Testosterone enanthate
Testosterone enanthate, sold under the brand names Delatestryl and Xyosted among others, is an androgen and anabolic steroid (AAS) medication which is used mainly in the treatment of low testosterone levels in men.[2][3][4] It is also used in hormone therapy for transgender men.[5] It is given by injection into muscle or subcutaneously usually once every one to four weeks.[4][6][1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Delatestryl, Xyosted, others |
| Other names | TE; Testosterone heptanoate; Testosterone 17β-heptanoate; NSC-17591 |
| Routes of administration | Intramuscular injection, subcutaneous injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Oral: very low Intramuscular: high |
| Metabolism | Liver |
| Elimination half-life | Intramuscular: 4–5 days[1] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.005.686 |
| Chemical and physical data | |
| Formula | C26H40O3 |
| Molar mass | 400.603 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Side effects of testosterone enanthate include symptoms of masculinization like acne, increased hair growth, voice changes, and increased sexual desire.[4] The drug is a synthetic androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT).[7][4] It has strong androgenic effects and moderate anabolic effects, which make it useful for producing masculinization and suitable for androgen replacement therapy.[4] Testosterone enanthate is a testosterone ester and a long-lasting prodrug of testosterone in the body.[6][2][3] Because of this, it is considered to be a natural and bioidentical form of testosterone.[8]
Testosterone enanthate was introduced for medical use in 1954.[9][3] Along with testosterone cypionate, testosterone undecanoate, and testosterone propionate, it is one of the most widely used testosterone esters.[7][3][4] In addition to its medical use, testosterone enanthate is used to improve physique and performance.[4] The drug is a controlled substance in many countries and so non-medical use is generally illicit.[4]
Medical uses
Testosterone enanthate is used primarily in androgen replacement therapy.[3] It is the most widely used form of testosterone in androgen replacement therapy.[3] The medication is specifically approved, in the United States, for the treatment of hypogonadism in men, delayed puberty in boys, and breast cancer in women.[10] It is also used in masculinizing hormone therapy for transgender men.[5]
| Route | Medication | Major brand names | Form | Dosage |
|---|---|---|---|---|
| Oral | Testosteronea | – | Tablet | 400–800 mg/day (in divided doses) |
| Testosterone undecanoate | Andriol, Jatenzo | Capsule | 40–80 mg/2–4x day (with meals) | |
| Methyltestosteroneb | Android, Metandren, Testred | Tablet | 10–50 mg/day | |
| Fluoxymesteroneb | Halotestin, Ora-Testryl, Ultandren | Tablet | 5–20 mg/day | |
| Metandienoneb | Dianabol | Tablet | 5–15 mg/day | |
| Mesteroloneb | Proviron | Tablet | 25–150 mg/day | |
| Buccal | Testosterone | Striant | Tablet | 30 mg 2x/day |
| Methyltestosteroneb | Metandren, Oreton Methyl | Tablet | 5–25 mg/day | |
| Sublingual | Testosteroneb | Testoral | Tablet | 5–10 mg 1–4x/day |
| Methyltestosteroneb | Metandren, Oreton Methyl | Tablet | 10–30 mg/day | |
| Intranasal | Testosterone | Natesto | Nasal spray | 11 mg 3x/day |
| Transdermal | Testosterone | AndroGel, Testim, TestoGel | Gel | 25–125 mg/day |
| Androderm, AndroPatch, TestoPatch | Non-scrotal patch | 2.5–15 mg/day | ||
| Testoderm | Scrotal patch | 4–6 mg/day | ||
| Axiron | Axillary solution | 30–120 mg/day | ||
| Androstanolone (DHT) | Andractim | Gel | 100–250 mg/day | |
| Rectal | Testosterone | Rektandron, Testosteronb | Suppository | 40 mg 2–3x/day |
| Injection (IM or SC) | Testosterone | Andronaq, Sterotate, Virosterone | Aqueous suspension | 10–50 mg 2–3x/week |
| Testosterone propionateb | Testoviron | Oil solution | 10–50 mg 2–3x/week | |
| Testosterone enanthate | Delatestryl | Oil solution | 50–250 mg 1x/1–4 weeks | |
| Xyosted | Auto-injector | 50–100 mg 1x/week | ||
| Testosterone cypionate | Depo-Testosterone | Oil solution | 50–250 mg 1x/1–4 weeks | |
| Testosterone isobutyrate | Agovirin Depot | Aqueous suspension | 50–100 mg 1x/1–2 weeks | |
| Testosterone phenylacetateb | Perandren, Androject | Oil solution | 50–200 mg 1x/3–5 weeks | |
| Mixed testosterone esters | Sustanon 100, Sustanon 250 | Oil solution | 50–250 mg 1x/2–4 weeks | |
| Testosterone undecanoate | Aveed, Nebido | Oil solution | 750–1,000 mg 1x/10–14 weeks | |
| Testosterone buciclatea | – | Aqueous suspension | 600–1,000 mg 1x/12–20 weeks | |
| Implant | Testosterone | Testopel | Pellet | 150–1,200 mg/3–6 months |
| Notes: Men produce about 3 to 11 mg testosterone per day (mean 7 mg/day in young men). Footnotes: a = Never marketed. b = No longer used and/or no longer marketed. Sources: See template. | ||||
| Medication | Brand names | Type | Route | Dosage |
|---|---|---|---|---|
| Testosterone undecanoate | Andriol, Jatenzo | Androgen | Oral | 40–80 mg/2–3x day (with meals) |
| Testosterone | Striant | Androgen | Buccal | 30 mg 2x/day |
| Natesto | Nasal spray | 11 mg 3x/day | ||
| AndroGel, others | Transdermal gel | 25–100 mg/day | ||
| Androderm, others | Transdermal patch | 2.5–10 mg/day | ||
| Axiron | Axillary solution | 30–120 mg/day | ||
| Testopel | Subcutaneous implant | 150–600 mg/3–6 months | ||
| Testosterone enanthate | Delatestryl, others | Androgen | Injection (IM or SC) | 50–100 mg/week or 100–250 mg/2–4 weeks |
| Testosterone cypionate | Depo-Testosterone, others | Androgen | Injection (IM or SC) | 50–100 mg/week or 100–250 mg/2–4 weeks |
| Testosterone isobutyrate | Agovirin Depot | Androgen | Injection (IM or SC) | 50–100 mg/week |
| Mixed testosterone esters | Sustanon 250, others | Androgen | Injection (IM or SC) | 250 mg/2–3 weeks or 500 mg/3–6 weeks |
| Testosterone undecanoate | Aveed, Nebido, others | Androgen | Injection (IM or SC) | 750–1,000 mg/10–14 weeks |
| GnRH analogue | Various | GnRH modulator | Parenteral (various) | Variable |
| Elagolix | Orilissa | GnRH antagonist | Oral | 150 mg/day or 200 mg/twice a day |
| Medroxyprogesterone acetatea | Provera, others | Progestin | Oral | 5–10 mg/day |
| Depo-Provera, others | Injection (IM) | 150 mg/3 months | ||
| Depo-SubQ Provera 104 | Injection (SC) | 104 mg/3 months | ||
| Lynestrenola | Orgametril, others | Progestin | Oral | 5–10 mg/day |
| Finasterideb | Propecia, Proscar | 5α-Reductase inhibitor | Oral | 1 mg/day |
| Dutasterideb | Avodart | 5α-Reductase inhibitor | Oral | 0.5 mg/day |
| Notes: Testes produce 3 to 11 mg testosterone per day (mean 7 mg/day in young men). Footnotes: a = For suppression of menses. b = For prevention/treatment of scalp hair loss. Sources: See template. | ||||
| Route | Medication | Major brand names | Form | Dosage |
|---|---|---|---|---|
| Oral | Testosterone undecanoate | Andriol, Jatenzo | Capsule | 40–80 mg 1x/1–2 days |
| Methyltestosterone | Metandren, Estratest | Tablet | 0.5–10 mg/day | |
| Fluoxymesterone | Halotestin | Tablet | 1–2.5 mg 1x/1–2 days | |
| Normethandronea | Ginecoside | Tablet | 5 mg/day | |
| Tibolone | Livial | Tablet | 1.25–2.5 mg/day | |
| Prasterone (DHEA)b | – | Tablet | 10–100 mg/day | |
| Sublingual | Methyltestosterone | Metandren | Tablet | 0.25 mg/day |
| Transdermal | Testosterone | Intrinsa | Patch | 150–300 μg/day |
| AndroGel | Gel, cream | 1–10 mg/day | ||
| Vaginal | Prasterone (DHEA) | Intrarosa | Insert | 6.5 mg/day |
| Injection | Testosterone propionatea | Testoviron | Oil solution | 25 mg 1x/1–2 weeks |
| Testosterone enanthate | Delatestryl, Primodian Depot | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone cypionate | Depo-Testosterone, Depo-Testadiol | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone isobutyratea | Femandren M, Folivirin | Aqueous suspension | 25–50 mg 1x/4–6 weeks | |
| Mixed testosterone esters | Climacterona | Oil solution | 150 mg 1x/4–8 weeks | |
| Omnadren, Sustanon | Oil solution | 50–100 mg 1x/4–6 weeks | ||
| Nandrolone decanoate | Deca-Durabolin | Oil solution | 25–50 mg 1x/6–12 weeks | |
| Prasterone enanthatea | Gynodian Depot | Oil solution | 200 mg 1x/4–6 weeks | |
| Implant | Testosterone | Testopel | Pellet | 50–100 mg 1x/3–6 months |
| Notes: Premenopausal women produce about 230 ± 70 μg testosterone per day (6.4 ± 2.0 mg testosterone per 4 weeks), with a range of 130 to 330 μg per day (3.6–9.2 mg per 4 weeks). Footnotes: a = Mostly discontinued or unavailable. b = Over-the-counter. Sources: See template. | ||||
| Route | Medication | Form | Dosage | |
|---|---|---|---|---|
| Oral | Methyltestosterone | Tablet | 30–200 mg/day | |
| Fluoxymesterone | Tablet | 10–40 mg 3x/day | ||
| Calusterone | Tablet | 40–80 mg 4x/day | ||
| Normethandrone | Tablet | 40 mg/day | ||
| Buccal | Methyltestosterone | Tablet | 25–100 mg/day | |
| Injection (IM or SC) | Testosterone propionate | Oil solution | 50–100 mg 3x/week | |
| Testosterone enanthate | Oil solution | 200–400 mg 1x/2–4 weeks | ||
| Testosterone cypionate | Oil solution | 200–400 mg 1x/2–4 weeks | ||
| Mixed testosterone esters | Oil solution | 250 mg 1x/week | ||
| Methandriol | Aqueous suspension | 100 mg 3x/week | ||
| Androstanolone (DHT) | Aqueous suspension | 300 mg 3x/week | ||
| Drostanolone propionate | Oil solution | 100 mg 1–3x/week | ||
| Metenolone enanthate | Oil solution | 400 mg 3x/week | ||
| Nandrolone decanoate | Oil solution | 50–100 mg 1x/1–3 weeks | ||
| Nandrolone phenylpropionate | Oil solution | 50–100 mg/week | ||
| Note: Dosages are not necessarily equivalent. Sources: See template. | ||||
Side effects
Side effects of testosterone enanthate include virilization among others.[4] Approximately 10 percent of testosterone enanthate will be converted to dihydrotestosterone in normal men.[11] Dihydrotestosterone (DHT) can promote masculine characteristics in both males and females. These masculine characteristics include: clitoral hypertrophy, androgenic alopecia, growth of body hair and deepening of the vocal cords. Dihydrotestosterone also plays an important role in male sexual function and may also be a contributing factor of ischemic priapism in males as shown in a study conducted on the use of finasteride to treat ischemic priapism in males. Testosterone enanthate can also lead to an increase in igf-1 and igf-bp.[12][13] Testosterone enanthate can also be converted to estradiol by aromatase,[14] which may lead to gynecomastia in males. Aromatase inhibitors can help to prevent the estrogenic activity of testosterone enanthate in the body.[15]
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone (DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Testosterone enanthate is a prodrug of testosterone and is an androgen and anabolic–androgenic steroid (AAS). That is, it is an agonist of the androgen receptor (AR).
Pharmacokinetics
Testosterone enanthate has an elimination half-life of 4.5 days and a mean residence time of 8.5 days when used as a depot intramuscular injection.[1] It requires frequent administration of approximately once per week, and large fluctuations in testosterone levels result with it, with levels initially being elevated and supraphysiological.[1]
| Testosterone ester | Form | Route | Tmax | t1/2 | MRT |
|---|---|---|---|---|---|
| Testosterone undecanoate | Oil-filled capsules | Oral | ? | 1.6 hours | 3.7 hours |
| Testosterone propionate | Oil solution | Intramuscular injection | ? | 0.8 days | 1.5 days |
| Testosterone enanthate | Castor oil solution | Intramuscular injection | 10 days | 4.5 days | 8.5 days |
| Testosterone undecanoate | Tea seed oil solution | Intramuscular injection | 13.0 days | 20.9 days | 34.9 days |
| Testosterone undecanoate | Castor oil solution | Intramuscular injection | 11.4 days | 33.9 days | 36.0 days |
| Testosterone buciclatea | Aqueous suspension | Intramuscular injection | 25.8 days | 29.5 days | 60.0 days |
| Notes: Testosterone cypionate has similar pharmacokinetics to TE. Footnotes: a = Never marketed. Sources: See template. | |||||
| Medication | Form | Major brand names | Duration |
|---|---|---|---|
| Testosterone | Aqueous suspension | Andronaq, Sterotate, Virosterone | 2–3 days |
| Testosterone propionate | Oil solution | Androteston, Perandren, Testoviron | 3–4 days |
| Testosterone phenylpropionate | Oil solution | Testolent | 8 days |
| Testosterone isobutyrate | Aqueous suspension | Agovirin Depot, Perandren M | 14 days |
| Mixed testosterone estersa | Oil solution | Triolandren | 10–20 days |
| Mixed testosterone estersb | Oil solution | Testosid Depot | 14–20 days |
| Testosterone enanthate | Oil solution | Delatestryl | 14–28 days |
| Testosterone cypionate | Oil solution | Depovirin | 14–28 days |
| Mixed testosterone estersc | Oil solution | Sustanon 250 | 28 days |
| Testosterone undecanoate | Oil solution | Aveed, Nebido | 100 days |
| Testosterone buciclated | Aqueous suspension | 20 Aet-1, CDB-1781e | 90–120 days |
| Nandrolone phenylpropionate | Oil solution | Durabolin | 10 days |
| Nandrolone decanoate | Oil solution | Deca Durabolin | 21–28 days |
| Methandriol | Aqueous suspension | Notandron, Protandren | 8 days |
| Methandriol bisenanthoyl acetate | Oil solution | Notandron Depot | 16 days |
| Metenolone acetate | Oil solution | Primobolan | 3 days |
| Metenolone enanthate | Oil solution | Primobolan Depot | 14 days |
| Note: All are via i.m. injection. Footnotes: a = TP, TV, and TUe. b = TP and TKL. c = TP, TPP, TiCa, and TD. d = Studied but never marketed. e = Developmental code names. Sources: See template. | |||
Chemistry
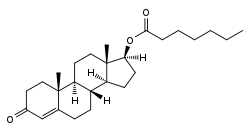
Testosterone enanthate, or testosterone 17β-heptanoate, is a synthetic androstane steroid and a derivative of testosterone.[16][17] It is an androgen ester; specifically, it is the C17β enanthate (heptanoate) ester of testosterone.[16][17]
| Androgen | Structure | Ester | Relative mol. weight | Relative T contentb | Durationc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position | Moiety | Type | Lengtha | Rank | Group | ||||
| Testosterone | – | – | – | – | 1.00 | 1.00 | 11 | Short | |
| Testosterone propionate | C17β | Propanoic acid | Straight-chain fatty acid | 3 | 1.19 | 0.84 | 10 | Short | |
| Testosterone isobutyrate | C17β | Isobutyric acid | Aromatic fatty acid | – (~3) | 1.24 | 0.80 | 9 | Moderate | |
| Testosterone cypionate | C17β | Cyclopentylpropanoic acid | Aromatic fatty acid | – (~6) | 1.43 | 0.70 | 8 | Moderate | |
| Testosterone phenylpropionate | C17β | Phenylpropanoic acid | Aromatic fatty acid | – (~6) | 1.46 | 0.69 | 7 | Moderate | |
| Testosterone isocaproate | C17β | Isohexanoic acid | Branched-chain fatty acid | – (~5) | 1.34 | 0.75 | 6 | Moderate | |
| Testosterone caproate | C17β | Hexanoic acid | Straight-chain fatty acid | 6 | 1.35 | 0.75 | 5 | Moderate | |
| Testosterone enanthate | C17β | Heptanoic acid | Straight-chain fatty acid | 7 | 1.39 | 0.72 | 4 | Moderate | |
| Testosterone decanoate | C17β | Decanoic acid | Straight-chain fatty acid | 10 | 1.53 | 0.65 | 3 | Long | |
| Testosterone undecanoate | C17β | Undecanoic acid | Straight-chain fatty acid | 11 | 1.58 | 0.63 | 2 | Long | |
| Testosterone buciclated | C17β | Bucyclic acide | Aromatic carboxylic acid | – (~9) | 1.58 | 0.63 | 1 | Long | |
| Footnotes: a = Length of ester in carbon atoms for straight-chain fatty acids or approximate length of ester in carbon atoms for aromatic fatty acids. b = Relative testosterone content by weight (i.e., relative androgenic/anabolic potency). c = Duration by intramuscular or subcutaneous injection in oil solution (except TiB and TB, which are in aqueous suspension). d = Never marketed. e = Bucyclic acid = trans-4-Butylcyclohexane-1-carboxylic acid. Sources: See individual articles. | |||||||||
History
Testosterone enanthate was described as early as 1952[18] and was first introduced for medical use in the United States in 1954 under the brand name Delatestryl.[9][3]
Society and culture
Generic names
Testosterone enanthate is the generic name of the drug and its USAN and BAN.[16][17][19][20] It has also referred to as testosterone heptanoate.[16][17][19][20]
Brand names
Testosterone enanthate is marketed primarily under the brand name Delatestryl.[16][17][19][20]
It is or has been marketed under a variety of other brand names as well, including, among others:[16][17][19][20]
- Andro LA
- Andropository
- Depandro
- Durathate
- Everone
- Testostroval
- Testrin
- Testro LA
- Xyosted
Availability
Testosterone enanthate is available in the United States and widely elsewhere throughout the world.[21][17][20] Testosterone enanthate (testosterone heptanoate) is often available in concentrations of 200mg per milliliter of fluid.[22]
Legal status
Testosterone enanthate, along with other AAS, is a schedule III controlled substance in the United States under the Controlled Substances Act and a schedule IV controlled substance in Canada under the Controlled Drugs and Substances Act.[23][24]
Research
As of October 2017, an auto-injection formulation of testosterone enanthate was in preregistration for the treatment of hypogonadism in the United States.[25]
Xyosted
On October 1, 2018, the U.S. Food and Drug Administration (FDA) announced the approval of Xyosted. Xyosted, a product of Antares Pharma, Inc., is a single-use disposable auto-injector that dispenses testosterone enanthate. Xyosted is the first FDA-approved subcutaneous testosterone enanthate product for testosterone replacement therapy in adult males.[26]
References
- Luetjens CM, Wistuba J, Weinbauer G, Nieschlag E (2007). "The Leydig Cell as a Target for Male Contraception". The Leydig Cell in Health and Disease. Contemporary Endocrinology. pp. 415–442. doi:10.1007/978-1-59745-453-7_29. ISBN 978-1-58829-754-9.
- Nieschlag E, Behre HM, Nieschlag S (26 July 2012). Testosterone: Action, Deficiency, Substitution. Cambridge University Press. pp. 315–. ISBN 978-1-107-01290-5.
- Nieschlag E, Behre HM, Nieschlag S (13 January 2010). Andrology: Male Reproductive Health and Dysfunction. Springer Science & Business Media. pp. 442–. ISBN 978-3-540-78355-8.
- Llewellyn W (2011). Anabolics. Molecular Nutrition Llc. pp. 208–211. ISBN 978-0-9828280-1-4.
- Irwig MS (April 2017). "Testosterone therapy for transgender men". The Lancet. Diabetes & Endocrinology. 5 (4): 301–311. doi:10.1016/S2213-8587(16)00036-X. PMID 27084565.
- Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1185, 1187. ISBN 978-0-7817-1750-2.
- Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- Santoro N, Braunstein GD, Butts CL, Martin KA, McDermott M, Pinkerton JV (April 2016). "Compounded Bioidentical Hormones in Endocrinology Practice: An Endocrine Society Scientific Statement". The Journal of Clinical Endocrinology and Metabolism. 101 (4): 1318–43. doi:10.1210/jc.2016-1271. PMID 27032319.
- "Testosterone Enanthate". p. 35t. in William Andrew Publishing (2007). "T". Pharmaceutical Manufacturing Encyclopedia. pp. 1t–242t. doi:10.1016/B978-0-8155-1526-5.50024-6. ISBN 978-0-8155-1526-5.
- "DELATESTRYL Package Insert" (PDF). Indevus Pharmaceuticals, Inc.
- "DHT (dihydrotestosterone): What is DHT's role in baldness?".
- Ashton WS, Degnan BM, Daniel A, Francis GL (1995). "Testosterone increases insulin-like growth factor-1 and insulin-like growth factor-binding protein". Annals of Clinical and Laboratory Science. 25 (5): 381–8. PMID 7486812.
- Hoeh MP, Levine LA (March 2015). "Management of Recurrent Ischemic Priapism 2014: A Complex Condition with Devastating Consequences". Sexual Medicine Reviews. 3 (1): 24–35. doi:10.1002/smrj.37. PMID 27784569.
- Ishikawa T, Glidewell-Kenney C, Jameson JL (February 2006). "Aromatase-independent testosterone conversion into estrogenic steroids is inhibited by a 5 alpha-reductase inhibitor". The Journal of Steroid Biochemistry and Molecular Biology. 98 (2–3): 133–8. doi:10.1016/j.jsbmb.2005.09.004. PMID 16386416.
- Ishikawa T, Glidewell-Kenney C, Jameson JL (February 2006). "Aromatase-independent testosterone conversion into estrogenic steroids is inhibited by a 5 alpha-reductase inhibitor". The Journal of Steroid Biochemistry and Molecular Biology. 98 (2–3): 133–8. doi:10.1016/j.jsbmb.2005.09.004. PMID 16386416.
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 1002–1004. ISBN 978-3-88763-075-1.
- Junkmann K (1952). "Über protrahiert wirksame Androgene" [Over protracted effective androgens]. Festschrift zum 75. Geburtstag. pp. 85–92. doi:10.1007/978-3-642-49902-9_11. ISBN 978-3-642-49610-3.
- Morton IK, Hall JM (6 December 2012). "Testosterone". Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. p. 270. ISBN 978-94-011-4439-1.
- "Testosterone". Drugs.com. October 1, 2018. Retrieved December 5, 2018.
- "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 17 December 2016.
- "Testosterone enanthate".
- Karch SB (21 December 2006). Drug Abuse Handbook, Second Edition. CRC Press. pp. 30–. ISBN 978-1-4200-0346-8.
- Lilley LL, Snyder JS, Collins SR (5 August 2016). Pharmacology for Canadian Health Care Practice. Elsevier Health Sciences. pp. 50–. ISBN 978-1-77172-066-3.
- "Testosterone enanthate auto-injection - Antares Pharma". AdisInsight. February 5, 2018. Retrieved December 5, 2018.
- Antares Receives FDA Approval of Xyosted (Testosterone Enanthate) Injection for Testosterone Replacement Therapy in Adult Males