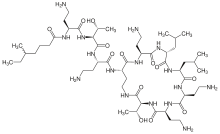
Polymyxin
Polymyxins are antibiotics. Polymyxins B and E (also known as colistin) are used in the treatment of Gram-negative bacterial infections. They work mostly by breaking up the bacterial cell membrane. They are part of a broader class of molecules called nonribosomal peptides.
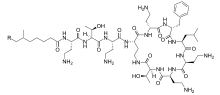
They are produced in nature by Gram-positive bacteria such as Paenibacillus polymyxa.
Medical use
Polymyxin antibiotics are relatively neurotoxic and nephrotoxic, so are usually used only as a last resort if modern antibiotics are ineffective or are contraindicated. Typical uses are for infections caused by strains of multiple drug-resistant Pseudomonas aeruginosa or carbapenemase-producing Enterobacteriaceae.[1][2] Polymyxins have less effect on Gram-positive organisms, and are sometimes combined with other agents (as with trimethoprim/polymyxin) to broaden the effective spectrum.[3]
Polymyxins B are not absorbed from the gastrointestinal tract, so they are only administered orally if the goal is to disinfect the GI tract.[3] Another route of administration is chosen for systemic treatment, e.g., parenteral (often intravenously) or by inhalation.[1][3] They are also used externally as a cream or drops to treat otitis externa (swimmers ear), and as a component of triple antibiotic ointment to treat and prevent skin infections.[3][4]
Mechanism of action
After binding to lipopolysaccharide (LPS) in the outer membrane of Gram-negative bacteria, polymyxins disrupt both the outer and inner membranes. The hydrophobic tail is important in causing membrane damage, suggesting a detergent-like mode of action.[1]
Removal of the hydrophobic tail of polymyxin B yields polymyxin nonapeptide, which still binds to LPS, but no longer kills the bacterial cell. However, it still detectably increases the permeability of the bacterial cell wall to other antibiotics, indicating that it still causes some degree of membrane disorganization.[5]
Gram-negative bacteria can develop resistance to polymyxins through various modifications of the LPS structure that inhibit the binding of polymyxins to LPS.[6]
Antibiotic resistance to this drug has been increasing, especially in southern China. Recently the gene mcr-1, which confers the antibiotic resistance, has been isolated from bacterial plasmids in Enterobacteriaceae.[7][8]
Chemistry

Polymyxins are a group of cyclic non-ribosomal polypeptide (NRPs) which are biosynthesized by bacteria belonging to the genus Bacillus, more specifically the subgenus Paenibacillus. Polymyxins consist of 10 amino acid residues, six of which are L-α,γ-diaminobutyric acid (L-DAB). The DAB residues cause polymyxins to have multiple positively charged groups at physiological pH. Seven amino acid residues form the main cyclic component, while the other three extend from one of the cyclic residues as a linear chain terminating in either 6-methyloctanoic acid or 6-methylheptanoic acid at the N-terminus. During cyclization, residue 10 is bound to the bridging residue 4.[9] The amino acid residues and DAB monomers are generally in the L (levo) configuration, however certain strains such as P. polymyxa PKB1 have been observed to incorporate DAB with the D (dextro) configuration at position 3 producing variations of polymyxin B.[10]
Polymyxin M is also known as "mattacin".[11]
Biosynthesis
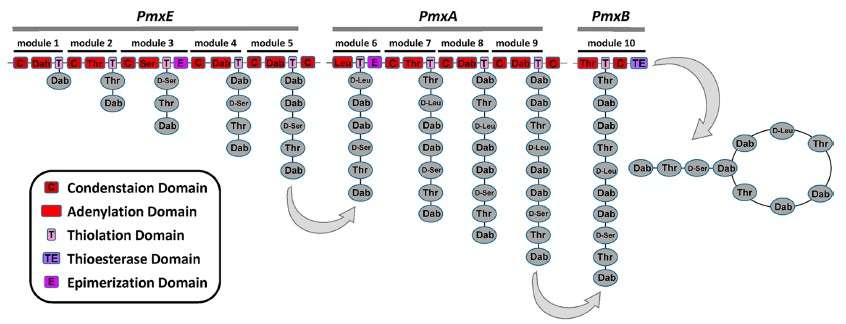
The polymyxins are produced by nonribosomal peptide synthetase systems in Gram-positive bacteria such as Paenibacillus polymyxa. Like other NRPs, polymyxins are assembled by synthetases with multiple modules, each containing a set of enzyme domains that sequentially operate on the growing chain by adding the next residue and extending the chain through peptide-bond formation and condensation reactions. The final steps involve a thioesterase domain at the C-terminal of the last module to cyclize the molecule and liberate the chain from the enzyme.[12]
Research
Polymyxins are used to neutralize or absorb LPS contaminants in samples, for example in immunological experiments. Minimization of LPS contamination can be important because LPS can evoke strong reactions from immune cells, distorting experimental results.
By increasing permeability of the bacterial membrane system, polymyxin is also used in clinical work to increase the release of secreted toxins, such as Shiga toxin, from Escherichia coli.[13]
The global problem of advancing antimicrobial resistance has led to a renewed interest in their use.[14]
See also
References
- Velkov, Tony; Roberts, Kade D; Nation, Roger L; Thompson, Philip E; Li, Jian (2013-06-01). "Pharmacology of polymyxins: new insights into an 'old' class of antibiotics". Future Microbiology. 8 (6): 711–724. doi:10.2217/fmb.13.39. ISSN 1746-0913. PMC 3852176. PMID 23701329.
- Falagas ME, Kasiakou SK (February 2006). "Toxicity of polymyxins: a systematic review of the evidence from old and recent studies". Crit Care. 10 (1): R27. doi:10.1186/cc3995. PMC 1550802. PMID 16507149.
- Poirel, L; Jayol, A; Nordmann, P (April 2017). "Polymyxins: Antibacterial Activity, Susceptibility Testing, and Resistance Mechanisms Encoded by Plasmids or Chromosomes". Clinical Microbiology Reviews. 30 (2): 557–596. doi:10.1128/CMR.00064-16. PMC 5355641. PMID 28275006.
- Ogbru, Omudhome. "Neomycin sulfate (Cortisporin): Drug Side Effects and Dosing". MedicineNet. Retrieved 11 June 2017.
- Tsubery, H.; Ofek, I.; Cohen, S.; Fridkin, M. (2000-01-01). "Structure activity relationship study of polymyxin B nonapeptide". Advances in Experimental Medicine and Biology. 479: 219–222. doi:10.1007/0-306-46831-X_18. ISBN 978-0-306-46409-6. ISSN 0065-2598. PMID 10897422.
- Tran AX, Lester ME, Stead CM, et al. (August 2005). "Resistance to the antimicrobial peptide polymyxin requires myristoylation of Escherichia coli and Salmonella typhimurium lipid A". J. Biol. Chem. 280 (31): 28186–28194. doi:10.1074/jbc.M505020200. PMID 15951433.
- Wolf, Joshua (2015). "Antibiotic resistance threatens the efficacy of prophylaxis". The Lancet Infectious Diseases. 15 (12): 1368–1369. doi:10.1016/S1473-3099(15)00317-5. PMID 26482598.
- Liu YY; et al. (18 November 2015). "Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study". The Lancet. 16 (2): 161–8. doi:10.1016/S1473-3099(15)00424-7. PMID 26603172.
- Dewick, Paul M. (2002-01-03). Medicinal Natural Products: A Biosynthetic Approach. John Wiley & Sons. ISBN 9780471496410.
- Shaheen, M.; Li, J.; Ross, A. C.; Vederas, J. C.; Jensen, S. E. Chem. Biol. 2011, 18, 1640−1648.
- Martin NI, Hu H, Moake MM, et al. (April 2003). "Isolation, structural characterization, and properties of mattacin (polymyxin M), a cyclic peptide antibiotic produced by Paenibacillus kobensis M". J. Biol. Chem. 278 (15): 13124–13132. doi:10.1074/jbc.M212364200. PMID 12569104.
- Kopp F, Marahiel MA (2007). "Macrocyclization strategies in polyketide and nonribosomal peptide biosynthesis". Nat Prod Rep. 24 (4): 735–49. doi:10.1039/b613652b. PMID 17653357.
- Yokoyama, K (2000). "Production of Shiga toxin by Escherichia coli measured with reference to the membrane vesicle-associated toxins". FEMS Microbiology Letters. 192 (1): 139–144. doi:10.1111/j.1574-6968.2000.tb09372.x. ISSN 0378-1097. PMID 11040442.
- Falagas, ME; Grammatikos, AP; Michalopoulos, A (2008). "Potential of old-generation antibiotics to address current need for new antibiotics". Expert Rev Anti Infect Ther. 6 (5): 593–600. doi:10.1586/14787210.6.5.593. PMID 18847400.