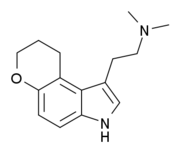
1-(2-Dimethylaminoethyl)dihydropyrano(3,2-e)indole
1-(2-Dimethylaminoethyl)dihydropyrano(3,2-e)indole (4,5-DHP-DMT) is a tricyclic tryptamine derivative which acts as a potent and reasonably selective partial agonist for the serotonin receptor 5-HT2A, with a Ki of 17.0 nM, and moderate selectivity over related serotonin receptors. It has lower 5-HT2 affinity and efficacy than the related compound AL-37350A, but higher lipophilicity.[1][2]
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C15H20N2O |
| Molar mass | 244.338 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
See also
References
- Macor JE, Fox CB, Johnson C, Koe BK, Lebel LA, Zorn SH (October 1992). "1-(2-Aminoethyl)-3-methyl-8,9-dihydropyrano[3,2-e]indole: a rotationally restricted phenolic analog of the neurotransmitter serotonin and agonist selective for serotonin (5-HT2-type) receptors". Journal of Medicinal Chemistry. 35 (20): 3625–32. doi:10.1021/jm00098a005. PMID 1433172.
- May JA, Chen HH, Rusinko A, Lynch VM, Sharif NA, McLaughlin MA (September 2003). "A novel and selective 5-HT2 receptor agonist with ocular hypotensive activity: (S)-(+)-1-(2-aminopropyl)-8,9-dihydropyrano[3,2-e]indole". Journal of Medicinal Chemistry. 46 (19): 4188–95. CiteSeerX 10.1.1.688.6169. doi:10.1021/jm030205t. PMID 12954071.
|
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.