Ethinylestradiol/segesterone acetate
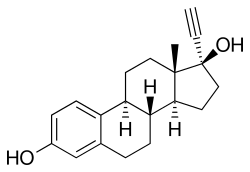
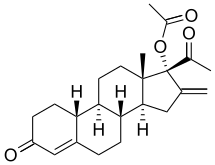
Ethinylestradiol/segesterone acetate (EE/SGA), sold under the brand name Annovera, is a contraceptive vaginal ring and combined form of hormonal birth control which contains ethinylestradiol, an estrogen, and segesterone acetate, a progestin.[1] It contains 17.4 mg ethinylestradiol and 103 mg segesterone acetate, releases an average of 13 μg ethinylestradiol and 0.15 mg segesterone acetate per day.[1][2]
 | |
 | |
| Combination of | |
|---|---|
| Ethinylestradiol | Estrogen |
| Segesterone acetate | Progestogen |
| Clinical data | |
| Trade names | Annovera |
| Other names | EE/SGA; EE/SA |
| Routes of administration | Vaginal ring |
| Identifiers | |
| KEGG | |
Annovera is inserted into the vagina and left for 21 days, then removed, washed and stored for 7 days, during which the user experiences a period (withdrawal bleeding.) This can be repeated 13 times, for one full year of use.[3] Unlike NuvaRing, another vaginal ring contraceptive, Annovera does not need to be refrigerated before being dispensed to a patient and can be stored at temperatures up to 30 degrees Celsius.[4]
The medication was developed by the Population Council, an international non-profit organization, and licensed to TherapeuticsMD. It was approved for use in the United States on 10 August 2018.[2]
See also
References
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/209627s000lbl.pdf
- https://ir.therapeuticsmd.com/news-releases/news-release-details/therapeuticsmd-announces-fda-approval-annoveratm-segesterone
- "FDA approves new vaginal ring for one year of birth control".
- "FDA prescribing information for Annovera" (PDF).
| Progestogens (and progestins) |
| ||||
|---|---|---|---|---|---|
| Antiprogestogens |
| ||||
| |||||
| ER |
| ||||||
|---|---|---|---|---|---|---|---|
| GPER |
| ||||||
| |||||||
| PR |
| ||||||
|---|---|---|---|---|---|---|---|
| mPR (PAQR) |
| ||||||
| |||||||