or montmorillonite, together with ions of similar ionic radius and low hydration energy such as potassium, but a small proportion of ammonium is also fixed in the silt fraction.[262] Only a small fraction of soil nitrogen is held this way.[263]
Losses
Usable nitrogen may be lost from soils when it is in the form of nitrate, as it is easily leached, contrary to ammonium which is easily fixed.[264] Further losses of nitrogen occur by denitrification, the process whereby soil bacteria convert nitrate (NO3−) to nitrogen gas, N2 or N2O. This occurs when poor soil aeration limits free oxygen, forcing bacteria to use the oxygen in nitrate for their respiratory process. Denitrification increases when oxidisable organic material is available, as in organic farming[264] and when soils are warm and slightly acidic, as currently happening in tropical areas.[265] Denitrification may vary throughout a soil as the aeration varies from place to place.[266] Denitrification may cause the loss of 10 to 20 percent of the available nitrates within a day and when conditions are favourable to that process, losses of up to 60 percent of nitrate applied as fertiliser may occur.[267]
Ammonia volatilisation occurs when ammonium reacts chemically with an alkaline soil, converting NH4+ to NH3.[268] The application of ammonium fertiliser to such a field can result in volatilisation losses of as much as 30 percent.[269]
All kinds of nitrogen losses, whether by leaching or volatilization, are responsible for a large part of aquifer pollution[270] and air pollution, with concomitant effects on soil acidification and eutrophication,[271] a novel combination of environmental threats (acidity and excess nitrogen) to which extant organisms are badly adapted, causing severe biodiversity losses in natural ecosystems.[272]
Phosphorus
After nitrogen, phosphorus is probably the element most likely to be deficient in soils, although it often turns to be the most deficient in tropical soils where the mineral pool is depleted under intense leaching and mineral weathering while, contrary to nitrogen, phosphorus reserves cannot be replenished from other sources.[273] The soil mineral apatite is the most common mineral source of phosphorus, from which it can be extracted by microbial and root exudates,[274][275] with an important contribution of arbuscular mycorrhizal fungi.[276] The most common form of organic phosphate is phytate, the principal storage form of phosphorus in many plant tissues. While there is on average 1000 lb per acre (1120 kg per hectare) of phosphorus in the soil, it is generally in the form of orthophosphate with low solubility, except when linked to ammonium or calcium, hence the use of diammonium phosphate or monocalcium phosphate as fertilizers.[277] Total phosphorus is about 0.1 percent by weight of the soil, but only one percent of that is directly available to plants. Of the part available, more than half comes from the mineralisation of organic matter. Agricultural fields may need to be fertilised to make up for the phosphorus that has been removed in the crop.[278]
When phosphorus does form solubilised ions of H2PO4−, if not taken up by plant roots they rapidly form insoluble phosphates of calcium or hydrous oxides of iron and aluminum. Phosphorus is largely immobile in the soil and is not leached but actually builds up in the surface layer if not cropped. The application of soluble fertilisers to soils may result in zinc deficiencies as zinc phosphates form, but soil pH levels, partly depending on the form of phosphorus in the fertiliser, strongly interact with this effect, in some cases resulting in increased zinc availability.[279] Lack of phosphorus may interfere with the normal opening of the plant leaf stomata, decreased stomatal conductance resulting in decreased photosynthesis and respiration rates[280] while decreased transpiration increases plant temperature.[281] Phosphorus is most available when soil pH is 6.5 in mineral soils and 5.5 in organic soils.[269]
Potassium
The amount of potassium in a soil may be as much as 80,000 lb per acre-foot, of which only 150 lb is available for plant growth. Common mineral sources of potassium are the mica biotite and potassium feldspar, KAlSi3O8. Rhizosphere bacteria, also called rhizobacteria, contribute through the production of organic acids to its solubilization.[282] When solubilised, half will be held as exchangeable cations on clay while the other half is in the soil water solution. Potassium fixation often occurs when soils dry and the potassium is bonded between layers of 2:1 expansive clay minerals such as illite, vermiculite or montmorillonite.[283] Under certain conditions, dependent on the soil texture, intensity of drying, and initial amount of exchangeable potassium, the fixed percentage may be as much as 90 percent within ten minutes. Potassium may be leached from soils low in clay.[284][285]
Calcium
Calcium is one percent by weight of soils and is generally available but may be low as it is soluble and can be leached. It is thus low in sandy and heavily leached soil or strongly acidic mineral soils, resulting in excessive concentration of free hydrogen ions in the soil solution, and therefore these soils require liming.[286] Calcium is supplied to the plant in the form of exchangeable ions and moderately soluble minerals. There are four forms of calcium in the soil. Soil calcium can be in insoluble forms such as calcite or dolomite, in the soil solution in the form of a divalent cation or retained in exchangeable form at the surface of mineral particles. Another form is when calcium complexes with organic matter, forming covalent bonds between organic compounds which contribute to structural stability.[287] Calcium is more available on the soil colloids than is potassium because the common mineral calcite, CaCO3, is more soluble than potassium-bearing minerals such as feldspar.[288]
Calcium uptake by roots is essential for plant nutrition, contrary to an old tenet that it was luxury consumption.[289] Calcium is considered as an essential component of plant cell membranes, a counterion for inorganic and organic anions in the vacuole, and an intracellular messenger in the cytosol, playing a role in cellular learning and memory.[290]
Magnesium
Magnesium is one of the dominant exchangeable cations in most soils (after calcium and potassium). Magnesium is an essential element for plants, microbes and animals, being involved in many catalytic reactions and in the synthesis of chlorophyll. Primary minerals that weather to release magnesium include hornblende, biotite and vermiculite. Soil magnesium concentrations are generally sufficient for optimal plant growth, but highly weathered and sandy soils may be magnesium deficient due to leaching by heavy precipitation.[205][291]
Sulfur
Most sulfur is made available to plants, like phosphorus, by its release from decomposing organic matter.[291] Deficiencies may exist in some soils (especially sandy soils) and if cropped, sulfur needs to be added. The application of large quantities of nitrogen to fields that have marginal amounts of sulfur may cause sulfur deficiency by a dilution effect when stimulation of plant growth by nitrogen increases the plant demand for sulfur.[292] A 15-ton crop of onions uses up to 19 lb of sulfur and 4 tons of alfalfa uses 15 lb per acre. Sulfur abundance varies with depth. In a sample of soils in Ohio, United States, the sulfur abundance varied with depths, 0–6 inches, 6–12 inches, 12–18 inches, 18–24 inches in the amounts: 1056, 830, 686, 528 lb per acre respectively.[293]
Micronutrients
The micronutrients essential in plant life, in their order of importance, include iron,[294] manganese,[295] zinc,[296] copper,[297] boron,[298] chlorine[299] and molybdenum.[300] The term refers to plants' needs, not to their abundance in soil. They are required in very small amounts but are essential to plant health in that most are required parts of enzyme systems which are involved in plant metabolism.[301] They are generally available in the mineral component of the soil, but the heavy application of phosphates can cause a deficiency in zinc and iron by the formation of insoluble zinc and iron phosphates.[302] Iron deficiency, stemming in plant chlorosis and rhizosphere acidification, may also result from excessive amounts of heavy metals or calcium minerals (lime) in the soil.[303][304] Excess amounts of soluble boron, molybdenum and chloride are toxic.[305][306]
Non-essential nutrients
Nutrients which enhance the health but whose deficiency does not stop the life cycle of plants include: cobalt, strontium, vanadium, silicon and nickel.[307] As their importance is evaluated they may be added to the list of essential plant nutrients, as is the case for silicon.[308]
Soil organic matter
Soil organic matter is made up of organic compounds and includes plant, animal and microbial material, both living and dead. A typical soil has a biomass composition of 70% microorganisms, 22% macrofauna, and 8% roots. The living component of an acre of soil may include 900 lb of earthworms, 2400 lb of fungi, 1500 lb of bacteria, 133 lb of protozoa and 890 lb of arthropods and algae.[309]
A few percent of the soil organic matter, with small residence time, consists of the microbial biomass and metabolites of bacteria, molds, and actinomycetes that work to break down the dead organic matter.[310][311] Were it not for the action of these micro-organisms, the entire carbon dioxide part of the atmosphere would be sequestered as organic matter in the soil. However, in the same time soil microbes contribute to carbon sequestration in the topsoil through the formation of stable humus.[312] In the aim to sequester more carbon in the soil for alleviating the greenhouse effect it would be more efficient in the long-term to stimulate humification than to decrease litter decomposition.[313]
The main part of soil organic matter is a complex assemblage of small organic molecules, collectively called humus or humic substances. The use of these terms, which do not rely on a clear chemical classification, has been considered as obsolete.[314] Other studies showed that the classical notion of molecule is not convenient for humus, which escaped most attempts done over two centuries to resolve it in unit components, but still is chemically distinct from polysaccharides, lignins and proteins.[315]
Most living things in soils, including plants, animals, bacteria, and fungi, are dependent on organic matter for nutrients and/or energy. Soils have organic compounds in varying degrees of decomposition which rate is dependent on the temperature, soil moisture, and aeration. Bacteria and fungi feed on the raw organic matter, which are fed upon by protozoa, which in turn are fed upon by nematodes, annelids and arthropods, themselves able to consume and transform raw or humified organic matter. This has been called the soil food web, through which all organic matter is processed as in a digestive system.[316] Organic matter holds soils open, allowing the infiltration of air and water, and may hold as much as twice its weight in water. Many soils, including desert and rocky-gravel soils, have little or no organic matter. Soils that are all organic matter, such as peat (histosols), are infertile.[317] In its earliest stage of decomposition, the original organic material is often called raw organic matter. The final stage of decomposition is called humus.
In grassland, much of the organic matter added to the soil is from the deep, fibrous, grass root systems. By contrast, tree leaves falling on the forest floor are the principal source of soil organic matter in the forest. Another difference is the frequent occurrence in the grasslands of fires that destroy large amounts of aboveground material but stimulate even greater contributions from roots. Also, the much greater acidity under any forests inhibits the action of certain soil organisms that otherwise would mix much of the surface litter into the mineral soil. As a result, the soils under grasslands generally develop a thicker A horizon with a deeper distribution of organic matter than in comparable soils under forests, which characteristically store most of their organic matter in the forest floor (O horizon) and thin A horizon.[318]
Humus
Humus refers to organic matter that has been decomposed by soil microflora and fauna to the point where it is resistant to further breakdown. Humus usually constitutes only five percent of the soil or less by volume, but it is an essential source of nutrients and adds important textural qualities crucial to soil health and plant growth.[319] Humus also feeds arthropods, termites and earthworms which further improve the soil.[320] The end product, humus, is suspended in colloidal form in the soil solution and forms a weak acid that can attack silicate minerals.[321] Humus has a high cation and anion exchange capacity that on a dry weight basis is many times greater than that of clay colloids. It also acts as a buffer, like clay, against changes in pH and soil moisture.[322]
Humic acids and fulvic acids, which begin as raw organic matter, are important constituents of humus. After the death of plants, animals, and microbes, microbes begin to feed on the residues through their production of extra-cellular enzymes, resulting finally in the formation of humus.[323] As the residues break down, only molecules made of aliphatic and aromatic hydrocarbons, assembled and stabilized by oxygen and hydrogen bonds, remain in the form of complex molecular assemblages collectively called humus.[315] Humus is never pure in the soil, because it reacts with metals and clays to form complexes which further contribute to its stability and to soil structure.[322] While the structure of humus has in itself few nutrients, to the exception of constitutive metals such as calcium, iron and aluminum, it is able to attract and link by weak bonds cation and anion nutrients that can further be released into the soil solution in response to selective root uptake and changes in soil pH, a process of paramount importance for the maintenance of fertility in tropical soils.[324]
Lignin is resistant to breakdown and accumulates within the soil. It also reacts with proteins,[325] which further increases its resistance to decomposition, including enzymatic decomposition by microbes.[326] Fats and waxes from plant matter have still more resistance to decomposition and persist in soils for thousand years, hence their use as tracers of past vegetation in buried soil layers.[327] Clay soils often have higher organic contents that persist longer than soils without clay as the organic molecules adhere to and are stabilised by the clay.[328] Proteins normally decompose readily, to the exception of scleroproteins, but when bound to clay particles they become more resistant to decomposition.[329] As for other proteins clay particles absorb the enzymes exuded by microbes, decreasing enzyme activity while protecting extracellular enzymes from degradation.[330] The addition of organic matter to clay soils can render that organic matter and any added nutrients inaccessible to plants and microbes for many years, while a study showed increased soil fertility following the addition of mature compost to a clay soil.[331] High soil tannin content can cause nitrogen to be sequestered as resistant tannin-protein complexes.[332][333]
Humus formation is a process dependent on the amount of plant material added each year and the type of base soil. Both are affected by climate and the type of organisms present.[334] Soils with humus can vary in nitrogen content but typically have 3 to 6 percent nitrogen. Raw organic matter, as a reserve of nitrogen and phosphorus, is a vital component affecting soil fertility.[317] Humus also absorbs water, and expands and shrinks between dry and wet states to a higher extent than clay, increasing soil porosity.[335] Humus is less stable than the soil's mineral constituents, as it is reduced by microbial decomposition, and over time its concentration diminishes without the addition of new organic matter. However, humus in its most stable forms may persist over centuries if not millennia.[336] Charcoal is a source of highly stable humus, called black carbon,[337] which had been used traditionally to improve the fertility of nutrient-poor tropical soils. This very ancient practice, as ascertained in the genesis of Amazonian dark earths, has been renewed and became popular under the name of biochar. It has been suggested that biochar could be used to sequester more carbon in the fight against the greenhouse effect.[338]
Climatological influence
The production, accumulation and degradation of organic matter are greatly dependent on climate. Temperature, soil moisture and topography are the major factors affecting the accumulation of organic matter in soils. Organic matter tends to accumulate under wet or cold conditions where decomposer activity is impeded by low temperature[339] or excess moisture which results in anaerobic conditions.[340] Conversely, excessive rain and high temperatures of tropical climates enables rapid decomposition of organic matter and leaching of plant nutrients. Forest ecosystems on these soils rely on efficient recycling of nutrients and plant matter by the living plant and microbial biomass to maintain their productivity, a process which is disturbed by human activities.[341] Excessive slope, in particular in the presence of cultivation for the sake of agriculture, may encourage the erosion of the top layer of soil which holds most of the raw organic material that would otherwise eventually become humus.[342]
Plant residue
Typical types and percentages of plant residue components
Cellulose and hemicellulose undergo fast decomposition by fungi and bacteria, with a half-life of 12–18 days in a temperate climate.[343] Brown rot fungi can decompose the cellulose and hemicellulose, leaving the lignin and phenolic compounds behind. Starch, which is an energy storage system for plants, undergoes fast decomposition by bacteria and fungi. Lignin consists of polymers composed of 500 to 600 units with a highly branched, amorphous structure, linked to cellulose, hemicellulose and pectin in plant cell walls. Lignin undergoes very slow decomposition, mainly by white rot fungi and actinomycetes; its half-life under temperate conditions is about six months.[343]
Horizons
A horizontal layer of the soil, whose physical features, composition and age are distinct from those above and beneath, is referred to as a soil horizon. The naming of a horizon is based on the type of material of which it is composed. Those materials reflect the duration of specific processes of soil formation. They are labelled using a shorthand notation of letters and numbers which describe the horizon in terms of its colour, size, texture, structure, consistency, root quantity, pH, voids, boundary characteristics and presence of nodules or concretions.[344] No soil profile has all the major horizons. Some, called entisols, may have only one horizon or are currently considered as having no horizon, in particular incipient soils from unreclaimed mining waste deposits,[345] moraines,[346] volcanic cones[347] sand dunes or alluvial terraces.[348] Upper soil horizons may be lacking in truncated soils following wind or water ablation, with concomitant downslope burying of soil horizons, a natural process aggravated by agricultural practices such as tillage.[349] The growth of trees is another source of disturbance, creating a micro-scale heterogeneity which is still visible in soil horizons once trees have died.[350] By passing from a horizon to another, from the top to the bottom of the soil profile, one goes back in time, with past events registered in soil horizons like in sediment layers. Sampling pollen, testate amoebae and plant remains in soil horizons may help to reveal environmental changes (e.g. climate change, land use change) which occurred in the course of soil formation.[351] Soil horizons can be dated by several methods such as radiocarbon, using pieces of charcoal provided they are of enough size to escape pedoturbation by earthworm activity and other mechanical disturbances.[352] Fossil soil horizons from paleosols can be found within sedimentary rock sequences, allowing the study of past environments.[353]
The exposure of parent material to favourable conditions produces mineral soils that are marginally suitable for plant growth, as is the case in eroded soils.[354] The growth of vegetation results in the production of organic residues which fall on the ground as litter for plant aerial parts (leaf litter) or are directly produced belowground for subterranean plant organs (root litter), and then release dissolved organic matter.[355] The remaining surficial organic layer, called the O horizon, produces a more active soil due to the effect of the organisms that live within it. Organisms colonise and break down organic materials, making available nutrients upon which other plants and animals can live.[356] After sufficient time, humus moves downward and is deposited in a distinctive organic-mineral surface layer called the A horizon, in which organic matter is mixed with mineral matter through the activity of burrowing animals, a process called pedoturbation. This natural process does not go to completion in the presence of conditions detrimental to soil life such as strong acidity, cold climate or pollution, stemming in the accumulation of undecomposed organic matter within a single organic horizon overlying the mineral soil[357] and in the juxtaposition of humified organic matter and mineral particles, without intimate mixing, in the underlying mineral horizons.[358]
Classification
Soil is classified into categories in order to understand relationships between different soils and to determine the suitability of a soil in a particular region. One of the first classification systems was developed by the Russian scientist Vasily Dokuchaev around 1880.[359] It was modified a number of times by American and European researchers, and developed into the system commonly used until the 1960s. It was based on the idea that soils have a particular morphology based on the materials and factors that form them. In the 1960s, a different classification system began to emerge which focused on soil morphology instead of parental materials and soil-forming factors. Since then it has undergone further modifications. The World Reference Base for Soil Resources (WRB)[360] aims to establish an international reference base for soil classification.
Uses
Soil is used in agriculture, where it serves as the anchor and primary nutrient base for plants. The types of soil and available moisture determine the species of plants that can be cultivated. Agricultural soil science was the primeval domain of soil knowledge, long time before the advent of pedology in the 19th century. However, as demonstrated by aeroponics, aquaponics and hydroponics, soil material is not an absolute essential for agriculture, and soilless cropping systems have been claimed as the future of agriculture for an endless growing mankind.[361]
Soil material is also a critical component in the mining, construction and landscape development industries.[362] Soil serves as a foundation for most construction projects. The movement of massive volumes of soil can be involved in surface mining, road building and dam construction. Earth sheltering is the architectural practice of using soil for external thermal mass against building walls. Many building materials are soil based. Loss of soil through urbanization is growing at a high rate in many areas and can be critical for the maintenance of subsistence agriculture.[363]
Soil resources are critical to the environment, as well as to food and fibre production, producing 98.8% of food consumed by humans.[364] Soil provides minerals and water to plants according to several processes involved in plant nutrition. Soil absorbs rainwater and releases it later, thus preventing floods and drought, flood regulation being one of the major ecosystem services provided by soil.[365] Soil cleans water as it percolates through it.[366] Soil is the habitat for many organisms: the major part of known and unknown biodiversity is in the soil, in the form of invertebrates (earthworms, woodlice, millipedes, centipedes, snails, slugs, mites, springtails, enchytraeids, nematodes, protists), bacteria, archaea, fungi and algae; and most organisms living above ground have part of them (plants) or spend part of their life cycle (insects) below-ground.[367] Above-ground and below-ground biodiversities are tightly interconnected,[334][368] making soil protection of paramount importance for any restoration or conservation plan.
The biological component of soil is an extremely important carbon sink since about 57% of the biotic content is carbon. Even on desert crusts, cyanobacteria, lichens and mosses capture and sequester a significant amount of carbon by photosynthesis. Poor farming and grazing methods have degraded soils and released much of this sequestered carbon to the atmosphere. Restoring the world's soils could offset the effect of increases in greenhouse gas emissions and slow global warming, while improving crop yields and reducing water needs.[369][370][371]
Waste management often has a soil component. Septic drain fields treat septic tank effluent using aerobic soil processes. Landfills use soil for daily cover. Land application of waste water relies on soil biology to aerobically treat BOD.
Organic soils, especially peat, serve as a significant fuel resource; but wide areas of peat production, such as sphagnum bogs, are now protected because of patrimonial interest.
Geophagy is the practice of eating soil-like substances. Both animals and human cultures occasionally consume soil for medicinal, recreational, or religious purposes. It has been shown that some monkeys consume soil, together with their preferred food (tree foliage and fruits), in order to alleviate tannin toxicity.[372]
Soils filter and purify water and affect its chemistry. Rain water and pooled water from ponds, lakes and rivers percolate through the soil horizons and the upper rock strata, thus becoming groundwater. Pests (viruses) and pollutants, such as persistent organic pollutants (chlorinated pesticides, polychlorinated biphenyls), oils (hydrocarbons), heavy metals (lead, zinc, cadmium), and excess nutrients (nitrates, sulfates, phosphates) are filtered out by the soil.[373] Soil organisms metabolise them or immobilise them in their biomass and necromass,[374] thereby incorporating them into stable humus.[375] The physical integrity of soil is also a prerequisite for avoiding landslides in rugged landscapes.[376]
Degradation
Land degradation[377] refers to a human-induced or natural process which impairs the capacity of land to function. Soils degradation involves the acidification, contamination, desertification, erosion or salination.
Soil acidification is beneficial in the case of alkaline soils, but it degrades land when it lowers crop productivity and increases soil vulnerability to contamination and erosion. Soils are often initially acid because their parent materials were acid and initially low in the basic cations (calcium, magnesium, potassium and sodium). Acidification occurs when these elements are leached from the soil profile by rainfall or by the harvesting of forest or agricultural crops. Soil acidification is accelerated by the use of acid-forming nitrogenous fertilizers and by the effects of acid precipitation.
Soil contamination at low levels is often within a soil's capacity to treat and assimilate waste material. Soil biota can treat waste by transforming it; soil colloids can adsorb the waste material. Many waste treatment processes rely on this treatment capacity. Exceeding treatment capacity can damage soil biota and limit soil function. Derelict soils occur where industrial contamination or other development activity damages the soil to such a degree that the land cannot be used safely or productively. Remediation of derelict soil uses principles of geology, physics, chemistry and biology to degrade, attenuate, isolate or remove soil contaminants to restore soil functions and values. Techniques include leaching, air sparging, chemical amendments, phytoremediation, bioremediation and natural degradation. An example of diffuse pollution with contaminants is the copper distribution in agricultural soils mainly due to fungicide applications in vineyards and other permanent crops.[378]

Desertification is an environmental process of ecosystem degradation in arid and semi-arid regions, often caused by human activity. It is a common misconception that droughts cause desertification. Droughts are common in arid and semiarid lands. Well-managed lands can recover from drought when the rains return. Soil management tools include maintaining soil nutrient and organic matter levels, reduced tillage and increased cover. These practices help to control erosion and maintain productivity during periods when moisture is available. Continued land abuse during droughts, however, increases land degradation. Increased population and livestock pressure on marginal lands accelerates desertification.
Erosion of soil is caused by water, wind, ice, and movement in response to gravity. More than one kind of erosion can occur simultaneously. Erosion is distinguished from weathering, since erosion also transports eroded soil away from its place of origin (soil in transit may be described as sediment). Erosion is an intrinsic natural process, but in many places it is greatly increased by human activity, especially poor land use practices. These include agricultural activities which leave the soil bare during times of heavy rain or strong winds, overgrazing, deforestation, and improper construction activity. Improved management can limit erosion. Soil conservation techniques which are employed include changes of land use (such as replacing erosion-prone crops with grass or other soil-binding plants), changes to the timing or type of agricultural operations, terrace building, use of erosion-suppressing cover materials (including cover crops and other plants), limiting disturbance during construction, and avoiding construction during erosion-prone periods.
A serious and long-running water erosion problem occurs in China, on the middle reaches of the Yellow River and the upper reaches of the Yangtze River. From the Yellow River, over 1.6 billion tons of sediment flow each year into the ocean. The sediment originates primarily from water erosion (gully erosion) in the Loess Plateau region of northwest China.
Soil piping is a particular form of soil erosion that occurs below the soil surface. It causes levee and dam failure, as well as sink hole formation. Turbulent flow removes soil starting at the mouth of the seep flow and the subsoil erosion advances up-gradient.[379] The term sand boil is used to describe the appearance of the discharging end of an active soil pipe.[380]
Soil salination is the accumulation of free salts to such an extent that it leads to degradation of the agricultural value of soils and vegetation. Consequences include corrosion damage, reduced plant growth, erosion due to loss of plant cover and soil structure, and water quality problems due to sedimentation. Salination occurs due to a combination of natural and human-caused processes. Arid conditions favour salt accumulation. This is especially apparent when soil parent material is saline. Irrigation of arid lands is especially problematic.[381] All irrigation water has some level of salinity. Irrigation, especially when it involves leakage from canals and overirrigation in the field, often raises the underlying water table. Rapid salination occurs when the land surface is within the capillary fringe of saline groundwater. Soil salinity control involves watertable control and flushing with higher levels of applied water in combination with tile drainage or another form of subsurface drainage.[382][383]
Reclamation
Soils which contain high levels of particular clays, such as smectites, are often very fertile. For example, the smectite-rich clays of Thailand's Central Plains are among the most productive in the world.
Many farmers in tropical areas, however, struggle to retain organic matter in the soils they work. In recent years, for example, productivity has declined in the low-clay soils of northern Thailand. Farmers initially responded by adding organic matter from termite mounds, but this was unsustainable in the long-term. Scientists experimented with adding bentonite, one of the smectite family of clays, to the soil. In field trials, conducted by scientists from the International Water Management Institute in cooperation with Khon Kaen University and local farmers, this had the effect of helping retain water and nutrients. Supplementing the farmer's usual practice with a single application of 200 kg bentonite per rai (6.26 rai = 1 hectare) resulted in an average yield increase of 73%. More work showed that applying bentonite to degraded sandy soils reduced the risk of crop failure during drought years.
In 2008, three years after the initial trials, IWMI scientists conducted a survey among 250 farmers in northeast Thailand, half of whom had applied bentonite to their fields. The average improvement for those using the clay addition was 18% higher than for non-clay users. Using the clay had enabled some farmers to switch to growing vegetables, which need more fertile soil. This helped to increase their income. The researchers estimated that 200 farmers in northeast Thailand and 400 in Cambodia had adopted the use of clays, and that a further 20,000 farmers were introduced to the new technique.[384]
If the soil is too high in clay, adding gypsum, washed river sand and organic matter will balance the composition. Adding organic matter (like ramial chipped wood for instance) to soil which is depleted in nutrients and too high in sand will boost its quality.[385]
History of studies and research
The history of the study of soil is intimately tied to humans' urgent need to provide food for themselves and forage for their animals. Throughout history, civilizations have prospered or declined as a function of the availability and productivity of their soils.[386]
Studies of soil fertility
The Greek historian Xenophon (450–355 BCE) is credited with being the first to expound upon the merits of green-manuring crops: "But then whatever weeds are upon the ground, being turned into earth, enrich the soil as much as dung."[387]
Columella's "Husbandry," circa 60 CE, advocated the use of lime and that clover and alfalfa (green manure) should be turned under, and was used by 15 generations (450 years) under the Roman Empire until its collapse.[387][388] From the fall of Rome to the French Revolution, knowledge of soil and agriculture was passed on from parent to child and as a result, crop yields were low. During the European Middle Ages, Yahya Ibn al-'Awwam's handbook,[389] with its emphasis on irrigation, guided the people of North Africa, Spain and the Middle East; a translation of this work was finally carried to the southwest of the United States when under Spanish influence.[390] Olivier de Serres, considered as the father of French agronomy, was the first to suggest the abandonment of fallowing and its replacement by hay meadows within crop rotations, and he highlighted the importance of soil (the French terroir) in the management of vineyards. His famous book Le Théâtre d'Agriculture et mesnage des champs[391] contributed to the rise of modern, sustainable agriculture and to the collapse of old agricultural practices such as soil improvement (amendment) for crops by the lifting of forest litter and assarting, which ruined the soils of western Europe during Middle Ages and even later on according to regions.[392]
Experiments into what made plants grow first led to the idea that the ash left behind when plant matter was burned was the essential element but overlooked the role of nitrogen, which is not left on the ground after combustion, a belief which prevailed until the 19th century.[393] In about 1635, the Flemish chemist Jan Baptist van Helmont thought he had proved water to be the essential element from his famous five years' experiment with a willow tree grown with only the addition of rainwater. His conclusion came from the fact that the increase in the plant's weight had apparently been produced only by the addition of water, with no reduction in the soil's weight.[394][395] John Woodward (d. 1728) experimented with various types of water ranging from clean to muddy and found muddy water the best, and so he concluded that earthy matter was the essential element. Others concluded it was humus in the soil that passed some essence to the growing plant. Still others held that the vital growth principal was something passed from dead plants or animals to the new plants. At the start of the 18th century, Jethro Tull demonstrated that it was beneficial to cultivate (stir) the soil, but his opinion that the stirring made the fine parts of soil available for plant absorption was erroneous.[394][396]
As chemistry developed, it was applied to the investigation of soil fertility. The French chemist Antoine Lavoisier showed in about 1778 that plants and animals must [combust] oxygen internally to live and was able to deduce that most of the 165-pound weight of van Helmont's willow tree derived from air.[397] It was the French agriculturalist Jean-Baptiste Boussingault who by means of experimentation obtained evidence showing that the main sources of carbon, hydrogen and oxygen for plants were air and water, while nitrogen was taken from soil.[398] Justus von Liebig in his book Organic chemistry in its applications to agriculture and physiology (published 1840), asserted that the chemicals in plants must have come from the soil and air and that to maintain soil fertility, the used minerals must be replaced.[399] Liebig nevertheless believed the nitrogen was supplied from the air. The enrichment of soil with guano by the Incas was rediscovered in 1802, by Alexander von Humboldt. This led to its mining and that of Chilean nitrate and to its application to soil in the United States and Europe after 1840.[400]
The work of Liebig was a revolution for agriculture, and so other investigators started experimentation based on it. In England John Bennet Lawes and Joseph Henry Gilbert worked in the Rothamsted Experimental Station, founded by the former, and (re)discovered that plants took nitrogen from the soil, and that salts needed to be in an available state to be absorbed by plants. Their investigations also produced the "superphosphate", consisting in the acid treatment of phosphate rock.[401] This led to the invention and use of salts of potassium (K) and nitrogen (N) as fertilizers. Ammonia generated by the production of coke was recovered and used as fertiliser.[402] Finally, the chemical basis of nutrients delivered to the soil in manure was understood and in the mid-19th century chemical fertilisers were applied. However, the dynamic interaction of soil and its life forms still awaited discovery.
In 1856 J. Thomas Way discovered that ammonia contained in fertilisers was transformed into nitrates,[403] and twenty years later Robert Warington proved that this transformation was done by living organisms.[404] In 1890 Sergei Winogradsky announced he had found the bacteria responsible for this transformation.[405]
It was known that certain legumes could take up nitrogen from the air and fix it to the soil but it took the development of bacteriology towards the end of the 19th century to lead to an understanding of the role played in nitrogen fixation by bacteria. The symbiosis of bacteria and leguminous roots, and the fixation of nitrogen by the bacteria, were simultaneously discovered by the German agronomist Hermann Hellriegel and the Dutch microbiologist Martinus Beijerinck.[401]
Crop rotation, mechanisation, chemical and natural fertilisers led to a doubling of wheat yields in western Europe between 1800 and 1900.[406]
Studies of soil formation
The scientists who studied the soil in connection with agricultural practices had considered it mainly as a static substrate. However, soil is the result of evolution from more ancient geological materials, under the action of biotic and abiotic (not associated with life) processes. After studies of the improvement of the soil commenced, other researchers began to study soil genesis and as a result also soil types and classifications.
In 1860, in Mississippi, Eugene W. Hilgard (1833-1916) studied the relationship between rock material, climate, vegetation, and the type of soils that were developed. He realised that the soils were dynamic, and considered the classification of soil types.[407] Unfortunately his work was not continued. At about the same time, Friedrich Albert Fallou was describing soil profiles and relating soil characteristics to their formation as part of his professional work evaluating forest and farm land for the principality of Saxony. His 1857 book, Anfangsgründe der Bodenkunde (First principles of soil science) established modern soil science.[408] Contemporary with Fallou's work, and driven by the same need to accurately assess land for equitable taxation, Vasily Dokuchaev led a team of soil scientists in Russia who conducted an extensive survey of soils, observing that similar basic rocks, climate and vegetation types lead to similar soil layering and types, and established the concepts for soil classifications. Due to language barriers, the work of this team was not communicated to western Europe until 1914 through a publication in German by Konstantin Glinka, a member of the Russian team.[409]
Curtis F. Marbut, influenced by the work of the Russian team, translated Glinka's publication into English,[410] and as he was placed in charge of the U.S. National Cooperative Soil Survey, applied it to a national soil classification system.[394]
References
- Chesworth, Ward, ed. (2008). Encyclopedia of soil science (PDF). Dordrecht, The Netherlands: Springer. ISBN 978-1-4020-3994-2. Archived (PDF) from the original on 5 September 2018. Retrieved 14 January 2019.
- "pedo-". Oxford English Dictionary (3rd ed.). Oxford University Press. September 2005. (Subscription or UK public library membership required.), from the ancient Greek πέδον "ground", "earth".
- Voroney, R. Paul & Heck, Richard J. (2007). "The soil habitat" (PDF). In Paul, Eldor A. (ed.). Soil microbiology, ecology and biochemistry (3rd ed.). Amsterdam: Elsevier. pp. 25–49. doi:10.1016/B978-0-08-047514-1.50006-8. ISBN 978-0-12-546807-7. Archived (PDF) from the original on 10 July 2018. Retrieved 15 January 2019.
- Danoff-Burg, James A. "The terrestrial influence: geology and soils". Earth Institute Center for Environmental Sustainability. New York: Columbia University Press. Retrieved 17 December 2017.
- Taylor, Sterling A. & Ashcroft, Gaylen L. (1972). Physical edaphology: the physics of irrigated and nonirrigated soils. San Francisco: W.H. Freeman. ISBN 978-0-7167-0818-6.
- McCarthy, David F. (2006). Essentials of soil mechanics and foundations: basic geotechnics (7th ed.). Upper Saddle River, New Jersey: Prentice Hall. ISBN 978-0-13-114560-3.
- Gilluly, James; Waters, Aaron Clement & Woodford, Alfred Oswald (1975). Principles of geology (4th ed.). San Francisco: W.H. Freeman. ISBN 978-0-7167-0269-6.
- Ponge, Jean-François (2015). "The soil as an ecosystem" (PDF). Biology and Fertility of Soils. 51 (6): 645–48. doi:10.1007/s00374-015-1016-1. Retrieved 17 December 2017.
- Yu, Charley; Kamboj, Sunita; Wang, Cheng & Cheng, Jing-Jy (2015). "Data collection handbook to support modeling impacts of radioactive material in soil and building structures" (PDF). Argonne National Laboratory. pp. 13–21. Archived (PDF) from the original on 4 August 2018. Retrieved 17 December 2017.
- Buol, Stanley W.; Southard, Randal J.; Graham, Robert C. & McDaniel, Paul A. (2011). Soil genesis and classification (7th ed.). Ames, Iowa: Wiley-Blackwell. ISBN 978-0-470-96060-8.
- Retallack, Gregory J.; Krinsley, David H; Fischer, Robert; Razink, Joshua J. & Langworthy, Kurt A. (2016). "Archean coastal-plain paleosols and life on land" (PDF). Gondwana Research. 40: 1–20. Bibcode:2016GondR..40....1R. doi:10.1016/j.gr.2016.08.003. Archived (PDF) from the original on 13 November 2018. Retrieved 15 January 2019.
- "Glossary of Terms in Soil Science". Agriculture and Agri-Food Canada. Archived from the original on 27 October 2018. Retrieved 15 January 2019.
- Amundson, Ronald. "Soil preservation and the future of pedology" (PDF). Faculty of Natural Resources. Songkhla, Thailand: Prince of Songkla University. Archived (PDF) from the original on 12 June 2018. Retrieved 15 January 2019.
- Küppers, Michael; Vincent, Jean-Baptiste. "Impacts and formation of regolith". Max Planck Institute for Solar System Research. Archived from the original on 4 August 2018. Retrieved 15 January 2019.
- Pouyat, Richard; Groffman, Peter; Yesilonis, Ian & Hernandez, Luis (2002). "Soil carbon pools and fluxes in urban ecosystems" (PDF). Environmental Pollution. 116 (Supplement 1): S107–S118. doi:10.1016/S0269-7491(01)00263-9. PMID 11833898. Retrieved 17 December 2017.
- Davidson, Eric A. & Janssens, Ivan A. (2006). "Temperature sensitivity of soil carbon decomposition and feedbacks to climate change" (PDF). Nature. 440 (9 March 2006): 165‒73. Bibcode:2006Natur.440..165D. doi:10.1038/nature04514. PMID 16525463. Retrieved 17 December 2017.
- Powlson, David (2005). "Climatology: will soil amplify climate change?". Nature. 433 (20 January 2005): 204‒05. Bibcode:2005Natur.433..204P. doi:10.1038/433204a. PMID 15662396.
- Bradford, Mark A.; Wieder, William R.; Bonan, Gordon B.; Fierer, Noah; Raymond, Peter A. & Crowther, Thomas W. (2016). "Managing uncertainty in soil carbon feedbacks to climate change" (PDF). Nature Climate Change. 6 (27 July 2016): 751–58. Bibcode:2016NatCC...6..751B. doi:10.1038/nclimate3071. Retrieved 17 December 2017.
- Dominati, Estelle; Patterson, Murray & Mackay, Alec (2010). "A framework for classifying and quantifying the natural capital and ecosystem services of soils" (PDF). Ecological Economics. 69 (9): 1858‒68. doi:10.1016/j.ecolecon.2010.05.002. Archived from the original (PDF) on 8 August 2017. Retrieved 17 December 2017.
- Dykhuizen, Daniel E. (1998). "Santa Rosalia revisited: why are there so many species of bacteria?" (PDF). Antonie van Leeuwenhoek. 73 (1): 25‒33. doi:10.1023/A:1000665216662. PMID 9602276. Retrieved 17 December 2017.
- Torsvik, Vigdis & Øvreås, Lise (2002). "Microbial diversity and function in soil: from genes to ecosystems". Current Opinion in Microbiology. 5 (3): 240‒45. doi:10.1016/S1369-5274(02)00324-7. PMID 12057676.
- Raynaud, Xavier & Nunan, Naoise (2014). "Spatial ecology of bacteria at the microscale in soil". PLOS ONE. 9 (1): e87217. Bibcode:2014PLoSO...987217R. doi:10.1371/journal.pone.0087217. PMC 3905020. PMID 24489873.
- Whitman, William B.; Coleman, David C. & Wiebe, William J. (1998). "Prokaryotes: the unseen majority". Proceedings of the National Academy of Sciences of the USA. 95 (12): 6578‒83. Bibcode:1998PNAS...95.6578W. doi:10.1073/pnas.95.12.6578. PMC 33863. PMID 9618454.
- Schlesinger, William H. & Andrews, Jeffrey A. (2000). "Soil respiration and the global carbon cycle" (PDF). Biogeochemistry. 48 (1): 7‒20. doi:10.1023/A:1006247623877. Retrieved 17 December 2017.
- Denmead, Owen Thomas & Shaw, Robert Harold (1962). "Availability of soil water to plants as affected by soil moisture content and meteorological conditions" (PDF). Agronomy Journal. 54 (5): 385‒90. doi:10.2134/agronj1962.00021962005400050005x. Retrieved 17 December 2017.
- House, Christopher H.; Bergmann, Ben A.; Stomp, Anne-Marie & Frederick, Douglas J. (1999). "Combining constructed wetlands and aquatic and soil filters for reclamation and reuse of water" (PDF). Ecological Engineering. 12 (1–2): 27–38. doi:10.1016/S0925-8574(98)00052-4. Retrieved 17 December 2017.
- Van Bruggen, Ariena H.C. & Semenov, Alexander M. (2000). "In search of biological indicators for soil health and disease suppression" (PDF). Applied Soil Ecology. 15 (1): 13–24. doi:10.1016/S0929-1393(00)00068-8. Retrieved 17 December 2017.
- "A citizen's guide to monitored natural attenuation" (PDF). Retrieved 17 December 2017.
- Linn, Daniel Myron; Doran, John W. (1984). "Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tilled and nontilled soils" (PDF). Soil Science Society of America Journal. 48 (6): 1267–72. Bibcode:1984SSASJ..48.1267L. doi:10.2136/sssaj1984.03615995004800060013x. Retrieved 17 December 2017.
- Miller, Raymond W.; Donahue, Roy Luther (1990). Soils: an introduction to soils and plant growth. Upper Saddle River, New Jersey: Prentice Hall. ISBN 978-0-13-820226-2.
- Bot, Alexandra; Benites, José (2005). The importance of soil organic matter: key to drought-resistant soil and sustained food and production (PDF). Rome: Food and Agriculture Organization of the United Nations. ISBN 978-92-5-105366-9. Retrieved 17 December 2017.
- McClellan, Tai. "Soil composition". University of Hawai‘i – College of Tropical Agriculture and Human Resources. Retrieved 29 April 2018.
- "Arizona Master Gardener Manual". Cooperative Extension, College of Agriculture, University of Arizona. 9 November 2017. Archived from the original on 29 May 2016. Retrieved 17 December 2017.
- Vannier, Guy (1987). "The porosphere as an ecological medium emphasized in Professor Ghilarov's work on soil animal adaptations" (PDF). Biology and Fertility of Soils. 3 (1): 39–44. doi:10.1007/BF00260577. Retrieved 29 July 2018.
- Torbert, H. Allen & Wood, Wes (1992). "Effect of soil compaction and water-filled pore space on soil microbial activity and N losses" (PDF). Communications in Soil Science and Plant Analysis. 23 (11): 1321‒31. doi:10.1080/00103629209368668. Retrieved 17 December 2017.
- Simonson 1957, p. 17.
- Bronick, Carol J. & Lal, Ratan (January 2005). "Soil structure and management: a review" (PDF). Geoderma. 124 (1/2): 3–22. Bibcode:2005Geode.124....3B. doi:10.1016/j.geoderma.2004.03.005. Retrieved 17 December 2017.
- "Soil and water". Food and Agriculture Organization of the United Nations. Retrieved 17 December 2017.
- Valentin, Christian; d'Herbès, Jean-Marc & Poesen, Jean (1999). "Soil and water components of banded vegetation patterns" (PDF). Catena. 37 (1): 1‒24. doi:10.1016/S0341-8162(99)00053-3. Retrieved 17 December 2017.
- Barber, Stanley A. (1995). "Chemistry of soil-nutrient associations". In Barber, Stanley A. (ed.). Soil nutrient bioavailability: a mechanistic approach (2nd ed.). New York: John Wiley & Sons. pp. 9–48. ISBN 978-0-471-58747-7.
- "Soil colloids: properties, nature, types and significance" (PDF). Tamil Nadu Agricultural University. Retrieved 17 December 2017.
- "Cation exchange capacity in soils, simplified". Retrieved 17 December 2017.
- Miller, Jarrod O. "Soil pH affects nutrient availability" (PDF). University of Maryland. Retrieved 17 December 2017.
- Goulding, Keith W.T.; Bailey, Neal J.; Bradbury, Nicola J.; Hargreaves, Patrick; Howe, MT; Murphy, Daniel V.; Poulton, Paul R. & Willison, Toby W. (1998). "Nitrogen deposition and its contribution to nitrogen cycling and associated soil processes". New Phytologist. 139 (1): 49‒58. doi:10.1046/j.1469-8137.1998.00182.x.
- Kononova, M.M. (2013). Soil organic matter: its nature, its role in soil formation and in soil fertility (2nd ed.). Amsterdam: Elsevier. ISBN 978-1-4831-8568-2.
- Bishop, Janice L.; Murchie, Scott L.; Pieters, Carlé L. & Zent, Aaron P. (2002). "A model for formation of dust, soil, and rock coatings on Mars: physical and chemical processes on the Martian surface". Journal of Geophysical Research. 107 (E11): 7-1–7-17. Bibcode:2002JGRE..107.5097B. doi:10.1029/2001JE001581.
- Navarro-González, Rafael; Rainey, Fred A.; Molina, Paola; Bagaley, Danielle R.; Hollen, Becky J.; de la Rosa, José; Small, Alanna M.; Quinn, Richard C.; Grunthaner, Frank J.; Cáceres, Luis; Gomez-Silva, Benito & McKay, Christopher P. (2003). "Mars-like soils in the Atacama desert, Chile, and the dry limit of microbial life" (PDF). Science. 302 (5647): 1018–21. Bibcode:2003Sci...302.1018N. doi:10.1126/science.1089143. PMID 14605363. Retrieved 17 December 2017.
- Van Schöll, Laura; Smits, Mark M. & Hoffland, Ellis (2006). "Ectomycorrhizal weathering of the soil minerals muscovite and hornblende". New Phytologist. 171 (4): 805–14. doi:10.1111/j.1469-8137.2006.01790.x. PMID 16918551.
- Jackson, Togwell A. & Keller, Walter David (1970). "A comparative study of the role of lichens and "inorganic" processes in the chemical weathering of recent Hawaiian lava flows". American Journal of Science. 269 (5): 446–66. Bibcode:1970AmJS..269..446J. doi:10.2475/ajs.269.5.446.
- Dojani, Stephanie; Lakatos, Michael; Rascher, Uwe; Waneck, Wolfgang; Luettge, Ulrich & Büdel, Burkhard (2007). "Nitrogen input by cyanobacterial biofilms of an inselberg into a tropical rainforest in French Guiana". Flora. 202 (7): 521–29. doi:10.1016/j.flora.2006.12.001.
- Kabala, Cesary & Kubicz, Justyna (2012). "Initial soil development and carbon accumulation on moraines of the rapidly retreating Werenskiold Glacier, SW Spitsbergen, Svalbard archipelago" (PDF). Geoderma. 175/176: 9–20. Bibcode:2012Geode.175....9K. doi:10.1016/j.geoderma.2012.01.025. Retrieved 26 May 2019.
- Jenny, Hans (1941). Factors of soil formation: a system of qunatitative pedology (PDF). New York: McGraw-Hill. Archived from the original (PDF) on 8 August 2017. Retrieved 17 December 2017.
- Ritter, Michael E. "The physical environment: an introduction to physical geography". Retrieved 17 December 2017.
- Gardner, Catriona M.K.; Laryea, Kofi Buna & Unger, Paul W. (1999). Soil physical constraints to plant growth and crop production (PDF) (1st ed.). Rome: Food and Agriculture Organization of the United Nations. Archived from the original (PDF) on 8 August 2017. Retrieved 24 December 2017.
- Six, Johan; Paustian, Keith; Elliott, Edward T. & Combrink, Clay (2000). "Soil structure and organic matter. I. Distribution of aggregate-size classes and aggregate-associated carbon" (PDF). Soil Science Society of America Journal. 64 (2): 681–89. Bibcode:2000SSASJ..64..681S. doi:10.2136/sssaj2000.642681x. Retrieved 24 December 2017.
- Håkansson, Inge & Lipiec, Jerzy (2000). "A review of the usefulness of relative bulk density values in studies of soil structure and compaction" (PDF). Soil and Tillage Research. 53 (2): 71–85. doi:10.1016/S0167-1987(99)00095-1. Retrieved 24 December 2017.
- Schwerdtfeger, W.J. (1965). "Soil resistivity as related to underground corrosion and cathodic protection". Journal of Research of the National Bureau of Standards. 69C (1): 71–77. doi:10.6028/jres.069c.012.
- Tamboli, Prabhakar Mahadeo (1961). The influence of bulk density and aggregate size on soil moisture retention (PDF). Ames, Iowa: Iowa State University. Retrieved 24 December 2017.
- Wallace, James S. & Batchelor, Charles H. (1997). "Managing water resources for crop production". Philosophical Transactions of the Royal Society B: Biological Sciences. 352 (1356): 937–47. doi:10.1098/rstb.1997.0073. PMC 1691982.
- Veihmeyer, Frank J. & Hendrickson, Arthur H. (1927). "Soil-moisture conditions in relation to plant growth". Plant Physiology. 2 (1): 71–82. doi:10.1104/pp.2.1.71. PMC 439946. PMID 16652508.
- Donahue, Miller & Shickluna 1977, p. 72.
- Van Breemen, Nico & Buurman, Peter (2003). Soil formation (PDF) (2nd ed.). Dordrecht, The Netherlands: Kluwer Academic Publishers. ISBN 978-0-306-48163-5. Retrieved 29 April 2018.
- Ratliff, Larry F.; Ritchie, Jerry T. & Cassel, D. Keith (1983). "Field-measured limits of soil water availability as related to laboratory-measured properties" (PDF). Soil Science Society of America Journal. 47 (4): 770–75. Bibcode:1983SSASJ..47..770R. doi:10.2136/sssaj1983.03615995004700040032x. Retrieved 29 April 2018.
- Wadleigh 1957, p. 48.
- Richards & Richards 1957, p. 50.
- Richards & Richards 1957, p. 56.
- Wadleigh 1957, p. 39.
- Richards & Richards 1957, p. 52.
- "Water movement in soils". Oklahoma State University. Retrieved 1 May 2018.
- Le Bissonnais, Yves (2016). "Aggregate stability and assessment of soil crustability and erodibility. I. Theory and methodology" (PDF). European Journal of Soil Science. 67 (1): 11–21. doi:10.1111/ejss.4_12311. Retrieved 5 May 2018.
- Easton, Zachary M. & Bock, Emily. "Soil and soil water relationships" (PDF). Virginia Tech. Retrieved 5 May 2018.
- Sims, J. Thomas; Simard, Régis R. & Joern, Brad Christopher (1998). "Phosphorus loss in agricultural drainage: historical perspective and current research" (PDF). Journal of Environmental Quality. 27 (2): 277–93. doi:10.2134/jeq1998.00472425002700020006x. Retrieved 6 May 2018.
- Brooks, Royal H. & Corey, Arthur T. (1966). "Properties of porous media affecting fluid flow" (PDF). Journal of the Irrigation and Drainage Division. 92 (2): 61–90. Retrieved 6 May 2018.
- McElrone, Andrew J.; Choat, Brendan; Gambetta, Greg A. & Brodersen, Craig R. "Water uptake and transport in vascular plants". The Nature Education Knowledge Project. Retrieved 6 May 2018.
- Steudle, Ernst (2000). "Water uptake by plant roots: an integration of views" (PDF). Plant and Soil. 226 (1): 45–56. doi:10.1023/A:1026439226716. Retrieved 6 May 2018.
- Wilcox, Carolyn S.; Ferguson, Joseph W.; Fernandez, George C.J. & Nowak, Robert S. (2004). "Fine root growth dynamics of four Mojave Desert shrubs as related to soil moisture and microsite" (PDF). Journal of Arid Environments. 56 (1): 129–48. Bibcode:2004JArEn..56..129W. doi:10.1016/S0140-1963(02)00324-5. Retrieved 6 May 2018.
- Hunter, Albert S. & Kelley, Omer J. (1946). "The extension of plant roots into dry soil". Plant Physiology. 21 (4): 445–51. doi:10.1104/pp.21.4.445. PMC 437296. PMID 16654059.
- Zhang, Yongqiang; Kendy, Eloise; Qiang, Yu; Liu, Changming; Shen, Yanjun & Sun, Hongyong (2004). "Effect of soil water deficit on evapotranspiration, crop yield, and water use efficiency in the North China Plain" (PDF). Agricultural Water Management. 64 (2): 107–22. doi:10.1016/S0378-3774(03)00201-4. Retrieved 6 May 2018.
- Oyewole, Olusegun Ayodeji; Inselsbacher, Erich & Näsholm, Torgny (2014). "Direct estimation of mass flow and diffusion of nitrogen compounds in solution and soil" (PDF). New Phytologist. 201 (3): 1056–64. doi:10.1111/nph.12553. PMID 24134319. Retrieved 10 May 2018.
- "the GCOS Essential Climate Variables". GCOS. 2013. Retrieved 5 November 2013.
- Brocca, L.; Hasenauer, S.; Lacava, T.; oramarco, T.; Wagner, W.; Dorigo, W.; Matgen, P.; Martínez-Fernández, J.; Llorens, P.; Latron, C.; Martin, C.; Bittelli, M. (2011). "Soil moisture estimation through ASCAT and AMSR-E sensors: An intercomparison and validation study across Europe". Remote Sensing of Environment. 115 (12): 3390–3408. Bibcode:2011RSEnv.115.3390B. doi:10.1016/j.rse.2011.08.003.
- Donahue, Miller & Shickluna 1977, pp. 72–74.
- "Soil and water". Food and Agriculture Organization of the United Nations. Retrieved 10 May 2018.
- Petersen, Lis Wollesen; Møldrup, Per; Jacobsen, Ole H. & Rolston, Dennis E. (1996). "Relations between specific surface area and soil physical and chemical properties" (PDF). Soil Science. 161 (1): 9–21. Bibcode:1996SoilS.161....9P. doi:10.1097/00010694-199601000-00003. Retrieved 10 May 2018.
- Gupta, Satish C. & Larson, William E. (1979). "Estimating soil water retention characteristics from particle size distribution, organic matter percent, and bulk density". Water Resources Research. 15 (6): 1633–35. Bibcode:1979WRR....15.1633G. CiteSeerX 10.1.1.475.497. doi:10.1029/WR015i006p01633.
- "Soil Water Potential". AgriInfo.in. Archived from the original on 17 August 2017. Retrieved 15 March 2019.
- Savage, Michael J.; Ritchie, Joe T.; Bland, William L. & Dugas, William A. (1996). "Lower limit of soil water availability" (PDF). Agronomy Journal. 88 (4): 644–51. doi:10.2134/agronj1996.00021962008800040024x. Retrieved 12 May 2018.
- Al-Ani, Tariq & Bierhuizen, Johan Frederik (1971). "Stomatal resistance, transpiration, and relative water content as influenced by soil moisture stress" (PDF). Acta Botanica Neerlandica. 20 (3): 318–26. doi:10.1111/j.1438-8677.1971.tb00715.x. Retrieved 12 May 2018.
- Donahue, Miller & Shickluna 1977, pp. 75–76.
- Rawls, W. J.; Brakensiek, D. L.; Saxtonn, K. E. (1982). "Estimation of Soil Water Properties" (PDF). Transactions of the ASAE. 25 (5): 1316–1320. doi:10.13031/2013.33720. Retrieved 17 March 2019.
- Donahue, Miller & Shickluna 1977, p. 85.
- "Soil water movement: saturated and unsaturated flow and vapour movement, soil moisture constants and their importance in irrigation" (PDF). Tamil Nadu Agricultural University. Retrieved 19 May 2018.
- Donahue, Miller & Shickluna 1977, p. 86.
- Donahue, Miller & Shickluna 1977, p. 88.
- Cueto-Felgueroso, Luis & Juanes, Ruben (2008). "Nonlocal interface dynamics and pattern formation in gravity-driven unsaturated flow through porous media" (PDF). Physical Review Letters. 101 (24): 244504. Bibcode:2008PhRvL.101x4504C. doi:10.1103/PhysRevLett.101.244504. PMID 19113626. Retrieved 21 May 2018.
- "Finger flow in coarse soils". Cornell University. Retrieved 21 May 2018.
- Ghestem, Murielle; Sidle, Roy C. & Stokes, Alexia (2011). "The influence of plant root systems on subsurface flow: implications for slope stability". BioScience. 61 (11): 869–79. doi:10.1525/bio.2011.61.11.6.
- Bartens, Julia; Day, Susan D.; Harris, J. Roger; Dove, Joseph E. & Wynn, Theresa M. (2008). "Can urban tree roots improve infiltration through compacted subsoils for stormwater management?" (PDF). Journal of Environmental Quality. 37 (6): 2048–57. doi:10.2134/jeq2008.0117. PMID 18948457. Retrieved 21 May 2018.
- Zhang, Guohua; Feng, Gary; Li, Xinhu; Xie, Congbao & P, Xiaoyu (2017). "Flood effect on groundwater recharge on a typical silt loam soil". Water. 9 (7): 523. doi:10.3390/w9070523.
- Nielsen, Donald R.; Biggar, James W. & Erh, Koon T. (1973). "Spatial variability of field-measured soil-water properties". Hilgardia. 42 (7): 215–59. doi:10.3733/hilg.v42n07p215. Archived from the original (PDF) on 12 June 2018. Retrieved 9 June 2018.
- Rimon, Yaara; Dahan, Ofer; Nativ, Ronit & Geyer, Stefan (2007). "Water percolation through the deep vadose zone and groundwater recharge: preliminary results based on a new vadose zone monitoring system". Water Resources Research. 43 (5): W05402. Bibcode:2007WRR....43.5402R. doi:10.1029/2006WR004855.
- Weiss, Peter T.; LeFevre, Greg & Gulliver, John S. (2008). "Contamination of soil and groundwater due to stormwater infiltration practices: a literature review". CiteSeerX 10.1.1.410.5113. Cite journal requires
|journal=(help) - Hagedorn, Charles; Hansen, Debra T. & Simonson, Gerald H. (1978). "Survival and movement of fecal indicator bacteria in soil under conditions of saturated flow" (PDF). Journal of Environmental Quality. 7 (1): 55–59. doi:10.2134/jeq1978.00472425000700010011x. Retrieved 24 June 2018.
- Donahue, Miller & Shickluna 1977, p. 90.
- Donahue, Miller & Shickluna 1977, p. 80.
- Ng, Charles W.W. & Pang, Wenyan (2000). "Influence of stress state on soil-water characteristics and slope stability" (PDF). Journal of Geotechnical and Geoenvironmental Engineering. 126 (2): 157–66. doi:10.1061/(ASCE)1090-0241(2000)126:2(157). Retrieved 1 July 2018.
- Richards, L.A. (1931). "Capillary conduction of liquids through porous mediums". Physics. 1 (5): 318–333. Bibcode:1931Physi...1..318R. doi:10.1063/1.1745010.
- Richardson, Lewis Fry (1922). Weather prediction by numerical process. Cambridge, The University press. p. 262.
- Ogden, Fred L.; Allen, Myron B.; Lai, Wencong; Zhu, Julian; Douglas, Craig C.; Seo, Mookwon; Talbot, Cary A. (2017). "The Soil Moisture Velocity Equation". J. Adv. Modeling Earth Syst. 9 (2): 1473–1487. Bibcode:2017JAMES...9.1473O. doi:10.1002/2017MS000931.
- Talbot, Cary A.; Ogden, Fred L. (2008). "A method for computing infiltration and redistribution in a discretized moisture content domain". Water Resour. Res. 44 (8): 8. Bibcode:2008WRR....44.8453T. doi:10.1029/2008WR006815.
- Ogden, Fred L.; Lai, Wencong; Steinke, Robert C.; Zhu, Julian; Talbot, Cary A.; Wilson, John L. (2015). "A new general 1-D vadose zone solution method". Water Resour. Res. 51: 4282–4300. Bibcode:2015WRR....51.4282O. doi:10.1002/2015WR017126.
- Šimůnek, J.; Saito, H.; Sakai, M.; van Genuchten, M. Th. (2013). "The HYDRUS-1D Software Package for Simulating the One-Dimensional Movement of Water, Heat, and Multiple Solutes in Variably-Saturated Media". Retrieved 15 March 2019.
- Bouma, Johan (1981). "Soil morphology and preferential flow along macropores" (PDF). Geoderma. 3 (4): 235–50. doi:10.1016/0378-3774(81)90009-3. Retrieved 1 July 2018.
- Luo, Lifang; Lin, Henry & Halleck, Phil (2008). "Quantifying soil structure and preferential flow in intact soil Using X-ray computed tomography". Soil Science Society of America Journal. 72 (4): 1058–69. Bibcode:2008SSASJ..72.1058L. CiteSeerX 10.1.1.455.2567. doi:10.2136/sssaj2007.0179.
- Beven, Keith & Germann, Peter (2013). "Macropores and water flow in soils revisited" (PDF). Water Resources Research. 49 (6): 3071–92. Bibcode:2013WRR....49.3071B. doi:10.1002/wrcr.20156.
- Aston, M.J. & Lawlor, David W. (1979). "The relationship between transpiration, root water uptake, and leaf water potential" (PDF). Journal of Experimental Botany. 30 (1): 169–81. doi:10.1093/jxb/30.1.169. Retrieved 8 July 2018.
- Powell, D.B.B. (1978). "Regulation of plant water potential by membranes of the endodermis in young roots" (PDF). Plant, Cell and Environment. 1 (1): 69–76. doi:10.1111/j.1365-3040.1978.tb00749.x. Retrieved 7 July 2018.
- Irvine, James; Perks, Michael P.; Magnani, Federico & Grace, John (1998). "The response of Pinus sylvestris to drought: stomatal control of transpiration and hydraulic conductance". Tree Physiology. 18 (6): 393–402. doi:10.1093/treephys/18.6.393. PMID 12651364.
- Jackson, Robert B.; Sperry, John S. & Dawson, Todd E. (2000). "Root water uptake and transport: using physiological processes in global predictions" (PDF). Trends in Plant Science. 5 (11): 482–88. doi:10.1016/S1360-1385(00)01766-0. PMID 11077257. Retrieved 8 July 2018.
- Steudle, Ernst (2000). "Water uptake by plant roots: an integration of views" (PDF). Plant and Soil. 226 (1): 45–56. doi:10.1023/A:1026439226716. Retrieved 8 July 2018.
- Donahue, Miller & Shickluna 1977, p. 92.
- Kaufmann, Merrill R. & Eckard, Alan N. (1971). "Evaluation of water stress control with polyethylene glycols by analysis of guttation". Plant Physiology. 47 (4): 453–6. doi:10.1104/pp.47.4.453. PMC 396708. PMID 16657642.
- Wadleigh 1957, p. 46.
- Kramer, Paul J. & Coile, Theodore S. (1940). "An estimation of the volume of water made available by root extension". Plant Physiology. 15 (4): 743–47. doi:10.1104/pp.15.4.743. PMC 437871. PMID 16653671.
- Lynch, Jonathan (1995). "Root architecture and plant productivity". Plant Physiology. 109 (1): 7–13. doi:10.1104/pp.109.1.7. PMC 157559. PMID 12228579.
- Comas, Louise H.; Eissenstat, David M. & Lakso, Alan N. (2000). "Assessing root death and root system dynamics in a study of grape canopy pruning". New Phytologist. 147 (1): 171–78. doi:10.1046/j.1469-8137.2000.00679.x.
- Donahue, Miller & Shickluna 1977, p. 94.
- Schlesinger, William H. & Jasechko, Scott (2014). "Transpiration in the global water cycle" (PDF). Agricultural and Forest Meteorology. 189/190: 115–17. Bibcode:2014AgFM..189..115S. doi:10.1016/j.agrformet.2014.01.011. Retrieved 22 July 2018.
- Erie, Leonard J.; French, Orrin F. & Harris, Karl (1968). Consumptive use of water by crops in Arizona (PDF). Tucson, Arizona: The University of Arizona. Retrieved 15 July 2018.
- Tolk, Judy A.; Howell, Terry A. & Evett, Steve R. (1999). "Effect of mulch, irrigation, and soil type on water use and yield of maize" (PDF). Soil and Tillage Research. 50 (2): 137–47. doi:10.1016/S0167-1987(99)00011-2. Retrieved 15 July 2018.
- Donahue, Miller & Shickluna 1977, pp. 97–99.
- Qi, Jingen; Marshall, John D. & Mattson, Kim G. (1994). "High soil carbon dioxide concentrations inhibit root respiration of Douglas fir". New Phytologist. 128 (3): 435–42. doi:10.1111/j.1469-8137.1994.tb02989.x.
- Karberg, Noah J.; Pregitzer, Kurt S.; King, John S.; Friend, Aaron L. & Wood, James R. (2005). "Soil carbon dioxide partial pressure and dissolved inorganic carbonate chemistry under elevated carbon dioxide and ozone" (PDF). Oecologia. 142 (2): 296–306. Bibcode:2005Oecol.142..296K. doi:10.1007/s00442-004-1665-5. PMID 15378342. Retrieved 26 August 2018.
- Chang, H.T. & Loomis, W.E. (1945). "Effect of carbon dioxide on absorption of water and nutrients by roots". Plant Physiology. 20 (2): 221–32. doi:10.1104/pp.20.2.221. PMC 437214. PMID 16653979.
- McDowell, Nate J.; Marshall, John D.; Qi, Jingen & Mattson, Kim (1999). "Direct inhibition of maintenance respiration in western hemlock roots exposed to ambient soil carbon dioxide concentrations" (PDF). Tree Physiology. 19 (9): 599–605. doi:10.1093/treephys/19.9.599. PMID 12651534. Retrieved 22 July 2018.
- Xu, Xia; Nieber, John L. & Gupta, Satish C. (1992). "Compaction effect on the gas diffusion coefficient in soils" (PDF). Soil Science Society of America Journal. 56 (6): 1743–50. Bibcode:1992SSASJ..56.1743X. doi:10.2136/sssaj1992.03615995005600060014x. Retrieved 29 July 2018.
- Smith, Keith A.; Ball, Tom; Conen, Franz; Dobbie, Karen E.; Massheder, Jonathan & Rey, Ana (2003). "Exchange of greenhouse gases between soil and atmosphere: interactions of soil physical factors and biological processes" (PDF). European Journal of Soil Science. 54 (4): 779–91. doi:10.1046/j.1351-0754.2003.0567.x. Retrieved 5 August 2018.
- Russell 1957, pp. 35–36.
- Ruser, Reiner; Flessa, Heiner; Russow, Rolf; Schmidt, G.; Buegger, Franz & Munch, J.C. (2006). "Emission of N2O, N2 and CO2 from soil fertilized with nitrate: effect of compaction, soil moisture and rewetting" (PDF). Soil Biology and Biochemistry. 38 (2): 263–74. doi:10.1016/j.soilbio.2005.05.005. Retrieved 5 August 2018.
- Hartmann, Adrian A.; Buchmann, Nina & Niklaus, Pascal A. (2011). "A study of soil methane sink regulation in two grasslands exposed to drought and N fertilization" (PDF). Plant and Soil. 342 (1/2): 265–75. doi:10.1007/s11104-010-0690-x. Retrieved 12 August 2018.
- Moore, Tim R. & Dalva, Moshe (1993). "The influence of temperature and water table position on carbon dioxide and methane emissions from laboratory columns of peatland soils" (PDF). Journal of Soil Science. 44 (4): 651–64. doi:10.1111/j.1365-2389.1993.tb02330.x. Retrieved 12 August 2018.
- Hiltpold, Ivan; Toepfer, Stefan; Kuhlmann, Ulrich & Turlings, Ted C.J. (2010). "How maize root volatiles affect the efficacy of entomopathogenic nematodes in controlling the western corn rootworm?" (PDF). Chemoecology. 20 (2): 155–62. doi:10.1007/s00049-009-0034-6. Retrieved 12 August 2018.
- Ryu, Choong-Min; Farag, Mohamed A.; Hu, Chia-Hui; Reddy, Munagala S.; Wei, Han-Xun; Paré, Paul W. & Kloepper, Joseph W. (2003). "Bacterial volatiles promote growth in Arabidopsis" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 100 (8): 4927–32. Bibcode:2003PNAS..100.4927R. doi:10.1073/pnas.0730845100. PMC 153657. PMID 12684534. Retrieved 12 August 2018.
- Hung, Richard; Lee, Samantha & Bennett, Joan W. (2015). "Fungal volatile organic compounds and their role in ecosystems" (PDF). Applied Microbiology and Biotechnology. 99 (8): 3395–405. doi:10.1007/s00253-015-6494-4. PMID 25773975. Retrieved 12 August 2018.
- Purrington, Foster Forbes; Kendall, Paricia A.; Bater, John E. & Stinner, Benjamin R. (1991). "Alarm pheromone in a gregarious poduromorph collembolan (Collembola: Hypogastruridae)" (PDF). Great Lakes Entomologist. 24 (2): 75–78. Retrieved 12 August 2018.
- Badri, Dayakar V.; Weir, Tiffany L.; Van der Lelie, Daniel & Vivanco, Jorge M. (2009). "Rhizosphere chemical dialogues: plant–microbe interactions". Current Opinion in Biotechnology. 20 (6): 642–50. doi:10.1016/j.copbio.2009.09.014. PMID 19875278.
- Salmon, Sandrine & Ponge, Jean-François (2001). "Earthworm excreta attract soil springtails: laboratory experiments on Heteromurus nitidus (Collembola: Entomobryidae)" (PDF). Soil Biology and Biochemistry. 33 (14): 1959–69. doi:10.1016/S0038-0717(01)00129-8. Retrieved 19 August 2018.
- Lambers, Hans; Mougel, Christophe; Jaillard, Benoît & Hinsinger, Philipe (2009). "Plant-microbe-soil interactions in the rhizosphere: an evolutionary perspective" (PDF). Plant and Soil. 321 (1/2): 83–115. doi:10.1007/s11104-009-0042-x. Retrieved 19 August 2018.
- Peñuelas, Josep; Asensio, Dolores; Tholl, Dorothea; Wenke, Katrin; Rosenkranz, Maaria; Piechulla, Birgit & Schnitzler, Jörg-Petter (2014). "Biogenic volatile emissions from the soil". Plant, Cell and Environment. 37 (8): 1866–91. doi:10.1111/pce.12340. PMID 24689847.
- Buzuleciu, Samuel A.; Crane, Derek P. & Parker, Scott L. (2016). "Scent of disinterred soil as an olfactory cue used by raccoons to locate nests of diamond-backed terrapins (Malaclemys terrapin)" (PDF). Herpetological Conservation and Biology. 11 (3): 539–51. Retrieved 19 August 2018.
- Saxton, Keith E. & Rawls, Walter J. (2006). "Soil water characteristic estimates by texture and organic matter for hydrologic solutions" (PDF). Soil Science Society of America Journal. 70 (5): 1569–78. Bibcode:2006SSASJ..70.1569S. CiteSeerX 10.1.1.452.9733. doi:10.2136/sssaj2005.0117. Retrieved 2 September 2018.
- College of Tropical Agriculture and Human Resources. "Soil Mineralogy". cms.ctahr.hawaii.edu/. University of Hawai‘i at Mānoa. Retrieved 2 September 2018.
- Sposito, Garrison (1984). The surface chemistry of soils (PDF). New York, New York: Oxford University Press. Retrieved 21 April 2019.
- Wynot, Christopher. "Theory of diffusion in colloidal suspensions" (PDF). Retrieved 21 April 2019.
- Donahue, Miller & Shickluna 1977, p. 103–06.
- Sposito, Garrison; Skipper, Neal T.; Sutton, Rebecca; Park, Sung-Ho; Soper, Alan K. & Greathouse, Jeffery A. (1999). "Surface geochemistry of the clay minerals". Proceedings of the National Academy of Sciences of the United States of America. 96 (7): 3358–64. Bibcode:1999PNAS...96.3358S. doi:10.1073/pnas.96.7.3358. PMC 34275. PMID 10097044.
- Bickmore, Barry R.; Rosso, Kevin M.; Nagy, Kathryn L.; Cygan, Randall T. & Tadanier, Christopher J. (2003). "Ab initio determination of edge surface structures for dioctahedral 2:1 phyllosilicates: implications for acid-base reactivity" (PDF). Clays and Clay Minerals. 51 (4): 359–71. Bibcode:2003CCM....51..359B. doi:10.1346/CCMN.2003.0510401. Retrieved 21 April 2019.
- Rajamathi, Michael; Thomas, Grace S. & Kamath, P. Vishnu (2001). "The many ways of making anionic clays" (PDF). Journal of Chemical Sciences. 113 (5–6): 671–80. doi:10.1007/BF02708799. Retrieved 27 April 2019.
- Moayedi, Hossein & Kazemian, Sina (2012). "Zeta potentials of suspended humus in multivalent cationic saline solution and its effect on electro-osomosis behavior" (PDF). Journal of Dispersion Science and Technology. 34 (2): 283–94. doi:10.1080/01932691.2011.646601. Retrieved 27 April 2019.
- Pettit, Robert E. "Organic matter, humus, humate, humic acid, fulvic acid and humin: their importance in soil fertility and plant health" (PDF). Retrieved 27 April 2019.
- Diamond, Sidney & Kinter, Earl B. (1965). "Mechanisms of soil-lime stabilization: an interpretative review" (PDF). Highway Research Record. 92: 83–102. Retrieved 27 April 2019.
- Woodruff, Clarence M. (1955). "The energies of replacement of calcium by potassium in soils" (PDF). Soil Science Society of America Journal. 19 (2): 167–71. Bibcode:1955SSASJ..19..167W. doi:10.2136/sssaj1955.03615995001900020014x. Retrieved 28 April 2019.
- Fronæus, Sture (1953). "On the application of the mass action law to cation exchange equilibria" (PDF). Acta Chemica Scandinavica. 7: 469–80. doi:10.3891/acta.chem.scand.07-0469. Retrieved 4 May 2019.
- Bolland, Mike D. A.; Posner, Alan M.; Quirk, James P. (1980). "pH-independent and pH-dependent surface charges on kaolinite". Clays and Clay Minerals. 28 (6): 412–18. Bibcode:1980CCM....28..412B. doi:10.1346/CCMN.1980.0280602.
- Silber, Avner; Levkovitch, Irit; Graber, Ellen R. (2010). "pH-dependent mineral release and surface properties of cornstraw biochar: agronomic implications" (PDF). Environmental Science and Technology. 44 (24): 9318–23. Bibcode:2010EnST...44.9318S. doi:10.1021/es101283d. PMID 21090742. Retrieved 4 May 2019.
- Dakora, Felix D.; Phillips, Donald D. (2002). "Root exudates as mediators of mineral acquisition in low-nutrient environments" (PDF). Plant and Soil. 245: 35–47. doi:10.1023/A:1020809400075. Archived from the original (PDF) on 19 August 2019. Retrieved 25 July 2019.
- Donahue, Miller & Shickluna 1977, p. 114.
- Singh, Jamuna Sharan; Raghubanshi, Akhilesh Singh; Singh, Raj S. & Srivastava, S. C. (1989). "Microbial biomass acts as a source of plant nutrient in dry tropical forest and savanna" (PDF). Nature. 338 (6215): 499–500. Bibcode:1989Natur.338..499S. doi:10.1038/338499a0. Retrieved 12 May 2019.
- Szatanik-Kloc, Alicja; Szerement, Justyna & Józefaciuk, Grzegorz (2017). "The role of cell walls and pectins in cation exchange and surface area of plant roots" (PDF). Journal of Plant Physiology. 215: 85–90. doi:10.1016/j.jplph.2017.05.017. PMID 28600926. Retrieved 25 July 2019.
- Donahue, Miller & Shickluna 1977, pp. 115–16.
- Gu, Baohua; Schulz, Robert K. (1991). "Anion retention in soil: possible application to reduce migration of buried technetium and iodine, a review" (PDF). doi:10.2172/5980032. Retrieved 19 May 2019. Cite journal requires
|journal=(help) - Sollins, Phillip; Robertson, G. Philip & Uehara, Goro (1988). "Nutrient mobility in variable- and permanent-charge soils" (PDF). Biogeochemistry. 6 (3): 181–99. doi:10.1007/BF02182995. Retrieved 19 May 2019.
- Sanders, W. M. H. (1964). "Extraction of soil phosphate by anion-exchange membrane". New Zealand Journal of Agricultural Research. 7 (3): 427–31. doi:10.1080/00288233.1964.10416423.
- Hinsinger, Philippe (2001). "Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review" (PDF). Plant and Soil. 237 (2): 173–95. doi:10.1023/A:1013351617532. Retrieved 19 May 2019.
- Chichester, Fredrick Wesley; Harward, Moyle E. & Youngberg, Chester T. (1970). "pH dependent ion exchange properties of soils and clays from Mazama pumice". Clays and Clay Minerals. 18 (2): 81–90. Bibcode:1970CCM....18...81C. doi:10.1346/CCMN.1970.0180203.
- Robertson, Bryan. "pH requirements of freshwater aquatic life" (PDF). Retrieved 26 May 2019.
- Chang, Raymond, ed. (2010). Chemistry (PDF) (10th ed.). New York, New York: McGraw-Hill. p. 663. ISBN 9780073511092. Retrieved 26 May 2019.
- Singleton, Peter L.; Edmeades, Doug C.; Smart, R. E. & Wheeler, David M. (2001). "The many ways of making anionic clays" (PDF). Journal of Chemical Sciences. 113 (5–6): 671–80. doi:10.1007/BF02708799. Retrieved 27 April 2019.
- Läuchli, André & Grattan, Steve R. (2012). "Soil pH extremes" (PDF). In Shabala, Sergey (ed.). Plant stress physiology (1st ed.). Wallingford, United Kingdom: CAB International. pp. 194–209. ISBN 978-1845939953. Retrieved 2 June 2019.
- Donahue, Miller & Shickluna 1977, pp. 116–17.
- Calmano, Wolfgang; Hong, Jihua & Förstner, Ulrich (1993). "Binding and mobilization of heavy metals in contaminated sediments affected by pH and redox potential" (PDF). Water Science and Technology. 28 (8–9): 223–35. doi:10.2166/wst.1993.0622. Retrieved 9 June 2019.
- Ren, Xiaoya; Zeng, Guangming; Tang, Lin; Wang, Jingjing; Wan, Jia; Liu, Yani; Yu, Jiangfang; Yi, Huan; Ye, Shujing & Deng, Rui (2018). "Sorption, transport and biodegradation: an insight into bioavailability of persistent organic pollutants in soil" (PDF). Science of the Total Environment. 610–611: 1154–63. Bibcode:2018ScTEn.610.1154R. doi:10.1016/j.scitotenv.2017.08.089. PMID 28847136. Retrieved 9 June 2019.
- Ponge, Jean-François (2003). "Humus forms in terrestrial ecosystems: a framework to biodiversity" (PDF). Soil Biology and Biochemistry. 35 (7): 935–45. CiteSeerX 10.1.1.467.4937. doi:10.1016/S0038-0717(03)00149-4. Retrieved 9 June 2019.
- Fujii, Kazumichi (2003). "Soil acidification and adaptations of plants and microorganisms in Bornean tropical forests". Ecological Research. 29 (3): 371–81. doi:10.1007/s11284-014-1144-3.
- Kauppi, Pekka; Kämäri, Juha; Posch, Maximilian & Kauppi, Lea (1986). "Acidification of forest soils: model development and application for analyzing impacts of acidic deposition in Europe" (PDF). Ecological Modelling. 33 (2–4): 231–53. doi:10.1016/0304-3800(86)90042-6. Retrieved 10 June 2019.
- Andriesse, J. P. (1969). "A study of the environment and characteristics of tropical podzols in Sarawak (East-Malaysia)" (PDF). Geoderma. 2 (3): 201–27. Bibcode:1969Geode...2..201A. doi:10.1016/0016-7061(69)90038-X. Retrieved 10 June 2019.
- Rengasamy, Pichu (2006). "World salinization with emphasis on Australia" (PDF). Journal of Experimental Botany. 57 (5): 1017–23. doi:10.1093/jxb/erj108. PMID 16510516. Retrieved 16 June 2019.
- Arnon, Daniel I. & Johnson, Clarence M. (1942). "Influence of hydrogen ion concentration on the growth of higher plants under controlled conditions". Plant Physiology. 17 (4): 525–39. doi:10.1104/pp.17.4.525. PMC 438054. PMID 16653803.
- Chaney, Rufus L.; Brown, John C. & Tiffin, Lee O. (1972). "Obligatory reduction of ferric chelates in iron uptake by soybeans". Plant Physiology. 50 (2): 208–13. doi:10.1104/pp.50.2.208. PMC 366111. PMID 16658143.
- Donahue, Miller & Shickluna 1977, pp. 116–19.
- Ahmad, Sagheer; Ghafoor, Abdul; Qadir, Manzoor & Aziz, M. Abbas (2006). "Amelioration of a calcareous saline-sodic soil by gypsum application and different crop rotations" (PDF). International Journal of Agriculture and Biology. 8 (2): 142–46. Retrieved 16 June 2019.
- McFee, William W.; Kelly, J. M. & Beck, R. H. (1976). "Acid precipitation effects on soil pH and base saturation of exchange sites" (PDF). USDA Forest Service, Northeastern Research Station, General Technical Reports. NE-23 (3): 725–35. Bibcode:1977WASP....7..401M. CiteSeerX 10.1.1.549.37. doi:10.1007/BF00284134. Retrieved 23 June 2019.
- Farina, Martin Patrick W.; Sumner, Malcolm E.; Plank, C. O. & Letzsch, W. Stephen (1980). "Exchangeable aluminum and pH as indicators of lime requirement for corn" (PDF). Soil Science Society of America Journal. 44 (5): 1036–41. Bibcode:1980SSASJ..44.1036F. doi:10.2136/sssaj1980.03615995004400050033x. Retrieved 30 June 2019.
- Donahue, Miller & Shickluna 1977, pp. 119–20.
- Sposito, Garrison; Skipper, Neal T.; Sutton, Rebecca; Park, Sun-Ho; Soper, Alan K. & Greathouse, Jeffery A. (1999). "Surface geochemistry of the clay minerals" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 96 (7): 3358–64. Bibcode:1999PNAS...96.3358S. doi:10.1073/pnas.96.7.3358. PMC 34275. PMID 10097044. Retrieved 7 July 2019.
- Sparks, Donald L. "Soil buffering and acidic and basic soils" (PDF). University of California, Department of Land, Air, and Water Resources. Retrieved 7 July 2019.
- Ulrich, Bernhard (1983). "Soil acidity and its relations to acid deposition". In Ulrich, Bernhard; Pankrath, Jürgen (eds.). Effects of accumulation of air pollutants in forest ecosystems (1st ed.). Dordrecht, The Netherlands: D. Reidel Publishing Company. pp. 127–46. ISBN 978-94-009-6985-8.
- Donahue, Miller & Shickluna 1977, pp. 120–21.
- Dean 1957, p. 80.
- Russel 1957, pp. 123–25.
- Brady, Nyle C.; Weil, Ray R. (2008). The nature and properties of soils (14th ed.). Upper Saddle River: Pearson.
- Van der Ploeg, Rienk R.; Böhm, Wolfgang & Kirkham, Mary Beth (1999). "On the origin of the theory of mineral nutrition of plants and the Law of the Minimum". Soil Science Society of America Journal. 63 (5): 1055–62. Bibcode:1999SSASJ..63.1055V. CiteSeerX 10.1.1.475.7392. doi:10.2136/sssaj1999.6351055x.
- Knecht, Magnus F. & Göransson, Anders (2004). "Terrestrial plants require nutrients in similar proportions". Tree Physiology. 24 (4): 447–60. doi:10.1093/treephys/24.4.447. PMID 14757584.
- Dean 1957, pp. 80–81.
- Roy, R. N.; Finck, Arnold; Blair, Graeme J.; Tandon, Hari Lal Singh (2006). "Chapter 4: Soil fertility and crop production" (PDF). Plant nutrition for food security: a guide for integrated nutrient management. Rome, Italy: Food and Agriculture Organization of the United Nations. pp. 43–90. ISBN 978-92-5-105490-1. Retrieved 21 July 2019.
- Parfitt, Roger L.; Giltrap, Donna J. & Whitton, Joe S. (1995). "Contribution of organic matter and clay minerals to the cation exchange capacity of soil" (PDF). Communications in Soil Science and Plant Analysis. 26 (9–10): 1343–55. doi:10.1080/00103629509369376. Retrieved 28 July 2019.
- Hajnos, Mieczyslaw; Jozefaciuk, Grzegorz; Sokołowska, Zofia; Greiffenhagen, Andreas & Wessolek, Gerd (2003). "Water storage, surface, and structural properties of sandy forest humus horizons" (PDF). Journal of Plant Nutrition and Soil Science. 166 (5): 625–34. doi:10.1002/jpln.200321161. Retrieved 28 July 2019.
- Donahue, Miller & Shickluna 1977, pp. 123–31.
- Donahue, Miller & Shickluna 1977, p. 125.
- Föhse, Doris; Claassen, Norbert & Jungk, Albrecht (1991). "Phosphorus efficiency of plants. II. Significance of root radius, root hairs and cation-anion balance for phosphorus influx in seven plant species" (PDF). Plant and Soil. 132 (2): 261–72. doi:10.1007/BF00010407. Retrieved 4 August 2019.
- Barber, Stanley A.; Walker, J. M. & Vasey, Edfred H. (1963). "Mechanisms for movement of plant nutrients from soil and fertilizer to plant root". Journal of Agricultural and Food Chemistry. 11 (3): 204–07. doi:10.1021/jf60127a017.
- Lawrence, Gregory B.; David, Mark B. & Shortle, Walter C. (1995). "A new mechanism for calcium loss in forest floor soils" (PDF). Nature. 378 (6553): 162–65. Bibcode:1995Natur.378..162L. doi:10.1038/378162a0. Retrieved 11 August 2019.
- Kramers, Hendrik Anthony (1940). "Brownian motion in a field of force and the diffusion model of chemical reactions" (PDF). Physica. 7 (4): 284–304. Bibcode:1940Phy.....7..284K. doi:10.1016/S0031-8914(40)90098-2. Retrieved 11 August 2019.
- Lin, Sijie; Reppert, Jason; Hu, Qian; Hudson, Joan S.; Reid, Michelle L.; Ratnikova, Tatsiana A.; Rao, Apparao M.; Luo, Hong & Ke, Pu Chun (2009). "Uptake, translocation, and transmission of carbon nanomaterials in rice plants" (PDF). Small. 5 (10): 1128–32. doi:10.1002/smll.200801556. PMID 19235197. Retrieved 11 August 2019.
- Donahue, Miller & Shickluna 1977, p. 126.
- "Plant nutrition". Northern Arizona University. Archived from the original on 14 May 2013. Retrieved 18 April 2019.
- Matimati, Ignatious; Verboom, G. Anthony & Cramer, Michael D. (2014). "Nitrogen regulation of transpiration controls mass-flow acquisition of nutrients". Journal of Experimental Botany. 65 (1): 159–68. doi:10.1093/jxb/ert367. PMC 3883293. PMID 24231035.
- Mengel, Dave. "Roots, growth and nutrient uptake" (PDF). Purdue University, Agronomy Department. Retrieved 18 April 2019.
- Sattelmacher, Burkhard (2001). "The apoplast and its significance for plant mineral nutrition". New Phytologist. 149 (2): 167–92. doi:10.1046/j.1469-8137.2001.00034.x.
- Hinsinger, Philippe; Plassard, Claude; Tang, Caixian & Jaillard, Benoît (2003). "Origins of root-mediated pH changes in the rhizosphere and their responses to environmental constraints: a review" (PDF). Plant and Soil. 248 (1): 43–59. doi:10.1023/A:1022371130939. Retrieved 18 August 2019.
- Chapin, F. Stuart III; Vitousek, Peter M. & Van Cleve, Keith (1986). "The nature of nutrient limitation in plant communities". American Naturalist. 127 (1): 48–58. doi:10.1086/284466. JSTOR 2461646.
- Alam, Syed Manzoor (1999). "Nutrient uptake by plants under stress conditions" (PDF). In Pessarakli, Mohammad (ed.). Handbook of plant and crop stress (2nd ed.). New York, New York: Marcel Dekker. pp. 285–313. ISBN 978-0824719487. Retrieved 1 September 2019.
- Donahue, Miller & Shickluna 1977, pp. 123–28.
- Rasmussen, Jim & Kuzyakov, Yakov (2009). "Carbon isotopes as proof for plant uptake of organic nitrogen: relevance of inorganic carbon uptake" (PDF). Soil Biology and Biochemistry. 41 (7): 1586–87. doi:10.1016/j.soilbio.2009.03.006. Retrieved 8 September 2019.
- Fitter, Alastair H.; Graves, Jonathan D.; Watkins, N. K.; Robinson, David & Scrimgeour, Charlie (1998). "Carbon transfer between plants and its control in networks of arbuscular mycorrhizas". Functional Ecology. 12 (3): 406–12. doi:10.1046/j.1365-2435.1998.00206.x.
- Manzoni, Stefano; Trofymow, John A.; Jackson, Robert B. & Porporato, Amilcare (2010). "Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter" (PDF). Ecological Monographs. 80 (1): 89–106. doi:10.1890/09-0179.1. Retrieved 15 September 2019.
- Teskey, Robert O.; Saveyn, An; Steppe, Kathy & McGuire, Mary Ann (2007). "Origin, fate and significance of CO2 in tree stems". New Phytologist. 177 (1): 17–32. doi:10.1111/j.1469-8137.2007.02286.x. PMID 18028298.
- Billings, William Dwight & Godfrey, Paul Joseph (1967). "Photosynthetic utilization of internal carbon dioxide by hollow-stemmed plants". Science. 158 (3797): 121–23. Bibcode:1967Sci...158..121B. doi:10.1126/science.158.3797.121. JSTOR 1722393. PMID 6054809.
- Wadleigh 1957, p. 41.
- Broadbent 1957, p. 153.
- Vitousek, Peter M. (1984). "Litterfall, nutrient cycling, and nutrient limitation in tropical forests" (PDF). Ecology. 65 (1): 285–98. doi:10.2307/1939481. JSTOR 1939481. Retrieved 29 September 2019.
- Donahue, Miller & Shickluna 1977, p. 128.
- Forde, Bryan G. & Clarkson, David T. (1999). "Nitrate and ammonium nutrition of plants: physiological and molecular perspectives" (PDF). Advances in Botanical Research. 30 (C): 1–90. doi:10.1016/S0065-2296(08)60226-8. Retrieved 29 September 2019.
- Hodge, Angela; Campbell, Colin D. & Fitter, Alastair H. (2001). "An arbuscular mycorrhizal fungus accelerates decomposition and acquires nitrogen directly from organic material" (PDF). Nature. 413 (6853): 297–99. Bibcode:2001Natur.413..297H. doi:10.1038/35095041. PMID 11565029. Retrieved 29 September 2019.
- Burke, Ingrid C.; Yonker, Caroline M.; Parton, William J.; Cole, C. Vernon; Flach, Klaus & Schimel, David S. (1989). "Texture, climate, and cultivation effects on soil organic matter content in U.S. grassland soils" (PDF). Soil Science Society of America Journal. 53 (3): 800–05. Bibcode:1989SSASJ..53..800B. doi:10.2136/sssaj1989.03615995005300030029x. Retrieved 6 October 2019.
- Enríquez, Susana; Duarte, Carlos M. & Sand-Jensen, Kaj (1993). "Patterns in decomposition rates among photosynthetic organisms: the importance of detritus C:N:P content" (PDF). Oecologia. 94 (4): 457–71. Bibcode:1993Oecol..94..457E. doi:10.1007/BF00566960. PMID 28313985. Retrieved 6 October 2019.
- Tiessen, Holm; Stewart, John W. B. & Bettany, Jeff R. (1982). "Cultivation effects on the amounts and concentration of carbon, nitrogen, and phosphorus in grassland soils" (PDF). Agronomy Journal. 74 (5): 831–35. doi:10.2134/agronj1982.00021962007400050015x. Retrieved 13 October 2019.
- Philippot, Laurent; Hallin, Sara & Schloter, Michael (2007). "Ecology of denitrifying prokaryotes in agricultural soil". In Sparks, Donald L. (ed.). Advances in agronomy, Volume 96. Amsterdam, The Netherlands: Elsevier. pp. 249–305. CiteSeerX 10.1.1.663.4557. ISBN 978-0-12-374206-3.
- Doran, John W. (1987). "Microbial biomass and mineralizable nitrogen distributions in no-tillage and plowed soils" (PDF). Biology and Fertility of Soils. 5 (1): 68–75. doi:10.1007/BF00264349. Retrieved 13 October 2019.
- Mahvi, Amir H.; Nouri, Jafar; Babaei, Ali A. & Nabizadeh, Ramin (2005). "Agricultural activities impact on groundwater nitrate pollution" (PDF). International Journal of Environmental Science and Technology. 2 (1): 41–47. doi:10.1007/BF03325856. Retrieved 20 October 2019.
- Scherer, Heinrich W.; Feils, E. & Beuters, Patrick (2014). "Ammonium fixation and release by clay minerals as influenced by potassium" (PDF). Plant, Soil and Environment. 60 (7): 325–31. doi:10.17221/202/2014-PSE. Retrieved 20 October 2019.
- Barak, Phillip; Jobe, Babou O.; Krueger, Armand R.; Peterson, Lloyd A. & Laird, David A. (1997). "Effects of long-term soil acidification due to nitrogen fertilizer inputs in Wisconsin" (PDF). Plant and Soil. 197 (1): 61–69. doi:10.1023/A:1004297607070. Retrieved 27 October 2019.
- Van Egmond, Klaas; Bresser, Ton & Bouwman, Lex (2002). "The European nitrogen case" (PDF). Ambio. 31 (2): 72–78. doi:10.1579/0044-7447-31.2.72. Retrieved 27 October 2019.
- Allison 1957, pp. 85–94.
- Broadbent 1957, pp. 152–55.
- Donahue, Miller & Shickluna 1977, pp. 128–31.
- Recous, Sylvie & Mary, Bruno (1990). "Microbial immobilization of ammonium and nitrate in cultivated soils" (PDF). Soil Biology and Biochemistry. 22 (7): 913–22. doi:10.1016/0038-0717(90)90129-N. Retrieved 3 November 2019.
- Verhoef, Herman A. & Brussaard, Lijbert (1990). "Decomposition and nitrogen mineralization in natural and agro-ecosystems: the contribution of soil animals" (PDF). Biogeochemistry. 11 (3): 175–211. doi:10.1007/BF00004496. Retrieved 3 November 2019.
- Chen, Baoqing; Liu, EnKe; Tian, Qizhuo; Yan, Changrong & Zhang, Yanqing (2014). "Soil nitrogen dynamics and crop residues: a review" (PDF). Agronomy for Sustainable Development. 34 (2): 429–42. doi:10.1007/s13593-014-0207-8. Retrieved 3 November 2019.
- Griffin, Timothy S.; Honeycutt, Charles W. & He, Zhijun (2002). "Effects of temperature, soil water status, and soil type on swine slurry nitrogen transformations" (PDF). Biology and Fertility of Soils. 36 (6): 442–46. doi:10.1007/s00374-002-0557-2. Retrieved 24 November 2019.
- Donahue, Miller & Shickluna 1977, pp. 129–30.
- Donahue, Miller & Shickluna 1977, p. 145.
- Lodwig, Emma; Hosie, Arthur H. F.; Bourdès, Alexandre; Findlay, Kim; Allaway, David; Karunakaran, Ramakrishnan; Downie, J. Allan & Poole, Philip S. (2003). "Amino-acid cycling drives nitrogen fixation in the legume–Rhizobium symbiosis" (PDF). Nature. 422 (6933): 722–26. Bibcode:2003Natur.422..722L. doi:10.1038/nature01527. PMID 12700763. Retrieved 10 November 2019.
- Donahue, Miller & Shickluna 1977, pp. 128–29.
- Hill, R. D.; Rinker, Robert G. & Wilson, H. Dale (1980). "Atmospheric nitrogen fixation by lightning". Journal of the Atmospheric Sciences. 37 (1): 179–92. Bibcode:1980JAtS...37..179H. doi:10.1175/1520-0469(1980)037<0179:ANFBL>2.0.CO;2.
- Allison 1957, p. 87.
- Ferris, Howard; Venette, Robert C.; Van der Meulen, Hans R. & Lau, Serrine S. (1998). "Nitrogen mineralization by bacterial-feeding nematodes: verification and measurement" (PDF). Plant and Soil. 203 (2): 159–71. doi:10.1023/A:1004318318307. Retrieved 17 November 2019.
- Violante, Antonio; de Cristofaro, Annunziata; Rao, Maria A. & Gianfreda, Liliana (1995). "Physicochemical properties of protein-smectite and protein-Al(OH)x-smectite complexes" (PDF). Clay Minerals. 30 (4): 325–36. Bibcode:1995ClMin..30..325V. doi:10.1180/claymin.1995.030.4.06. Retrieved 24 November 2019.
- Vogel, Cordula; Mueller, Carsten W.; Höschen, Carmen; Buegger, Franz; Heister, Katja; Schulz, Stefanie; Schloter, Michael & Kögel-Knabner, Ingrid (2014). "Submicron structures provide preferential spots for carbon and nitrogen sequestration in soils" (PDF). Nature Communications. 5 (2947): 1–7. Bibcode:2014NatCo...5.2947V. doi:10.1038/ncomms3947. PMC 3896754. PMID 24399306. Retrieved 24 November 2019.
- Ruamps, Léo Simon; Nunan, Naoise & Chenu, Claire (2011). "Microbial biogeography at the soil pore scale" (PDF). Soil Biology and Biochemistry. 43 (2): 280–86. doi:10.1016/j.soilbio.2010.10.010. Retrieved 1 December 2019.
- Quiquampoix, Hervé & Burns, Richard G. (2007). "Interactions between proteins and soil mineral surfaces: environmental and health consequences" (PDF). Elements. 3 (6): 401–06. doi:10.2113/GSELEMENTS.3.6.401. Retrieved 24 November 2019.
- Nieder, Rolf; Benbi, Dinesh K. & Scherer, Heinrich W. (2011). "Fixation and defixation of ammonium in soils: a review" (PDF). Biology and Fertility of Soils. 47 (1): 1–14. doi:10.1007/s00374-010-0506-4. Retrieved 8 December 2019.
- Allison 1957, p. 90.
- Kramer, Sasha B.; Reganold, John P.; Glover, Jerry D.; Bohannan, Brendan J.M. & Mooney, Harold A. (2006). "Reduced nitrate leaching and enhanced denitrifier activity and efficiency in organically fertilized soils" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 103 (12): 4522–27. Bibcode:2006PNAS..103.4522K. doi:10.1073/pnas.0600359103. PMC 1450204. PMID 16537377. Retrieved 8 December 2019.
- Robertson, G. Philip (1989). "Nitrification and denitrification in humid tropical ecosystems: potential controls on nitrogen retention" (PDF). In Proctor, John (ed.). Mineral nutrients in tropical forest and savanna ecosystems. Cambridge, Massachusetts: Blackwell Scientific. pp. 55–69. ISBN 978-0632025596. Retrieved 15 December 2019.
- Parkin, Timothy B. & Robinson, Joseph A. (1989). "Stochastic models of soil denitrification" (PDF). Applied and Environmental Microbiology. 55 (1): 72–77. doi:10.1128/AEM.55.1.72-77.1989. PMC 184056. PMID 16347838. Retrieved 15 December 2019.
- Donahue, Miller & Shickluna 1977, p. 130.
- Rao, Desiraju L.N. & Batra, Lalit (1983). "Ammonia volatilization from applied nitrogen in alkali soils" (PDF). Plant and Soil. 70 (2): 219–28. doi:10.1007/BF02374782. Retrieved 15 December 2019.
- Donahue, Miller & Shickluna 1977, p. 131.
- Lallouette, Vincent; Magnier, Julie; Petit, Katell & Michon, Janik (2014). "Agricultural practices and nitrates in aquatic environments" (PDF). The Brief. 11 (December): 1–16. Retrieved 15 December 2019.
- Choudhury, Abu T.M.A. & Kennedy, Ivan R. (2005). "Nitrogen fertilizer losses from rice soils and control of environmental pollution problems" (PDF). Communications in Soil Science and Plant Analysis. 36 (11–12): 1625–39. doi:10.1081/css-200059104. Retrieved 15 December 2019.
- Roth, Tobias; Kohli, Lukas; Rihm, Beat & Achermann, Beat (2013). "Nitrogen deposition is negatively related to species richness and species composition of vascular plants and bryophytes in Swiss mountain grassland" (PDF). Agriculture, Ecosystems and Environment. 178: 121–26. doi:10.1016/j.agee.2013.07.002. Retrieved 15 December 2019.
- Vitousek, Peter M. (1984). "Litterfall, nutrient cycling, and nutrient limitation in tropical forests" (PDF). Ecology. 65 (1): 285–98. doi:10.2307/1939481. JSTOR 1939481. Retrieved 22 December 2019.
- Kucey, Reg M.N. (1983). "Phosphate-solubilizing bacteria and fungi in various cultivated and virgin Alberta soils". Canadian Journal of Soil Science. 63 (4): 671–78. doi:10.4141/cjss83-068.
- Khorassani, Reza; Hettwer, Ursula; Ratzinger, Astrid; Steingrobe, Bernd; Karlovsky, Petr & Claassen, Norbert (2011). "Citramalic acid and salicylic acid in sugar beet root exudates solubilize soil phosphorus". BMC Plant Biology. 11 (121): 1–8. doi:10.1186/1471-2229-11-121. PMC 3176199. PMID 21871058.
- Duponnois, Robin; Colombet, Aline; Hien, Victor & Thioulouse, Jean (2005). "The mycorrhizal fungus Glomus intraradices and rock phosphate amendment influence plant growth and microbial activity in the rhizosphere of Acacia holosericea" (PDF). Soil Biology and Biochemistry. 37 (8): 1460–68. doi:10.1016/j.soilbio.2004.09.016. Retrieved 22 December 2019.
- Syers, John Keith; Johnston, A. Edward & Curtin, Denis (2008). Efficiency of soil and fertilizer phosphorus use: reconciling changing concepts of soil phosphorus behaviour with agronomic information (PDF). Rome, Italy: Food and Agriculture Organization of the United Nations. ISBN 978-92-5-105929-6. Retrieved 29 December 2019.
- Olsen & Fried 1957, p. 96.
- Lambert, Raphaël; Grant, Cynthia & Sauvé, Sébastien (2007). "Cadmium and zinc in soil solution extracts following the application of phosphate fertilizers" (PDF). Science of the Total Environment. 378 (3): 293–305. Bibcode:2007ScTEn.378..293L. doi:10.1016/j.scitotenv.2007.02.008. PMID 17400282. Retrieved 5 January 2020.
- Terry, Norman & Ulrich, Albert (1973). "Effects of phosphorus deficiency on the photosynthesis and respiration of leaves of sugar beet" (PDF). Plant Physiology. 51 (1): 43–47. doi:10.1104/pp.51.1.43. PMC 367354. PMID 16658294. Retrieved 5 January 2020.
- Pallas, James E. Jr; Michel, B.E. & Harris, D.G. (1967). "Photosynthesis, transpiration, leaf temperature, and stomatal activity of cotton plants under varying water potentials" (PDF). Plant Physiology. 42 (1): 76–88. doi:10.1104/pp.42.1.76. PMC 1086491. PMID 16656488. Retrieved 12 January 2020.
- Meena, Vijay Singh; Maurya, Bihari Ram; Verma, Jai Prakash; Aeron, Abhinav; Kumar, Ashok; Kim, Kangmin & Bajpai, Vivek K. (2015). "Potassium solubilizing rhizobacteria (KSR): isolation, identification, and K-release dynamics from waste mica" (PDF). Ecological Engineering. 81: 340–47. doi:10.1016/j.ecoleng.2015.04.065. Retrieved 12 January 2020.
- Sawhney, Brij L. (1972). "Selective sorption and fixation of cations by clay minerals: a review". Clays and Clay Minerals. 20 (2): 93–100. Bibcode:1972CCM....20...93S. doi:10.1346/CCMN.1972.0200208.
- Donahue, Miller & Shickluna 1977, pp. 134–35.
- Reitemeier 1957, pp. 101–04.
- Loide, Valli (2004). "About the effect of the contents and ratios of soil's available calcium, potassium and magnesium in liming of acid soils" (PDF). Agronomy Research. 2 (1): 71–82. Retrieved 19 January 2020.
- Wuddivira, Mark N.; Camps-Roach, Geremy (2007). "Effects of organic matter and calcium on soil structural stability" (PDF). European Journal of Soil Science. 58 (3): 722–27. doi:10.1111/j.1365-2389.2006.00861.x. Retrieved 19 January 2020.
- Donahue, Miller & Shickluna 1977, pp. 135–36.
- Smith, Garth S.; Cornforth, Ian S. (1982). "Concentrations of nitrogen, phosphorus, sulphur, magnesium, and calcium in North Island pastures in relation to plant and animal nutrition". New Zealand Journal of Agricultural Research. 25 (3): 373–87. doi:10.1080/00288233.1982.10417901.
- White, Philip J.; Broadley, Martin R. (2003). "Calcium in plants" (PDF). Annals of Botany. 92 (4): 487–511. doi:10.1093/aob/mcg164. PMC 4243668. PMID 12933363. Retrieved 19 January 2020.
- Donahue, Miller & Shickluna 1977, p. 136.
- Jarrell, Wesley M.; Beverly, Reuben B. (1981). "The dilution effect in plant nutrition studies". Advances in Agronomy. 34: 197–224. doi:10.1016/S0065-2113(08)60887-1. ISBN 9780120007349.
- Jordan & Reisenauer 1957, p. 107.
- Holmes & Brown 1957, pp. 111.
- Sherman 1957, p. 135.
- Seatz & Jurinak 1957, p. 115.
- Reuther 1957, p. 128.
- Russel 1957, p. 121.
- Stout & Johnson 1957, p. 146.
- Stout & Johnson 1957, p. 141.
- Welsh, Ross M. (1995). "Micronutrient nutrition of plants" (PDF). Critical Reviews in Plant Sciences. 14 (1): 49–82. doi:10.1080/713608066. Retrieved 9 February 2020.
- Summer, Malcolm E.; Farina, Mart P. W. (1986). "Phosphorus interactions with other nutrients and lime in field cropping systems" (PDF). In Stewart, Bobby A. (ed.). Advances in soil science. 5. New York, New York: Springer. pp. 201–36. doi:10.1007/978-1-4613-8660-5_5. ISBN 978-1-4613-8660-5. Retrieved 9 February 2020.
- Lešková, Alexandra; Giehl, Ricardo F.H.; Hartmann, Anja; Fargašová, Agáta & von Wirén, Nicolaus (2017). "Heavy metals induce iron deficiency responses at different hierarchic and regulatory levels". Plant Physiology. 174 (3): 1648–68. doi:10.1104/pp.16.01916. PMC 5490887. PMID 28500270.
- M’Sehli, Wissal; Youssfi, Sabah; Donnini, Silvia; Dell’Orto, Marta; De Nisi, Patricia; Zocchi, Graziano; Abdelly, Chedly & Gharsalli, Mohamed (2008). "Root exudation and rhizosphere acidification by two lines of Medicago ciliaris in response to lime-induced iron deficiency" (PDF). Plant and Soil. 312 (151): 151–62. doi:10.1007/s11104-008-9638-9. Retrieved 9 February 2020.
- Donahue, Miller & Shickluna 1977, pp. 136–37.
- Stout & Johnson 1957, p. 107.
- Pereira, B.F. Faria; He, Zhenli; Stoffella, Peter J.; Montes, Celia R.; Melfi, Adolpho J. & Baligar, Virupax C. (2012). "Nutrients and nonessential elements in soil after 11 Years of wastewater irrigation" (PDF). Journal of Environmental Quality. 41 (3): 920–27. doi:10.2134/jeq2011.0047. PMID 22565273. Retrieved 16 February 2020.
- Richmond, Kathryn E. & Sussman, Michael (2003). "Got silicon? The non-essential beneficial plant nutrient" (PDF). Current Opinion in Plant Biology. 6 (3): 268–72. doi:10.1016/S1369-5266(03)00041-4. PMID 12753977. Retrieved 16 February 2020.
- Pimentel, David; Harvey, Celia; Resosudarmo, Resosudarmo; Sinclair, K.; Kurz, D.; McNair, M.; Crist, S.; Shpritz, L.; Fitton, L.; Saffouri, R. & Blair, R. (1995). "Environmental and economic costs of soil erosion and conservation benefits" (PDF). Science. 267 (5201): 1117–23. Bibcode:1995Sci...267.1117P. doi:10.1126/science.267.5201.1117. PMID 17789193. Archived from the original (PDF) on 13 December 2016. Retrieved 23 February 2020.
- Schnürer, Johan; Clarholm, Marianne & Rosswall, Thomas (1985). "Microbial biomass and activity in an agricultural soil with different organic matter contents" (PDF). Soil Biology and Biochemistry. 17 (5): 611–18. doi:10.1016/0038-0717(85)90036-7. Retrieved 1 March 2020.
- Sparling, Graham P. (1992). "Ratio of microbial biomass carbon to soil organic carbon as a sensitive indicator of changes in soil organic matter" (PDF). Australian Journal of Soil Research. 30 (2): 195–207. doi:10.1071/SR9920195. Retrieved 1 March 2020.
- Varadachari, Chandrika & Ghosh, Kunal (1984). "On humus formation" (PDF). Plant and Soil. 77 (2): 305–13. doi:10.1007/BF02182933. Retrieved 8 March 2020.
- Prescott, Cindy E. (2010). "Litter decomposition: what controls it and how can we alter it to sequester more carbon in forest soils?" (PDF). Biogeochemistry. 101 (1): 133–49. doi:10.1007/s10533-010-9439-0. Retrieved 1 March 2020.
- Lehmann, Johannes & Kleber, Markus (2015). "The contentious nature of soil organic matter". Nature. 528 (7580): 60–68. Bibcode:2015Natur.528...60L. doi:10.1038/nature16069. PMID 26595271.
- Piccolo, Alessandro (2002). "The supramolecular structure of humic substances: a novel understanding of humus chemistry and implications in soil science" (PDF). Advances in Agronomy. 75: 57–134. doi:10.1016/S0065-2113(02)75003-7. ISBN 9780120007936. Retrieved 15 March 2020.
- Scheu, Stefan (2002). "The soil food web: structure and perspectives" (PDF). European Journal of Soil Biology. 38 (1): 11–20. doi:10.1016/S1164-5563(01)01117-7. Retrieved 8 March 2020.
- Foth, Henry D. (1984). Fundamentals of soil science (7th ed.). New York, New York: Wiley. p. 151. ISBN 978-0-471-88926-7.
- Ponge, Jean-François (2003). "Humus forms in terrestrial ecosystems: a framework to biodiversity". Soil Biology and Biochemistry. 35 (7): 935–45. CiteSeerX 10.1.1.467.4937. doi:10.1016/s0038-0717(03)00149-4. Retrieved 22 March 2020.
- Pettit, Robert E. "Organic matter, humus, humate, humic acid, fulvic acid and humin: their importance in soil fertility and plant health" (PDF). Retrieved 5 April 2020.
- Ji, Rong; Kappler, Andreas & Brune, Andreas (2000). "Transformation and mineralization of synthetic 14C-labeled humic model compounds by soil-feeding termites". Soil Biology and Biochemistry. 32 (8–9): 1281–91. CiteSeerX 10.1.1.476.9400. doi:10.1016/S0038-0717(00)00046-8.
- Gilluly, James; Waters, Aaron Clement; Woodford, Alfred Oswald (1975). Principles of geology (4th ed.). San Francisco, California: W.H. Freeman. p. 216. ISBN 978-0-7167-0269-6.
- Piccolo, Alessandro (1996). "Humus and soil conservation". In Piccolo, Alessandro (ed.). Humic substances in terrestrial ecosystems. Amsterdam, The Netherlands: Elsevier. pp. 225–64. doi:10.1016/B978-044481516-3/50006-2. ISBN 978-0-444-81516-3. Retrieved 22 March 2020.
- Varadachari, Chandrika & Ghosh, Kunal (1984). "On humus formation". Plant and Soil. 77 (2): 305–13. doi:10.1007/BF02182933. Retrieved 5 April 2020.
- Mendonça, Eduardo S. & Rowell, David L. (1996). "Mineral and organic fractions of two oxisols and their influence on effective cation-exchange capacity". Soil Science Society of America Journal. 60 (6): 1888–92. Bibcode:1996SSASJ..60.1888M. doi:10.2136/sssaj1996.03615995006000060038x. Retrieved 12 April 2020.
- Heck, Tobias; Faccio, Greta; Richter, Michael & Thöny-Meyer, Linda (2013). "Enzyme-catalyzed protein crosslinking". Applied Microbiology and Biotechnology. 97 (2): 461–75. doi:10.1007/s00253-012-4569-z. PMC 3546294. PMID 23179622.
- Lynch, D. L. & Lynch, C. C. (1958). "Resistance of protein–lignin complexes, lignins and humic acids to microbial attack". Nature. 181 (4621): 1478–79. Bibcode:1958Natur.181.1478L. doi:10.1038/1811478a0. PMID 13552710.
- Dawson, Lorna A. & Hillier, Stephen (2010). "Measurement of soil characteristics for forensic applications" (PDF). Surface and Interface Analysis. 42 (5): 363–77. doi:10.1002/sia.3315. Retrieved 19 April 2020.
- Manjaiah, K.M.; Kumar, Sarvendra; Sachdev, M. S.; Sachdev, P. & Datta, S. C. (2010). "Study of clay–organic complexes". Current Science. 98 (7): 915–21. Retrieved 19 April 2020.
- Theng, Benny K.G. (1982). "Clay-polymer interactions: summary and perspectives". Clays and Clay Minerals. 30 (1): 1–10. Bibcode:1982CCM....30....1T. CiteSeerX 10.1.1.608.2942. doi:10.1346/CCMN.1982.0300101.
- Tietjen, Todd & Wetzel, Robert G. (2003). "Extracellular enzyme-clay mineral complexes: enzyme adsorption, alteration of enzyme activity, and protection from photodegradation" (PDF). Aquatic Ecology. 37 (4): 331–39. doi:10.1023/B:AECO.0000007044.52801.6b. Retrieved 26 April 2020.
- Melero, Sebastiana; Madejón, Engracia; Ruiz, Juan Carlos & Herencia, Juan Francisco (2007). "Chemical and biochemical properties of a clay soil under dryland agriculture system as affected by organic fertilization". European Journal of Agronomy. 26 (3): 327–34. doi:10.1016/j.eja.2006.11.004.
- Joanisse, Gilles D.; Bradley, Robert L.; Preston, Caroline M. & Bending, Gary D. (2009). "Sequestration of soil nitrogen as tannin–protein complexes may improve the competitive ability of sheep laurel (Kalmia angustifolia) relative to black spruce (Picea mariana)". New Phytologist. 181 (1): 187–98. doi:10.1111/j.1469-8137.2008.02622.x. PMID 18811620.
- Fierer, Noah; Schimel, Joshua P.; Cates, Rex G. & Zou, Jiping (2001). "Influence of balsam poplar tannin fractions on carbon and nitrogen dynamics in Alaskan taiga floodplain soils". Soil Biology and Biochemistry. 33 (12–13): 1827–39. doi:10.1016/S0038-0717(01)00111-0. Retrieved 3 May 2020.
- Ponge, Jean-François (2003). "Humus forms in terrestrial ecosystems: a framework to biodiversity" (PDF). Soil Biology and Biochemistry. 35 (7): 935–45. CiteSeerX 10.1.1.467.4937. doi:10.1016/S0038-0717(03)00149-4. Archived from the original on 29 January 2016.
- Peng, Xinhua & Horn, Rainer (2007). "Anisotropic shrinkage and swelling of some organic and inorganic soils". European Journal of Soil Science. 58 (1): 98–107. doi:10.1111/j.1365-2389.2006.00808.x.
- Wang, Yang; Amundson, Ronald & Trumbmore, Susan (1996). "Radiocarbon dating of soil organic matter" (PDF). Quaternary Research. 45 (3): 282–88. doi:10.1006/qres.1996.0029. Retrieved 10 May 2020.
- Brodowski, Sonja; Amelung, Wulf; Haumaier, Ludwig & Zech, Wolfgang (2007). "Black carbon contribution to stable humus in German arable soils". Geoderma. 139 (1–2): 220–28. doi:10.1016/j.geoderma.2007.02.004. Retrieved 17 May 2020.
- Criscuoli, Irene; Alberti, Giorgio; Baronti, Silvia; Favilli, Filippo; Martinez, Cristina; Calzolari, Costanza; Pusceddu, Emanuela; Rumpel, Cornelia; Viola, Roberto & Miglietta, Franco (2014). "Carbon sequestration and fertility after centennial time scale incorporation of charcoal into soil". PLOS ONE. 9 (3): e91114. doi:10.1371/journal.pone.0091114. PMC 3948733. PMID 24614647.
- Wagai, Rota; Mayer, Lawrence M.; Kitayama, Kanehiro & Knicker, Heike (2008). "Climate and parent material controls on organic matter storage in surface soils: a three-pool, density-separation approach". Geoderma. 147 (1–2): 23–33. Bibcode:2008Geode.147...23W. doi:10.1016/j.geoderma.2008.07.010. Retrieved 24 May 2020.
- Minayeva, Tatiana Y.; Trofimov, Sergey Ya.; Chichagova, Olga A.; Dorofeyeva, E. I.; Sirin, Andrey A.; Glushkov, Igor V.; Mikhailov, N. D.; Kromer, Bernd (2008). "Carbon accumulation in soils of forest and bog ecosystems of southern Valdai in the Holocene". Biology Bulletin. 35 (5): 524–32. doi:10.1134/S1062359008050142. Retrieved 24 May 2020.
- Vitousek, Peter M. & Sanford, Robert L. (1986). "Nutrient cycling in moist tropical forest". Annual Review of Ecology and Systematics. 17: 137–67. doi:10.1146/annurev.es.17.110186.001033.
- Rumpel, Cornelia; Chaplot, Vincent; Planchon, Olivier; Bernadou, J.; Valentin, Christian & Mariotti, André (2006). "Preferential erosion of black carbon on steep slopes with slash and burn agriculture". Catena. 65 (1): 30–40. doi:10.1016/j.catena.2005.09.005. Retrieved 31 May 2020.
- Paul, Eldor A.; Paustian, Keith H.; Elliott, E. T.; Cole, C. Vernon (1997). Soil organic matter in temperate agroecosystems: long-term experiments in North America. Boca Raton, Florida: CRC Press. p. 80. ISBN 978-0-8493-2802-2.
- "Horizons". Soils of Canada. Archived from the original on 22 September 2019. Retrieved 7 June 2020.
- Frouz, Jan; Prach, Karel; Pizl, Václav; Háněl, Ladislav; Starý, Josef; Tajovský, Karel; Materna, Jan; Balík, Vladimír; Kalčík, Jiří & Řehounková, Klára (2008). "Interactions between soil development, vegetation and soil fauna during spontaneous succession in post mining sites" (PDF). European Journal of Soil Biology. 44 (1): 109–21. doi:10.1016/j.ejsobi.2007.09.002. Retrieved 21 June 2020.
- Kabala, Cezary & Zapart, Justyna (2012). "Initial soil development and carbon accumulation on moraines of the rapidly retreating Werenskiold Glacier, SW Spitsbergen, Svalbard archipelago" (PDF). Geoderma. 175–176: 9–20. doi:10.1016/j.geoderma.2012.01.025. Retrieved 21 June 2020.
- Ugolini, Fiorenzo C. & Dahlgren, Randy A. (2002). "Soil development in volcanic ash" (PDF). Global Environmental Research. 6 (2): 69–81. Retrieved 28 June 2020.
- Huggett, Richard J. (1998). "Soil chronosequences, soil development, and soil evolution: a critical review" (PDF). Catena. 32 (3): 155–72. doi:10.1016/S0341-8162(98)00053-8. Retrieved 28 June 2020.
- De Alba, Saturnio; Lindstrom, Michael; Schumacher, Thomas E. & Malo, Douglas D. (2004). "Soil landscape evolution due to soil redistribution by tillage: a new conceptual model of soil catena evolution in agricultural landscapes" (PDF). Catena. 58 (1): 77–100. doi:10.1016/j.catena.2003.12.004. Retrieved 28 June 2020.
- Phillips, Jonathan D. & Marion, Daniel A. (2004). "Pedological memory in forest soil development" (PDF). Forest Ecology and Management. 188 (1): 363–80. doi:10.1016/j.foreco.2003.08.007. Retrieved 5 July 2020.
- Mitchell, Edward A.D.; Van der Knaap, Willem O.; Van Leeuwen, Jacqueline F.N.; Buttler, Alexandre; Warner, Barry G. & Gobat, Jean-Michel (2001). "The palaeoecological history of the Praz-Rodet bog (Swiss Jura) based on pollen, plant macrofossils and testate amoebae(Protozoa)" (PDF). The Holocene. 11 (1): 65–80. doi:10.1191/095968301671777798. Retrieved 5 July 2020.
- Carcaillet, Christopher (2001). "Soil particles reworking evidences by AMS 14C dating of charcoal" (PDF). Comptes Rendus de l'Académie des Sciences, Series IIA, Earth and Planetary Science. 332 (1): 21–28. doi:10.1016/S1251-8050(00)01485-3. Retrieved 14 June 2020.
- Retallack, Gregory J. (1991). "Untangling the effects of burial alteration and ancient soil formation" (PDF). Annual Review of Earth and Planetary Sciences. 19 (1): 183–206. doi:10.1146/annurev.ea.19.050191.001151. Retrieved 14 June 2020.
- Bakker, Martha M.; Govers, Gerard; Jones, Robert A. & Rounsevell, Mark D.A. (2007). "The effect of soil erosion on Europe's crop yields" (PDF). Ecosystems. 10 (7): 1209–19. doi:10.1007/s10021-007-9090-3. Retrieved 19 July 2020.
- Uselman, Shauna M.; Qualls, Robert G. & Lilienfein, Juliane (2007). "Contribution of root vs. leaf litter to dissolved organic carbon leaching through soil" (PDF). Soil Science Society of America Journal. 71 (5): 1555–63. doi:10.2136/sssaj2006.0386. Retrieved 19 July 2020.
- Schulz, Stefanie; Brankatschk, Robert; Dümig, Alexander; Kögel-Knabner, Ingrid; Schloter, Michae & Zeyer, Josef (2013). "The role of microorganisms at different stages of ecosystem development for soil formation" (PDF). Biogeosciences. 10 (6): 3983–96. doi:10.5194/bg-10-3983-2013. Archived from the original (PDF) on 19 July 2020. Retrieved 19 July 2020.
- Gillet, Servane & Ponge, Jean-François (2002). "Humus forms and metal pollution in soil" (PDF). European Journal of Soil Science. 53 (4): 529–39. doi:10.1046/j.1365-2389.2002.00479.x. Retrieved 12 July 2020.
- Bardy, Marion; Fritsch, Emmanuel; Derenne, Sylvie; Allard, Thierry; do Nascimento, Nadia Régina & Bueno, Guilherme (2008). "Micromorphology and spectroscopic characteristics of organic matter in waterlogged podzols of the upper Amazon basin" (PDF). Geoderma. 145 (3): 222–30. doi:10.1016/j.geoderma.2008.03.008. Retrieved 19 July 2020.
- Dokuchaev, Vasily Vasilyevich (1967). "Russian Chernozem" (PDF). Jerusalem, Israel: Israel Program for Scientific Translations. Retrieved 26 July 2020.
- IUSS Working Group WRB (2015). World Reference Base for Soil Resources 2014: international soil classification system for naming soils and creating legends for soil maps, update 2015 (PDF). Rome, Italy: Food and Agriculture Organization. ISBN 978-92-5-108370-3. Retrieved 26 July 2020.
- AlShrouf, Ali (2017). "Hydroponics, aeroponic and aquaponic as compared with conventional farming" (PDF). American Scientific Research Journal for Engineering, Technology, and Sciences. 27 (1): 247–55. Retrieved 2 August 2020.
- Leake, Simon; Haege, Elke (2014). Soils for landscape development: selection, specification and validation. Clayton, Victoria, Australia: CSIRO Publishing. ISBN 978-0643109650.
- Pan, Xian-Zhang; Zhao, Qi-Guo (2007). "Measurement of urbanization process and the paddy soil loss in Yixing city, China between 1949 and 2000" (PDF). Catena. 69 (1): 65–73. Retrieved 2 August 2020.
- Kopittke, Peter M.; Menzies, Neal W.; Wang, Peng; McKenna, Brigid A.; Lombi, Enzo (2019). "Soil and the intensification of agriculture for global food security" (PDF). Environment International. 132: 105078. doi:10.1016/j.envint.2019.105078. ISSN 0160-4120. PMID 31400601. Retrieved 9 August 2020.
- Stürck, Julia; Poortinga, Ate; Verburg, Peter H. (2014). "Mapping ecosystem services: the supply and demand of flood regulation services in Europe" (PDF). Ecological Indicators. 38: 198–211. doi:10.1016/j.ecolind.2013.11.010. Retrieved 16 August 2020.
- Van Cuyk, Sheila; Siegrist, Robert; Logan, Andrew; Masson, Sarah; Fischer, Elizabeth; Figueroa, Linda (2001). "Hydraulic and purification behaviors and their interactions during wastewater treatment in soil infiltration systems" (PDF). Water Research. 35 (4): 953–64. doi:10.1016/S0043-1354(00)00349-3. Retrieved 9 August 2020.
- Jeffery, Simon; Gardi, Ciro; Arwyn, Jones (2010). European atlas of soil biodiversity (PDF). Luxembourg, Luxembourg: Publications Office of the European Union. doi:10.2788/94222. ISBN 978-92-79-15806-3. Retrieved 9 August 2020.
- De Deyn, Gerlinde B.; Van der Putten, Wim H. (2005). "Linking aboveground and belowground diversity" (PDF). Trends in Ecology and Evolution. 20 (11): 625–33. doi:10.1016/j.tree.2005.08.009. PMID 16701446. Retrieved 16 August 2020.
- Hansen, J.; et al. (2008). "Target atmospheric CO2: Where should humanity aim?". Open Atmospheric Science Journal. 2 (1): 217–31. arXiv:0804.1126. Bibcode:2008OASJ....2..217H. doi:10.2174/1874282300802010217.
- Lal, R. (11 June 2004). "Soil Carbon Sequestration Impacts on Global Climate Change and Food Security". Science. 304 (5677): 1623–27. Bibcode:2004Sci...304.1623L. doi:10.1126/science.1097396. PMID 15192216.
- Blakeslee, Thomas R. (24 February 2010). "Greening Deserts for Carbon Credits". Renewable Energy World.com. Archived from the original on 1 November 2012. Retrieved 23 October 2012.
- Setz, EZF; Enzweiler J; Solferini VN; Amendola MP; Berton RS (1999). "Geophagy in the golden-faced saki monkey (Pithecia pithecia chrysocephala) in the Central Amazon" (PDF). Journal of Zoology. 247 (1): 91–103. doi:10.1111/j.1469-7998.1999.tb00196.x.
- Kohne, John Maximilian; Koehne, Sigrid; Simunek, Jirka (2009). "A review of model applications for structured soils: a) Water flow and tracer transport" (PDF). Journal of Contaminant Hydrology. 104 (1–4): 4–35. Bibcode:2009JCHyd.104....4K. CiteSeerX 10.1.1.468.9149. doi:10.1016/j.jconhyd.2008.10.002. PMID 19012994. Archived from the original (PDF) on 7 November 2017. Retrieved 1 November 2017.
- Diplock, EE; Mardlin DP; Killham KS; Paton GI (2009). "Predicting bioremediation of hydrocarbons: laboratory to field scale". Environmental Pollution. 157 (6): 1831–40. doi:10.1016/j.envpol.2009.01.022. PMID 19232804.
- Moeckel, Claudia; Nizzetto, Luca; Di Guardo, Antonio; Steinnes, Eiliv; Freppaz, Michele; Filippa, Gianluca; Camporini, Paolo; Benner, Jessica; Jones, Kevin C. (2008). "Persistent organic pollutants in boreal and montane soil profiles: distribution, evidence of processes and implications for global cycling". Environmental Science and Technology. 42 (22): 8374–80. Bibcode:2008EnST...42.8374M. doi:10.1021/es801703k. PMID 19068820.
- Rezaei, Khalil; Guest, Bernard; Friedrich, Anke; Fayazi, Farajollah; Nakhaei, Mohamad; Aghda, Seyed Mahmoud Fatemi; Beitollahi, Ali (2009). "Soil and sediment quality and composition as factors in the distribution of damage at the December 26, 2003, Bam area earthquake in SE Iran (M (s)=6.6)". Journal of Soils and Sediments. 9: 23–32. doi:10.1007/s11368-008-0046-9.
- Johnson, D.L.; Ambrose, S.H.; Bassett, T.J.; Bowen, M.L.; Crummey, D.E.; Isaacson, J.S.; Johnson, D.N.; Lamb, P.; Saul, M.; Winter-Nelson, A. E. (1997). "Meanings of environmental terms". Journal of Environmental Quality. 26 (3): 581–89. doi:10.2134/jeq1997.00472425002600030002x.
- Ballabio, Cristiano; Panagos, Panos; Lugato, Emanuele; Huang, Jen-How; Orgiazzi, Alberto; Jones, Arwyn; Fernández-Ugalde, Oihane; Borrelli, Pasquale; Montanarella, Luca (15 September 2018). "Copper distribution in European topsoils: An assessment based on LUCAS soil survey". Science of the Total Environment. 636: 282–298. Bibcode:2018ScTEn.636..282B. doi:10.1016/j.scitotenv.2018.04.268. ISSN 0048-9697. PMID 29709848.
- Jones, j. a. a. (1976). "Soil piping and stream channel initiation". Water Resources Research. 7 (3): 602–10. Bibcode:1971WRR.....7..602J. doi:10.1029/WR007i003p00602.
- Dooley, Alan (June 2006). "Sandboils 101: Corps has experience dealing with common flood danger". Engineer Update. US Army Corps of Engineers. Archived from the original on 18 April 2008. Retrieved 14 May 2008.
- ILRI (1989). "Effectiveness and Social/Environmental Impacts of Irrigation Projects: a Review" (PDF). In: Annual Report 1988 of the International Institute for Land Reclamation and Improvement (ILRI). Wageningen, The Netherlands. pp. 18–34. Archived (PDF) from the original on 19 February 2009.
- Drainage Manual: A Guide to Integrating Plant, Soil, and Water Relationships for Drainage of Irrigated Lands. Interior Dept., Bureau of Reclamation. 1993. ISBN 978-0-16-061623-5.
- "Free articles and software on drainage of waterlogged land and soil salinity control". Archived from the original on 16 August 2010. Retrieved 28 July 2010.
- International Water Management Institute (2010). "Improving soils and boosting yields in Thailand" (PDF). Success Stories (2). doi:10.5337/2011.0031. Archived (PDF) from the original on 7 June 2012.
- "Provide for your garden's basic needs ... and the plants will take it from there". USA Weekend. 10 March 2011. Archived from the original on 9 February 2013.
- Hillel, Daniel (1993). Out of the Earth: civilization and the life of the soil. Berkeley: University of California Press. ISBN 978-0-520-08080-5.
- Donahue, Miller & Shickluna 1977, p. 4.
- Kellogg 1957, p. 1.
- Ibn al-'Awwam (1864). Le livre de l'agriculture, traduit de l'arabe par Jean Jacques Clément-Mullet (PDF). Filāḥah.French (in French). Paris: Librairie A. Franck. Retrieved 17 December 2017.
- Jelinek, Lawrence J. (1982). Harvest empire: a history of California agriculture. San Francisco: Boyd and Fraser. ISBN 978-0-87835-131-2.
- de Serres, Olivier (1600). Le Théâtre d'Agriculture et mesnage des champs (in French). Paris: Jamet Métayer. Retrieved 17 December 2017.
- Virto, Iñigo; Imaz, María José; Fernández-Ugalde, Oihane; Gartzia-Bengoetxea, Nahia; Enrique, Alberto & Bescansa, Paloma (2015). "Soil degradation and soil quality in western Europe: current situation and future perspectives". Sustainability. 7 (1): 313–65. doi:10.3390/su7010313.
- Van der Ploeg, Rienk R.; Schweigert, Peter & Bachmann, Joerg (2001). "Use and misuse of nitrogen in agriculture: the German story". Scientific World Journal. 1 (S2): 737–44. doi:10.1100/tsw.2001.263. PMC 6084271. PMID 12805882.
- Brady, Nyle C. (1984). The nature and properties of soils (9th ed.). New York: Collier Macmillan. ISBN 978-0-02-313340-4.
- Kellogg 1957, p. 3.
- Kellogg 1957, p. 2.
- de Lavoisier, Antoine-Laurent (1777). "Mémoire sur la combustion en général" (PDF). Mémoires de l'Académie Royale des Sciences (in French). Retrieved 17 December 2017.
- Boussingault, Jean-Baptiste (1860–1874). Agronomie, chimie agricole et physiologie, volumes 1–5 (PDF) (in French). Paris: Mallet-Bachelier. Retrieved 17 December 2017.
- von Liebig, Justus (1840). Organic chemistry in its applications to agriculture and physiology (PDF). London: Taylor and Walton. Retrieved 17 December 2017.
- Way, J. Thomas (1849). "On the composition and money value of the different varieties of guano". Journal of the Royal Agricultural Society of England. 10: 196–230. Retrieved 17 December 2017.
- Kellogg 1957, p. 4.
- Tandon, Hari L.S. "A short history of fertilisers". Fertiliser Development and Consultation Organisation. Archived from the original on 23 January 2017. Retrieved 17 December 2017.
- Way, J. Thomas (1852). "On the power of soils to absorb manure". Journal of the Royal Agricultural Society of England. 13: 123–43. Retrieved 17 December 2017.
- Warington, Robert (1878). Note on the appearance of nitrous acid during the evaporation of water: a report of experiments made in the Rothamsted laboratory. London: Harrison and Sons.
- Winogradsky, Sergei (1890). "Sur les organismes de la nitrification" (PDF). Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences (in French). 110 (1): 1013–16. Retrieved 17 December 2017.
- Kellogg 1957, pp. 1–4.
- Hilgard, Eugene W. (1921). Soils: their formation, properties, composition, and relations to climate and plant growth in the humid and arid regions. London: The Macmillan Company. Retrieved 17 December 2017.
- Fallou, Friedrich Albert (1857). Anfangsgründe der Bodenkunde (PDF) (in German). Dresden: G. Schönfeld's Buchhandlung. Archived from the original (PDF) on 15 December 2018. Retrieved 15 December 2018.
- Glinka, Konstantin Dmitrievich (1914). Die Typen der Bodenbildung: ihre Klassifikation und geographische Verbreitung (in German). Berlin: Borntraeger.
- Glinka, Konstantin Dmitrievich (1927). The great soil groups of the world and their development. Ann Arbor, Michigan: Edwards Brothers.
Bibliography
- Donahue, Roy Luther; Miller, Raymond W.; Shickluna, John C. (1977). Soils: An Introduction to Soils and Plant Growth. Prentice-Hall. ISBN 978-0-13-821918-5.
- "Arizona Master Gardener". Cooperative Extension, College of Agriculture, University of Arizona. Retrieved 27 May 2013.
- Stefferud, Alfred, ed. (1957). Soil: The Yearbook of Agriculture 1957. United States Department of Agriculture. OCLC 704186906.
- Kellogg. "We Seek; We Learn". In Stefferud (1957).
- Simonson. "What Soils Are". In Stefferud (1957).
- Russell. "Physical Properties". In Stefferud (1957).
- Richards & Richards. "Soil Moisture". In Stefferud (1957).
- Wadleigh. "Growth of Plants". In Stefferud (1957).
- Allaway. "pH, Soil Acidity, and Plant Growth". In Stefferud (1957).
- Coleman & Mehlich. "The Chemistry of Soil pH". In Stefferud (1957).
- Dean. "Plant Nutrition and Soil Fertility". In Stefferud (1957).
- Allison. "Nitrogen and Soil Fertility". In Stefferud (1957).
- Olsen & Fried. "Soil Phosphorus and Fertility". In Stefferud (1957).
- Reitemeier. "Soil Potassium and Fertility". In Stefferud (1957).
- Jordan & Reisenauer. "Sulfur and Soil Fertility". In Stefferud (1957).
- Holmes & Brown. "Iron and Soil Fertility". In Stefferud (1957).
- Seatz & Jurinak. "Zinc and Soil Fertility". In Stefferud (1957).
- Russel. "Boron and Soil Fertility". In Stefferud (1957).
- Reuther. "Copper and Soil Fertility". In Stefferud (1957).
- Sherman. "Manganese and Soil Fertility". In Stefferud (1957).
- Stout & Johnson. "Trace Elements". In Stefferud (1957).
- Broadbent. "Organic Matter". In Stefferud (1957).
- Clark. "Living Organisms in the Soil". In Stefferud (1957).
- Flemming. "Soil Management and Insect Control". In Stefferud (1957).
Further reading
- Soil-Net.com A free schools-age educational site teaching about soil and its importance.
- Adams, J.A. 1986. Dirt. College Station, Texas: Texas A&M University Press ISBN 0-89096-301-0
- Certini, G., Scalenghe, R. 2006. Soils: Basic concepts and future challenges. Cambridge Univ Press, Cambridge.
- David R. Montgomery, Dirt: The Erosion of Civilizations, ISBN 978-0-520-25806-8
- Faulkner, Edward H. 1943. Plowman's Folly. New York, Grosset & Dunlap. ISBN 0-933280-51-3
- LandIS Free Soilscapes Viewer Free interactive viewer for the Soils of England and Wales
- Jenny, Hans. 1941. Factors of Soil Formation: A System of Quantitative Pedology
- Logan, W.B. 1995. Dirt: The ecstatic skin of the earth. ISBN 1-57322-004-3
- Mann, Charles C. September 2008. " Our good earth" National Geographic Magazine
- "97 Flood". USGS. Archived from the original on 24 June 2008. Retrieved 8 July 2008. Photographs of sand boils.
- Soil Survey Division Staff. 1999. Soil survey manual. Soil Conservation Service. U.S. Department of Agriculture Handbook 18.
- Soil Survey Staff. 1975. Soil Taxonomy: A basic system of soil classification for making and interpreting soil surveys. USDA-SCS Agric. Handb. 436. United States Government Printing Office, Washington, DC.
- Soils (Matching suitable forage species to soil type), Oregon State University
- Gardiner, Duane T. "Lecture 1 Chapter 1 Why Study Soils?". ENV320: Soil Science Lecture Notes. Texas A&M University-Kingsville. Archived from the original on 9 February 2018. Retrieved 7 January 2019.
- Janick, Jules. 2002. Soil notes, Purdue University
- LandIS Soils Data for England and Wales a pay source for GIS data on the soils of England and Wales and soils data source; they charge a handling fee to researchers.
External links
| Wikiversity has learning resources about Soil Formation |
| The Wikibook Historical Geology has a page on the topic of: Soils and paleosols |
- Short video explaining soil basics
- The Soil Water Compendium (soil water content sensors explained)
- Global Soil Partnership
- FAO Soils Portal
- World Reference Base for Soil Resources
- ISRIC – World Soil Information (ICSU World Data Centre for Soils)
- World Soil Library and Maps
- Wossac the world soil survey archive and catalogue
- Canadian Society of Soil Science
- Soil Science Society of America
- USDA-NRCS Web Soil Survey
- European Soil Portal (wiki)
- National Soil Resources Institute UK
- Plant and Soil Sciences eLibrary
- Copies of the reference 'Soil: The Yearbook of Agriculture 1957' in multiple formats
