Phytoestrogen
A phytoestrogen is a plant-derived xenoestrogen (see estrogen) not generated within the endocrine system, but consumed by eating phytoestrogenic plants. Also called a "dietary estrogen", it is a diverse group of naturally occurring nonsteroidal plant compounds that, because of its structural similarity with estradiol (17-β-estradiol), have the ability to cause estrogenic and/or antiestrogenic effects.[1] Phytoestrogens are not essential nutrients because their absence from the diet does not cause a disease, nor are they known to participate in any normal biological function.[1]
Its name comes from the Greek phyto ("plant") and estrogen, the hormone which gives fertility to female mammals. The word "estrus" - Greek οίστρος - means "sexual desire", and "gene" - Greek γόνο - is "to generate". It has been hypothesized that plants use a phytoestrogen as part of their natural defence against the overpopulation of herbivore animals by controlling female fertility.[2][3]
The similarities, at molecular level, of an estrogen and a phytoestrogen allow them to mildly mimic and sometimes act as an antagonist of estrogen.[1] Phytoestrogens were first observed in 1926,[1][4] but it was unknown if they could have any effect in human or animal metabolism. In the 1940s and early 1950s, it was noticed that some pastures of subterranean clover and red clover (phytoestrogen-rich plants) had adverse effects on the fecundity of grazing sheep.[1][5][6][7]
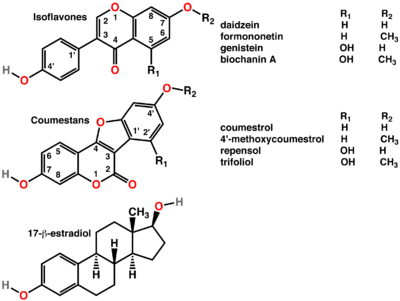
Structure
Phytoestrogens mainly belong to a large group of substituted natural phenolic compounds: the coumestans, prenylflavonoids and isoflavones are three of the most active in estrogenic effects in this class. The best-researched are isoflavones, which are commonly found in soy and red clover. Lignans have also been identified as phytoestrogens, although they are not flavonoids.[1] Mycoestrogens have similar structures and effects, but are not components of plants; these are mold metabolites of Fusarium, especially common on cereal grains,[8][9][10] but also occurring elsewhere, e.g. on various forages.[11] Although mycoestrogens are rarely taken into account in discussions about phytoestrogens, these are the compounds that initially generated the interest on the topic.[12]
Mechanism of action
Phytoestrogens exert their effects primarily through binding to estrogen receptors (ER).[13] There are two variants of the estrogen receptor, alpha (ER-α) and beta (ER-β) and many phytoestrogens display somewhat higher affinity for ER-β compared to ER-α.[13]
The key structural elements that enable phytoestrogens to bind with high affinity to estrogen receptors and display estradiol-like effects are:[1]
- The phenolic ring that is indispensable for binding to estrogen receptor
- The ring of isoflavones mimicking a ring of estrogens at the receptors binding site
- Low molecular weight similar to estrogens (MW=272)
- Distance between two hydroxyl groups at the isoflavones nucleus similar to that occurring in estradiol
- Optimal hydroxylation pattern
In addition to interaction with ERs, phytoestrogens may also modulate the concentration of endogenous estrogens by binding or inactivating some enzymes, and may affect the bioavailability of sex hormones by depressing or stimulating the synthesis of sex hormone-binding globulin (SHBG).[7]
Emerging evidence shows that some phytoestrogens bind to and transactivate peroxisome proliferator-activated receptors (PPARs).[14][15] In vitro studies show an activation of PPARs at concentrations above 1 μM, which is higher than the activation level of ERs.[16][17] At the concentration below 1 μM, activation of ERs may play a dominant role. At higher concentrations (>1 μM), both ERs and PPARs are activated. Studies have shown that both ERs and PPARs influence each other and therefore induce differential effects in a dose-dependent way. The final biological effects of genistein are determined by the balance among these pleiotrophic actions.[14][15][16]
| Ligand | Other names | Relative binding affinities (RBA, %)a | Absolute binding affinities (Ki, nM)a | Action | ||
|---|---|---|---|---|---|---|
| ERα | ERβ | ERα | ERβ | |||
| Estradiol | E2; 17β-Estradiol | 100 | 100 | 0.115 (0.04–0.24) | 0.15 (0.10–2.08) | Estrogen |
| Estrone | E1; 17-Ketoestradiol | 16.39 (0.7–60) | 6.5 (1.36–52) | 0.445 (0.3–1.01) | 1.75 (0.35–9.24) | Estrogen |
| Estriol | E3; 16α-OH-17β-E2 | 12.65 (4.03–56) | 26 (14.0–44.6) | 0.45 (0.35–1.4) | 0.7 (0.63–0.7) | Estrogen |
| Estetrol | E4; 15α,16α-Di-OH-17β-E2 | 4.0 | 3.0 | 4.9 | 19 | Estrogen |
| Alfatradiol | 17α-Estradiol | 20.5 (7–80.1) | 8.195 (2–42) | 0.2–0.52 | 0.43–1.2 | Metabolite |
| 16-Epiestriol | 16β-Hydroxy-17β-estradiol | 7.795 (4.94–63) | 50 | ? | ? | Metabolite |
| 17-Epiestriol | 16α-Hydroxy-17α-estradiol | 55.45 (29–103) | 79–80 | ? | ? | Metabolite |
| 16,17-Epiestriol | 16β-Hydroxy-17α-estradiol | 1.0 | 13 | ? | ? | Metabolite |
| 2-Hydroxyestradiol | 2-OH-E2 | 22 (7–81) | 11–35 | 2.5 | 1.3 | Metabolite |
| 2-Methoxyestradiol | 2-MeO-E2 | 0.0027–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Hydroxyestradiol | 4-OH-E2 | 13 (8–70) | 7–56 | 1.0 | 1.9 | Metabolite |
| 4-Methoxyestradiol | 4-MeO-E2 | 2.0 | 1.0 | ? | ? | Metabolite |
| 2-Hydroxyestrone | 2-OH-E1 | 2.0–4.0 | 0.2–0.4 | ? | ? | Metabolite |
| 2-Methoxyestrone | 2-MeO-E1 | <0.001–<1 | <1 | ? | ? | Metabolite |
| 4-Hydroxyestrone | 4-OH-E1 | 1.0–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestrone | 4-MeO-E1 | <1 | <1 | ? | ? | Metabolite |
| 16α-Hydroxyestrone | 16α-OH-E1; 17-Ketoestriol | 2.0–6.5 | 35 | ? | ? | Metabolite |
| 2-Hydroxyestriol | 2-OH-E3 | 2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestriol | 4-MeO-E3 | 1.0 | 1.0 | ? | ? | Metabolite |
| Estradiol sulfate | E2S; Estradiol 3-sulfate | <1 | <1 | ? | ? | Metabolite |
| Estradiol disulfate | Estradiol 3,17β-disulfate | 0.0004 | ? | ? | ? | Metabolite |
| Estradiol 3-glucuronide | E2-3G | 0.0079 | ? | ? | ? | Metabolite |
| Estradiol 17β-glucuronide | E2-17G | 0.0015 | ? | ? | ? | Metabolite |
| Estradiol 3-gluc. 17β-sulfate | E2-3G-17S | 0.0001 | ? | ? | ? | Metabolite |
| Estrone sulfate | E1S; Estrone 3-sulfate | <1 | <1 | >10 | >10 | Metabolite |
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | ? | ? | ? | Estrogen |
| Estradiol 17β-benzoate | E2-17B | 11.3 | 32.6 | ? | ? | Estrogen |
| Estrone methyl ether | Estrone 3-methyl ether | 0.145 | ? | ? | ? | Estrogen |
| ent-Estradiol | 1-Estradiol | 1.31–12.34 | 9.44–80.07 | ? | ? | Estrogen |
| Equilin | 7-Dehydroestrone | 13 (4.0–28.9) | 13.0–49 | 0.79 | 0.36 | Estrogen |
| Equilenin | 6,8-Didehydroestrone | 2.0–15 | 7.0–20 | 0.64 | 0.62 | Estrogen |
| 17β-Dihydroequilin | 7-Dehydro-17β-estradiol | 7.9–113 | 7.9–108 | 0.09 | 0.17 | Estrogen |
| 17α-Dihydroequilin | 7-Dehydro-17α-estradiol | 18.6 (18–41) | 14–32 | 0.24 | 0.57 | Estrogen |
| 17β-Dihydroequilenin | 6,8-Didehydro-17β-estradiol | 35–68 | 90–100 | 0.15 | 0.20 | Estrogen |
| 17α-Dihydroequilenin | 6,8-Didehydro-17α-estradiol | 20 | 49 | 0.50 | 0.37 | Estrogen |
| Δ8-Estradiol | 8,9-Dehydro-17β-estradiol | 68 | 72 | 0.15 | 0.25 | Estrogen |
| Δ8-Estrone | 8,9-Dehydroestrone | 19 | 32 | 0.52 | 0.57 | Estrogen |
| Ethinylestradiol | EE; 17α-Ethynyl-17β-E2 | 120.9 (68.8–480) | 44.4 (2.0–144) | 0.02–0.05 | 0.29–0.81 | Estrogen |
| Mestranol | EE 3-methyl ether | ? | 2.5 | ? | ? | Estrogen |
| Moxestrol | RU-2858; 11β-Methoxy-EE | 35–43 | 5–20 | 0.5 | 2.6 | Estrogen |
| Methylestradiol | 17α-Methyl-17β-estradiol | 70 | 44 | ? | ? | Estrogen |
| Diethylstilbestrol | DES; Stilbestrol | 129.5 (89.1–468) | 219.63 (61.2–295) | 0.04 | 0.05 | Estrogen |
| Hexestrol | Dihydrodiethylstilbestrol | 153.6 (31–302) | 60–234 | 0.06 | 0.06 | Estrogen |
| Dienestrol | Dehydrostilbestrol | 37 (20.4–223) | 56–404 | 0.05 | 0.03 | Estrogen |
| Benzestrol (B2) | – | 114 | ? | ? | ? | Estrogen |
| Chlorotrianisene | TACE | 1.74 | ? | 15.30 | ? | Estrogen |
| Triphenylethylene | TPE | 0.074 | ? | ? | ? | Estrogen |
| Triphenylbromoethylene | TPBE | 2.69 | ? | ? | ? | Estrogen |
| Tamoxifen | ICI-46,474 | 3 (0.1–47) | 3.33 (0.28–6) | 3.4–9.69 | 2.5 | SERM |
| Afimoxifene | 4-Hydroxytamoxifen; 4-OHT | 100.1 (1.7–257) | 10 (0.98–339) | 2.3 (0.1–3.61) | 0.04–4.8 | SERM |
| Toremifene | 4-Chlorotamoxifen; 4-CT | ? | ? | 7.14–20.3 | 15.4 | SERM |
| Clomifene | MRL-41 | 25 (19.2–37.2) | 12 | 0.9 | 1.2 | SERM |
| Cyclofenil | F-6066; Sexovid | 151–152 | 243 | ? | ? | SERM |
| Nafoxidine | U-11,000A | 30.9–44 | 16 | 0.3 | 0.8 | SERM |
| Raloxifene | – | 41.2 (7.8–69) | 5.34 (0.54–16) | 0.188–0.52 | 20.2 | SERM |
| Arzoxifene | LY-353,381 | ? | ? | 0.179 | ? | SERM |
| Lasofoxifene | CP-336,156 | 10.2–166 | 19.0 | 0.229 | ? | SERM |
| Ormeloxifene | Centchroman | ? | ? | 0.313 | ? | SERM |
| Levormeloxifene | 6720-CDRI; NNC-460,020 | 1.55 | 1.88 | ? | ? | SERM |
| Ospemifene | Deaminohydroxytoremifene | 2.63 | 1.22 | ? | ? | SERM |
| Bazedoxifene | – | ? | ? | 0.053 | ? | SERM |
| Etacstil | GW-5638 | 4.30 | 11.5 | ? | ? | SERM |
| ICI-164,384 | – | 63.5 (3.70–97.7) | 166 | 0.2 | 0.08 | Antiestrogen |
| Fulvestrant | ICI-182,780 | 43.5 (9.4–325) | 21.65 (2.05–40.5) | 0.42 | 1.3 | Antiestrogen |
| Propylpyrazoletriol | PPT | 49 (10.0–89.1) | 0.12 | 0.40 | 92.8 | ERα agonist |
| 16α-LE2 | 16α-Lactone-17β-estradiol | 14.6–57 | 0.089 | 0.27 | 131 | ERα agonist |
| 16α-Iodo-E2 | 16α-Iodo-17β-estradiol | 30.2 | 2.30 | ? | ? | ERα agonist |
| Methylpiperidinopyrazole | MPP | 11 | 0.05 | ? | ? | ERα antagonist |
| Diarylpropionitrile | DPN | 0.12–0.25 | 6.6–18 | 32.4 | 1.7 | ERβ agonist |
| 8β-VE2 | 8β-Vinyl-17β-estradiol | 0.35 | 22.0–83 | 12.9 | 0.50 | ERβ agonist |
| Prinaberel | ERB-041; WAY-202,041 | 0.27 | 67–72 | ? | ? | ERβ agonist |
| ERB-196 | WAY-202,196 | ? | 180 | ? | ? | ERβ agonist |
| Erteberel | SERBA-1; LY-500,307 | ? | ? | 2.68 | 0.19 | ERβ agonist |
| SERBA-2 | – | ? | ? | 14.5 | 1.54 | ERβ agonist |
| Coumestrol | – | 9.225 (0.0117–94) | 64.125 (0.41–185) | 0.14–80.0 | 0.07–27.0 | Xenoestrogen |
| Genistein | – | 0.445 (0.0012–16) | 33.42 (0.86–87) | 2.6–126 | 0.3–12.8 | Xenoestrogen |
| Equol | – | 0.2–0.287 | 0.85 (0.10–2.85) | ? | ? | Xenoestrogen |
| Daidzein | – | 0.07 (0.0018–9.3) | 0.7865 (0.04–17.1) | 2.0 | 85.3 | Xenoestrogen |
| Biochanin A | – | 0.04 (0.022–0.15) | 0.6225 (0.010–1.2) | 174 | 8.9 | Xenoestrogen |
| Kaempferol | – | 0.07 (0.029–0.10) | 2.2 (0.002–3.00) | ? | ? | Xenoestrogen |
| Naringenin | – | 0.0054 (<0.001–0.01) | 0.15 (0.11–0.33) | ? | ? | Xenoestrogen |
| 8-Prenylnaringenin | 8-PN | 4.4 | ? | ? | ? | Xenoestrogen |
| Quercetin | – | <0.001–0.01 | 0.002–0.040 | ? | ? | Xenoestrogen |
| Ipriflavone | – | <0.01 | <0.01 | ? | ? | Xenoestrogen |
| Miroestrol | – | 0.39 | ? | ? | ? | Xenoestrogen |
| Deoxymiroestrol | – | 2.0 | ? | ? | ? | Xenoestrogen |
| β-Sitosterol | – | <0.001–0.0875 | <0.001–0.016 | ? | ? | Xenoestrogen |
| Resveratrol | – | <0.001–0.0032 | ? | ? | ? | Xenoestrogen |
| α-Zearalenol | – | 48 (13–52.5) | ? | ? | ? | Xenoestrogen |
| β-Zearalenol | – | 0.6 (0.032–13) | ? | ? | ? | Xenoestrogen |
| Zeranol | α-Zearalanol | 48–111 | ? | ? | ? | Xenoestrogen |
| Taleranol | β-Zearalanol | 16 (13–17.8) | 14 | 0.8 | 0.9 | Xenoestrogen |
| Zearalenone | ZEN | 7.68 (2.04–28) | 9.45 (2.43–31.5) | ? | ? | Xenoestrogen |
| Zearalanone | ZAN | 0.51 | ? | ? | ? | Xenoestrogen |
| Bisphenol A | BPA | 0.0315 (0.008–1.0) | 0.135 (0.002–4.23) | 195 | 35 | Xenoestrogen |
| Endosulfan | EDS | <0.001–<0.01 | <0.01 | ? | ? | Xenoestrogen |
| Kepone | Chlordecone | 0.0069–0.2 | ? | ? | ? | Xenoestrogen |
| o,p'-DDT | – | 0.0073–0.4 | ? | ? | ? | Xenoestrogen |
| p,p'-DDT | – | 0.03 | ? | ? | ? | Xenoestrogen |
| Methoxychlor | p,p'-Dimethoxy-DDT | 0.01 (<0.001–0.02) | 0.01–0.13 | ? | ? | Xenoestrogen |
| HPTE | Hydroxychlor; p,p'-OH-DDT | 1.2–1.7 | ? | ? | ? | Xenoestrogen |
| Testosterone | T; 4-Androstenolone | <0.0001–<0.01 | <0.002–0.040 | >5000 | >5000 | Androgen |
| Dihydrotestosterone | DHT; 5α-Androstanolone | 0.01 (<0.001–0.05) | 0.0059–0.17 | 221–>5000 | 73–1688 | Androgen |
| Nandrolone | 19-Nortestosterone; 19-NT | 0.01 | 0.23 | 765 | 53 | Androgen |
| Dehydroepiandrosterone | DHEA; Prasterone | 0.038 (<0.001–0.04) | 0.019–0.07 | 245–1053 | 163–515 | Androgen |
| 5-Androstenediol | A5; Androstenediol | 6 | 17 | 3.6 | 0.9 | Androgen |
| 4-Androstenediol | – | 0.5 | 0.6 | 23 | 19 | Androgen |
| 4-Androstenedione | A4; Androstenedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| 3α-Androstanediol | 3α-Adiol | 0.07 | 0.3 | 260 | 48 | Androgen |
| 3β-Androstanediol | 3β-Adiol | 3 | 7 | 6 | 2 | Androgen |
| Androstanedione | 5α-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Etiocholanedione | 5β-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Methyltestosterone | 17α-Methyltestosterone | <0.0001 | ? | ? | ? | Androgen |
| Ethinyl-3α-androstanediol | 17α-Ethynyl-3α-adiol | 4.0 | <0.07 | ? | ? | Estrogen |
| Ethinyl-3β-androstanediol | 17α-Ethynyl-3β-adiol | 50 | 5.6 | ? | ? | Estrogen |
| Progesterone | P4; 4-Pregnenedione | <0.001–0.6 | <0.001–0.010 | ? | ? | Progestogen |
| Norethisterone | NET; 17α-Ethynyl-19-NT | 0.085 (0.0015–<0.1) | 0.1 (0.01–0.3) | 152 | 1084 | Progestogen |
| Norethynodrel | 5(10)-Norethisterone | 0.5 (0.3–0.7) | <0.1–0.22 | 14 | 53 | Progestogen |
| Tibolone | 7α-Methylnorethynodrel | 0.5 (0.45–2.0) | 0.2–0.076 | ? | ? | Progestogen |
| Δ4-Tibolone | 7α-Methylnorethisterone | 0.069–<0.1 | 0.027–<0.1 | ? | ? | Progestogen |
| 3α-Hydroxytibolone | – | 2.5 (1.06–5.0) | 0.6–0.8 | ? | ? | Progestogen |
| 3β-Hydroxytibolone | – | 1.6 (0.75–1.9) | 0.070–0.1 | ? | ? | Progestogen |
| Footnotes: a = (1) Binding affinity values are of the format "median (range)" (# (#–#)), "range" (#–#), or "value" (#) depending on the values available. The full sets of values within the ranges can be found in the Wiki code. (2) Binding affinities were determined via displacement studies in a variety of in-vitro systems with labeled estradiol and human ERα and ERβ proteins (except the ERβ values from Kuiper et al. (1997), which are rat ERβ). Sources: See template page. | ||||||
Ecology
These compounds in plants are an important part of their defense system, mainly against fungi.[18]
Phytoestrogens are ancient naturally occurring substances, and as dietary phytochemicals they are considered as co-evolutive with mammals. In the human diet, phytoestrogens are not the only source of exogenous estrogens. Xenoestrogens (novel, man-made), are found as food additives[19] and ingredients, and also in cosmetics, plastics, and insecticides. Environmentally, they have similar effects as phytoestrogens, making it difficult to clearly separate the action of these two kind of agents in studies done on populations.[20]
Avian studies
The consumption of plants with unusual content of phytoestrogens, under drought conditions, has been shown to decrease fertility in quail.[21] Parrot food as available in nature has shown only weak estrogenic activity. Studies have been conducted on screening methods for environmental estrogens present in manufactured supplementary food, with the purpose of aiding reproduction of endangered species.[22]
Food sources
According to one study of nine common phytoestrogens in a Western diet, foods with the highest relative phytoestrogen content were nuts and oilseeds, followed by soy products, cereals and breads, legumes, meat products, and other processed foods that may contain soy, vegetables, fruits, alcoholic, and nonalcoholic beverages. Flax seed and other oilseeds contained the highest total phytoestrogen content, followed by soybeans and tofu.[23] The highest concentrations of isoflavones are found in soybeans and soybean products followed by legumes, whereas lignans are the primary source of phytoestrogens found in nuts and oilseeds (e.g. flax) and also found in cereals, legumes, fruits and vegetables. Phytoestrogen content varies in different foods, and may vary significantly within the same group of foods (e.g. soy beverages, tofu) depending on processing mechanisms and type of soybean used. Legumes (in particular soybeans), whole grain cereals, and some seeds are high in phytoestrogens.
A more comprehensive list of foods known to contain phytoestrogens includes:
- Soybeans and soy products
- Tempeh
- Linseed (flax)
- Sesame seeds
- Wheat berries
- Fenugreek (contains diosgenin, but also used to make Testofen, a compound taken by men to increase testosterone).
- Oats
- Barley
- Beans
- Lentils
- Yams
- Rice
- Alfalfa
- Mung beans
- Apples
- Carrots
- Pomegranates[24]
- Wheat germ
- Rice bran
- Lupin
- Kudzu
- Coffee
- Licorice root
- Mint
- Ginseng
- Hops,[25]
- Bourbon whiskey
- Beer,[26]
- Fennel
- Anise.[27]
- Red clover (sometimes a constituent of green manure).
- Spinach[28]
An epidemiological study of women in the United States found that the dietary intake of phytoestrogens in healthy post-menopausal Caucasian women is less than one milligram daily.[29]
Effects on humans
In human beings, phytoestrogens are readily absorbed into the circulatory system, circulate in plasma, and are excreted in the urine. Metabolic influence is different from that of grazing animals due to the differences between ruminant versus monogastric digestive systems.[20]
A wide range of beneficial effects of phytoestrogens on the cardiovascular, metabolic, central nervous systems as well as reduction of risk of cancer and post menopausal symptoms have been claimed. However, there is also concern that phytoestrogens may act as endocrine disruptors that adversely affect health. Based on currently available evidence, it is not clear whether the potential health benefits of phytoestrogens outweigh their risks.[30]
Males
It is unclear if phytoestrogens have any effect on male sexuality, with conflicting results about the potential effects of isoflavones originating from soy. A 2010 meta-analysis of fifteen placebo-controlled studies led by Dr. Mark Messina, executive director of the Soy Nutrition Institute, said that "neither soy foods nor isoflavone supplements alter measures of bioavailable testosterone concentrations in men."[31] Some studies showed that isoflavone supplementation had no effect on sperm concentration, count, or motility, and had no effects on testicular or ejaculate volume.[32][33] Sperm count decline and increasing rate of testicular cancers in the West may be linked to a higher presence of isoflavone phytoestrogens in the diet while in utero, but such a link has not been definitively proven.[34] Furthermore, there is some evidence that phytoestrogens may affect male fertility, but "further investigation is needed before a firm conclusion can be drawn".[35]
Females
It is unclear if phytoestrogens have any effect on the cause or prevention of cancer in females.[36] Some epidemiological studies have suggested a protective effect against breast cancer.[36][37] Other epidemiological studies found that consumption of soy estrogens is safe for patients with breast cancer, and that it may decrease mortality and recurrence rates.[38][39] It remains unclear if phytoestrogens can minimize some of the deleterious effects of low estrogen levels (hypoestrogenism) resulting from oophorectomy, menopause, or other causes.[36] A Cochrane Review of the use of phytoestrogens to relieve the vasomotor symptoms of menopause (hot flashes) stated that there was no conclusive evidence to suggest any benefit to their use, although genistein effects should be further investigated.[40]
Infant formula
Some studies have found that some concentrations of isoflavones may have effects on intestinal cells. At low doses, genistein acted as a weak estrogen and stimulated cell growth; at high doses, it inhibited proliferation and altered cell cycle dynamics. This biphasic response correlates with how genistein is thought to exert its effects.[41] Some reviews express the opinion that more research is needed to answer the question of what effect phytoestrogens may have on infants,[42][43] but their authors did not find any adverse effects. Studies conclude there are no adverse effects in human growth, development, or reproduction as a result of the consumption of soy-based infant formula compared to conventional cow-milk formula.[44][45][46] The American Academy of Pediatrics states: "although isolated soy protein-based formulas may be used to provide nutrition for normal growth and development, there are few indications for their use in place of cow milk-based formula. These indications include (a) for infants with galactosemia and hereditary lactase deficiency (rare) and (b) in situations in which a vegetarian diet is preferred."[47]
Ethnopharmacology
In some countries, phytoestrogenic plants have been used for centuries in the treatment of menstrual and menopausal problems, as well as for fertility problems.[48] Plants used that have been shown to contain phytoestrogens include Pueraria mirifica,[49] and its close relative, kudzu,[50] Angelica,[51] fennel,[27] and anise. In a rigorous study, the use of one such source of phytoestrogen, red clover, has been shown to be safe, but ineffective in relieving menopausal symptoms[52] (black cohosh is also used for menopausal symptoms, but does not contain phytoestrogens.[53])
References
- Yildiz F (2005). Phytoestrogens in Functional Foods. Taylor & Francis Ltd. pp. 3–5, 210–211. ISBN 978-1-57444-508-4.
- Hughes CL (Jun 1988). "Phytochemical mimicry of reproductive hormones and modulation of herbivore fertility by phytoestrogens". Environmental Health Perspectives. 78: 171–4. doi:10.1289/ehp.8878171. PMC 1474615. PMID 3203635.
- Bentley GR, Mascie-Taylor CG (2000). Infertility in the modern world: present and future prospects. Cambridge, UK: Cambridge University Press. pp. 99–100. ISBN 978-0-521-64387-0.
- Varner JE, Bonner J (1966). Plant Biochemistry. Academic Press. ISBN 978-0-12-114856-0.
- Bennetts HW, Underwood EJ, Shier FL (1946). "A specific breeding problem of sheep on subterranean clover pastures in Western Australia". Australian Veterinary Journal. 22 (1): 2–12. doi:10.1111/j.1751-0813.1946.tb15473.x. PMID 21028682.
- Cunningham IJ, Hogan KG (1954). "Oestrogens in New Zealand pasture plants". N. Z. Vet. J. 2 (4): 128–134. doi:10.1080/00480169.1954.33166.
- Johnston I (2003). Phytochem Functional Foods. CRC Press Inc. pp. 66–68. ISBN 978-0-8493-1754-5.
- Bennett GA, Shotwell OI (1979). "Zearalenone in cereal grains". J. Amer. Oil. Chemists Soc. 56 (9): 812–819. doi:10.1007/bf02909525.
- Kuiper-Goodman T, Scott PM, Watanabe H (1987). "Risk assessment of the mycotoxin zearalenone". Regul. Toxicol. Pharmacol. 7 (3): 253–306. doi:10.1016/0273-2300(87)90037-7. PMID 2961013.
- Zinedine A, Soriano JM, Moltó JC, Mañes J (2007). "Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: an oestrogenic mycotoxin". Food Chem. Toxicol. 45 (1): 1–18. doi:10.1016/j.fct.2006.07.030. PMID 17045381.
- Gallo A, Giuberti G, Frisvad JC, Bertuzzi T, Nielsen KF (2015). "Review on Mycotoxin Issues in Ruminants: Occurrence in Forages, Effects of Mycotoxin Ingestion on Health Status and Animal Performance and Practical Strategies to Counteract Their Negative Effects". Toxins (Basel). 7 (8): 3057–111. doi:10.3390/toxins7083057. PMC 4549740. PMID 26274974.
- Naz RK (1999). Endocrine Disruptors: Effects on Male and Female Reproductive Systems. CRC Press Inc. p. 90. ISBN 978-0-8493-3164-0.
- Turner JV, Agatonovic-Kustrin S, Glass BD (Aug 2007). "Molecular aspects of phytoestrogen selective binding at estrogen receptors". Journal of Pharmaceutical Sciences. 96 (8): 1879–85. doi:10.1002/jps.20987. PMID 17518366.
- Dang ZC, Lowik C (Jul 2005). "Dose-dependent effects of phytoestrogens on bone". Trends in Endocrinology and Metabolism. 16 (5): 207–13. doi:10.1016/j.tem.2005.05.001. PMID 15922618.
- Dang ZC (May 2009). "Dose-dependent effects of soy phyto-oestrogen genistein on adipocytes: mechanisms of action". Obesity Reviews. 10 (3): 342–9. doi:10.1111/j.1467-789X.2008.00554.x. PMID 19207876.
- Dang ZC, Audinot V, Papapoulos SE, Boutin JA, Löwik CW (Jan 2003). "Peroxisome proliferator-activated receptor gamma (PPARgamma ) as a molecular target for the soy phytoestrogen genistein". The Journal of Biological Chemistry. 278 (2): 962–7. doi:10.1074/jbc.M209483200. PMID 12421816.
- Dang Z, Löwik CW (May 2004). "The balance between concurrent activation of ERs and PPARs determines daidzein-induced osteogenesis and adipogenesis". Journal of Bone and Mineral Research. 19 (5): 853–61. doi:10.1359/jbmr.040120. PMID 15068509.
- Leegood RC, Lea P (1998). Plant Biochemistry and Molecular Biology. John Wiley & Sons. p. 211. ISBN 978-0-471-97683-7.
- Amadasi A, Mozzarelli A, Meda C, Maggi A, Cozzini P (2009). "Identification of xenoestrogens in food additives by an integrated in silico and in vitro approach". Chem. Res. Toxicol. 22 (1): 52–63. doi:10.1021/tx800048m. PMC 2758355. PMID 19063592.
- Korach KS (1998). Reproductive and Developmental Toxicology. Marcel Dekker Ltd. pp. 278–279. ISBN 978-0-8247-9857-4.
- Leopold AS, Erwin M, Oh J, Browning B (January 1976). "Phytoestrogens: adverse effects on reproduction in California quail". Science. 191 (4222): 98–100. Bibcode:1976Sci...191...98S. doi:10.1126/science.1246602. PMID 1246602.
- Fidler AE, Zwart S, Pharis RP, Weston RJ, Lawrence SB, Jansen P, Elliott G, Merton DV (2000). "Screening the foods of an endangered parrot, the kakapo (Strigops habroptilus), for oestrogenic activity using a recombinant yeast bioassay". Reproduction, Fertility, and Development. 12 (3–4): 191–9. doi:10.1071/RD00041. PMID 11302429.
- Thompson LU, Boucher BA, Liu Z, Cotterchio M, Kreiger N (2006). "Phytoestrogen content of foods consumed in Canada, including isoflavones, lignans, and coumestan". Nutrition and Cancer. 54 (2): 184–201. doi:10.1207/s15327914nc5402_5. PMID 16898863.
- van Elswijk DA, Schobel UP, Lansky EP, Irth H, van der Greef J (Jan 2004). "Rapid dereplication of estrogenic compounds in pomegranate (Punica granatum) using on-line biochemical detection coupled to mass spectrometry". Phytochemistry. 65 (2): 233–41. doi:10.1016/j.phytochem.2003.07.001. PMID 14732284.
- Chadwick LR, Nikolic D, Burdette JE, Overk CR, Bolton JL, van Breemen RB, Fröhlich R, Fong HH, Farnsworth NR, Pauli GF (Dec 2004). "Estrogens and congeners from spent hops (Humulus lupulus)". Journal of Natural Products. 67 (12): 2024–32. doi:10.1021/np049783i. PMID 15620245.
- Rosenblum ER, Stauber RE, Van Thiel DH, Campbell IM, Gavaler JS (Dec 1993). "Assessment of the estrogenic activity of phytoestrogens isolated from bourbon and beer". Alcoholism: Clinical and Experimental Research. 17 (6): 1207–9. doi:10.1111/j.1530-0277.1993.tb05230.x. PMID 8116832.
- Albert-Puleo M (Dec 1980). "Fennel and anise as estrogenic agents". Journal of Ethnopharmacology. 2 (4): 337–44. doi:10.1016/S0378-8741(80)81015-4. PMID 6999244.
- Bacciottini, Lucia; Falchetti, Alberto; Pampaloni, Barbara; Bartolini, Elisa; Carossino, Anna Maria; Brandi, Maria Luisa (2007). "Phytoestrogens: food or drug?". Clinical Cases in Mineral and Bone Metabolism. 4 (2): 123–130. ISSN 1724-8914. PMC 2781234. PMID 22461212.
- de Kleijn MJ, van der Schouw YT, Wilson PW, Adlercreutz H, Mazur W, Grobbee DE, Jacques PF (Jun 2001). "Intake of dietary phytoestrogens is low in postmenopausal women in the United States: the Framingham study(1-4)". The Journal of Nutrition. 131 (6): 1826–32. doi:10.1093/jn/131.6.1826. PMID 11385074.
- Rietjens IM, Louisse J, Beekmann K (June 2017). "The potential health effects of dietary phytoestrogens". British Journal of Pharmacology. 174 (11): 1263–1280. doi:10.1111/bph.13622. PMC 5429336. PMID 27723080.
- Hamilton-Reeves JM, Vazquez G, Duval SJ, Phipps WR, Kurzer MS, Messina MJ (Aug 2010). "Clinical studies show no effects of soy protein or isoflavones on reproductive hormones in men: results of a meta-analysis". Fertility and Sterility. 94 (3): 997–1007. doi:10.1016/j.fertnstert.2009.04.038. PMID 19524224.
- Dabrowski WM (2004). Toxins in Food. CRC Press Inc. p. 95. ISBN 978-0-8493-1904-4.
- Mitchell JH, Cawood E, Kinniburgh D, Provan A, Collins AR, Irvine DS (Jun 2001). "Effect of a phytoestrogen food supplement on reproductive health in normal males". Clinical Science. 100 (6): 613–8. doi:10.1042/CS20000212. PMID 11352776.
- Patisaul HB, Jefferson W (2010). "The pros and cons of phytoestrogens". Frontiers in Neuroendocrinology. 31 (4): 400–19. doi:10.1016/j.yfrne.2010.03.003. PMC 3074428. PMID 20347861.
- Cederroth CR, Auger J, Zimmermann C, Eustache F, Nef S (2010). "Soy, phyto-oestrogens and male reproductive function: a review". International Journal of Andrology. 33 (2): 304–16. doi:10.1111/j.1365-2605.2009.01011.x. PMID 19919579.
- Bilal I, Chowdhury A, Davidson J, Whitehead S (2014). "Phytoestrogens and prevention of breast cancer: The contentious debate". World Journal of Clinical Oncology. 5 (4): 705–12. doi:10.5306/wjco.v5.i4.705. PMC 4129534. PMID 25302172.
- Ingram D, Sanders K, Kolybaba M, Lopez D (Oct 1997). "Case-control study of phyto-oestrogens and breast cancer". Lancet. 350 (9083): 990–4. doi:10.1016/S0140-6736(97)01339-1. PMID 9329514.
- Shu XO, Zheng Y, Cai H, Gu K, Chen Z, Zheng W, Lu W (Dec 2009). "Soy food intake and breast cancer survival". JAMA. 302 (22): 2437–43. doi:10.1001/jama.2009.1783. PMC 2874068. PMID 19996398.
- Fritz H, Seely D, Flower G, Skidmore B, Fernandes R, Vadeboncoeur S, Kennedy D, Cooley K, Wong R, Sagar S, Sabri E, Fergusson D (2013). "Soy, red clover, and isoflavones and breast cancer: a systematic review". PLOS ONE. 8 (11): e81968. Bibcode:2013PLoSO...881968F. doi:10.1371/journal.pone.0081968. PMC 3842968. PMID 24312387.
- Lethaby A, Marjoribanks J, Kronenberg F, Roberts H, Eden J, Brown J (2013). "Phytoestrogens for menopausal vasomotor symptoms". The Cochrane Database of Systematic Reviews (12): CD001395. doi:10.1002/14651858.CD001395.pub4. PMID 24323914.
- Chen AC, Donovan SM (Jun 2004). "Genistein at a concentration present in soy infant formula inhibits Caco-2BBe cell proliferation by causing G2/M cell cycle arrest". The Journal of Nutrition. 134 (6): 1303–8. doi:10.1093/jn/134.6.1303. PMID 15173388.
- Miniello VL, Moro GE, Tarantino M, Natile M, Granieri L, Armenio L (Sep 2003). "Soy-based formulas and phyto-oestrogens: a safety profile". Acta Paediatrica. 91 (441): 93–100. doi:10.1111/j.1651-2227.2003.tb00655.x. PMID 14599051.
- Chen A, Rogan WJ (2004). "Isoflavones in soy infant formula: a review of evidence for endocrine and other activity in infants". Annual Review of Nutrition. 24 (1): 33–54. doi:10.1146/annurev.nutr.24.101603.064950. PMID 15189112.
- Strom BL, Schinnar R, Ziegler EE, Barnhart KT, Sammel MD, Macones GA, Stallings VA, Drulis JM, Nelson SE, Hanson SA (Aug 2001). "Exposure to soy-based formula in infancy and endocrinological and reproductive outcomes in young adulthood". JAMA. 286 (7): 807–14. doi:10.1001/jama.286.7.807. PMID 11497534.
- Giampietro PG, Bruno G, Furcolo G, Casati A, Brunetti E, Spadoni GL, Galli E (Feb 2004). "Soy protein formulas in children: no hormonal effects in long-term feeding". Journal of Pediatric Endocrinology & Metabolism. 17 (2): 191–6. doi:10.1515/JPEM.2004.17.2.191. PMID 15055353.
- Merritt RJ, Jenks BH (May 2004). "Safety of soy-based infant formulas containing isoflavones: the clinical evidence". The Journal of Nutrition. 134 (5): 1220S–1224S. doi:10.1093/jn/134.5.1220S. PMID 15113975.
- Bhatia J, Greer F (May 2008). "Use of soy protein-based formulas in infant feeding". Pediatrics. 121 (5): 1062–8. doi:10.1542/peds.2008-0564. PMID 18450914.
- Muller-Schwarze D (2006). Chemical Ecology of Vertebrates. Cambridge University Press. p. 287. ISBN 978-0-521-36377-8.
- Lee YS, Park JS, Cho SD, Son JK, Cherdshewasart W, Kang KS (December 2002). "Requirement of metabolic activation for estrogenic activity of Pueraria mirifica". Journal of Veterinary Science. 3 (4): 273–277. doi:10.4142/jvs.2002.3.4.273. PMID 12819377. Archived from the original on 20 November 2008.
- Delmonte P, Rader JI (2006). "Analysis of isoflavones in foods and dietary supplements". Journal of AOAC International. 89 (4): 1138–1146. PMID 16915857.
- Brown D, Walton N (1999). Chemicals from plants: Perspectives on plant secondary products. World Scientific Publishing. pp. 21, 141. ISBN 978-981-02-2773-9.
- Geller SE, Shulman LP, van Breemen RB, Banuvar S, Zhou Y, Epstein G, Hedayat S, Nikolic D, Krause EC, Piersen CE, Bolton JL, Pauli GF, Farnsworth NR (2009). "Safety and efficacy of black cohosh and red clover for the management of vasomotor symptoms: A randomized controlled trial". Menopause. 16 (6): 1156–1166. doi:10.1097/gme.0b013e3181ace49b. PMC 2783540. PMID 19609225.
- Kennelly EJ, Baggett S, Nuntanakorn P, Ososki AL, Mori SA, Duke J, Coleton M, Kronenberg F (Jul 2002). "Analysis of thirteen populations of black cohosh for formononetin". Phytomedicine. 9 (5): 461–467. doi:10.1078/09447110260571733. PMID 12222669.