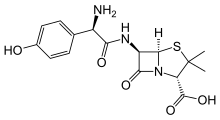
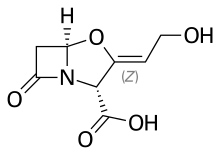
Amoxicillin/clavulanic acid
Amoxicillin/clavulanic acid, also known as co-amoxiclav, is an antibiotic useful for the treatment of a number of bacterial infections.[3] It is a combination consisting of amoxicillin, a β-lactam antibiotic, and potassium clavulanate, a β-lactamase inhibitor.[3] It is specifically used for otitis media, strep throat, pneumonia, cellulitis, urinary tract infections, and animal bites.[3] It is taken by mouth or by injection into a vein.[2]
 | |
 | |
| Combination of | |
|---|---|
| Amoxicillin | Penicillin antibiotic |
| Clavulanic acid | Beta-lactamase inhibitor |
| Clinical data | |
| Trade names | Augmentin, Clavulin, other[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685024 |
| License data |
|
| Pregnancy category | |
| Routes of administration | By mouth, intravenous[2] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H27KN4O10S |
| Molar mass | 602.66 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include diarrhea, vomiting, and allergic reactions.[3] It also increases the risk of yeast infections, headaches, and blood clotting problems.[2][4] It is not recommended in people with a history of a penicillin allergy.[2] It is relatively safe for use during pregnancy.[3]
Amoxicillin/clavulanic acid was approved for medical use in the United States in 1984.[3] It is on the World Health Organization's List of Essential Medicines.[5] It is available as a generic medication.[3] In 2017, it was the 116th most commonly prescribed medication in the United States, with more than six million prescriptions.[6][7]
Medical uses
Amoxicillin/clavulanic acid is widely used to treat or prevent many infections caused by susceptible bacteria, such as:
- urinary tract infections
- respiratory tract infections
- skin and soft tissue infections
- sinus infections
- tonsillitis
- cat scratches
- infections caused by the bacterial flora of the mouth, such as:
It is also used for tuberculosis that is resistant to other treatments.[3]
This combination results in an antibiotic with an increased spectrum of action and restored efficacy against amoxicillin-resistant bacteria that produce β-lactamase.
Adverse effects
Possible side effects include diarrhea, vomiting, nausea, thrush, and skin rash. These do not usually require medical attention. As with all antimicrobial agents, antibiotic-associated diarrhea due to Clostridium difficile infection—sometimes leading to pseudomembranous colitis—may occur during or after treatment with amoxicillin/clavulanic acid.[9]
Rarely, cholestatic jaundice (also referred to as cholestatic hepatitis, a form of liver toxicity) has been associated with amoxicillin/clavulanic acid. The reaction may occur up to several weeks after treatment has stopped, and usually takes weeks to resolve. It is more frequent in men, older people, and those who have taken long courses of treatment; the estimated overall incidence is one in 100,000 exposures.[9] In the United Kingdom, co-amoxiclav carries a warning from the Committee on Safety of Medicines to this effect.[8]
As all aminopenicillins, amoxicillin has been associated with Stevens–Johnson syndrome/toxic epidermal necrolysis, although these reactions are very rare.[9][10]
History
British scientists working at Beecham (now part of GlaxoSmithKline), filed for US patent protection for the drug combination in 1979. They marketed it under the trade name Augmentin.[8] A patent was granted in 1985.[11]
Preparations
Amoxicillin/clavulanic acid is the International Nonproprietary Name (INN) and co-amoxiclav is the British Approved Name (BAN).
Many branded products indicate their strengths as the quantity of amoxicillin. Augmentin 250, for example, contains 250 mg of amoxicillin and 125 mg of clavulanic acid.[8][12]
An intravenous preparation has been available in the UK since 1985,[13] but no parenteral preparation is available in the US; the nearest equivalent is ampicillin/sulbactam.
Suspensions of amoxicillin/clavulanic acid are available for use in children. They must be refrigerated to maintain effectiveness.
Veterinary use
Amoxicillin/clavulanic acid is used in numerous animals for a variety of conditions:
- Dogs: periodontitis, kennel cough[14][15]
- Cats: urinary tract infections, skin and soft tissue infections
- Calves: enteritis, navel ill
- Cattle: respiratory tract infections, soft tissue infections, metritis, mastitis
- Pigs: respiratory tract infections, colibacillosis, mastitis, metritis, agalactia
In combination with prednisolone, it is used for intramammary infusion for the treatment of mastitis in lactating cows. Trade names include Clavaseptin, Clavamox, and Synulox.
Amoxicillin/clavulanic acid is banned from use in domestic-food animals (cattle, swine, etc.) in both the US and Europe; in the UK, Synulox can be used in domestic-food animals as long as a specified withdrawal period is observed.
Bacterial resistance
Bacterial antibiotic resistance is a growing problem in veterinary medicine. Amoxicillin/clavulanic acid is reported to be effective against clinical Klebsiella infections, but is not efficacious against Pseudomonas infections.[16]
References
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 97. ISBN 9781284057560.
- World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. p. 102. hdl:10665/44053. ISBN 9789241547659.
- "Amoxicillin and Clavulanate Potassium". The American Society of Health-System Pharmacists. Archived from the original on 29 November 2016. Retrieved 8 December 2016.
- Gillies, M; Ranakusuma, A; Hoffmann, T; Thorning, S; McGuire, T; Glasziou, P; Del Mar, C (17 November 2014). "Common harms from amoxicillin: a systematic review and meta-analysis of randomized placebo-controlled trials for any indication". Canadian Medical Association Journal. 187: E21-31. doi:10.1503/cmaj.140848. PMC 4284189. PMID 25404399.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- "Amoxicillin; Clavulanate Potassium - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
- British National Formulary (57 ed.). March 2009.
- Gordon D (2010). "Amoxicillin–Clavulanic Acid (Co-Amoxiclav)". In Grayson ML; et al. (eds.). Kucers' the Use of Antibiotics: a Clinical Review of Antibacterial, Antifungal, Antiparasitic and Antiviral Drugs. London: Hodder Arnold/ASM Press. pp. 193–4. ISBN 0-340-92767-4.
- Harr T, French LE (2010). "Toxic epidermal necrolysis and Stevens-Johnson syndrome". Orphanet Journal of Rare Diseases. 5: 39. doi:10.1186/1750-1172-5-39. PMC 3018455. PMID 21162721.
- US 4441609
- "Augmentin -- Prescribing Information" (PDF). December 2006. Archived (PDF) from the original on 20 December 2013.
- Davies BE, Boon R, Horton R, Reubi FC, Descoeudres CE (October 1988). "Pharmacokinetics of amoxycillin and clavulanic acid in haemodialysis patients following intravenous administration of Augmentin". British Journal of Clinical Pharmacology. 26 (4): 385–90. doi:10.1111/j.1365-2125.1988.tb03395.x. PMC 1386558. PMID 3190988.
- "Canine Infectious Tracheobronchitis (Kennel Cough)". Archived from the original on 21 May 2006. Retrieved 30 May 2013.
- "Kennel Cough - Symptoms and Treatment". Archived from the original on 10 May 2013. Retrieved 30 May 2013.
- Federation of Veterinarians in Europe Position Paper: "Antibiotic Resistance & Prudent Use of Antibiotics in Veterinary Medicine"
External links
- "Amoxicillin / clavulanic acid". Drug Information Portal. U.S. National Library of Medicine.