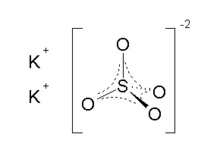
Potassium sulfate
Potassium sulfate (US) or potassium sulphate (UK), also called sulphate of potash (SOP), arcanite, or archaically potash of sulfur, is the inorganic compound with formula K2SO4, a white water-soluble solid. It is commonly used in fertilizers, providing both potassium and sulfur.
 Arcanite | |
 | |
| Names | |
|---|---|
| Other names
Potassium sulphate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.029.013 |
| E number | E515(i) (acidity regulators, ...) |
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| K2SO4 | |
| Molar mass | 174.259 g/mol |
| Appearance | White solid |
| Odor | odorless |
| Density | 2.66 g/cm3[1] |
| Melting point | 1,069[2] °C (1,956 °F; 1,342 K) |
| Boiling point | 1,689 °C (3,072 °F; 1,962 K) |
| 111 g/L (20 °C) 120 g/L (25 °C) 240 g/L (100 °C) | |
| Solubility | slightly soluble in glycerol insoluble in acetone, alcohol, CS2 |
| −67.0·10−6 cm3/mol | |
Refractive index (nD) |
1.495 |
| Structure | |
| orthorhombic | |
| Hazards | |
| Main hazards | Irritant |
| Safety data sheet | External MSDS |
| R-phrases (outdated) | R22 |
| S-phrases (outdated) | S36 |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
6600 mg/kg (oral, rat)[3] |
| Related compounds | |
Other anions |
Potassium selenate Potassium tellurate |
Other cations |
Lithium sulfate Sodium sulfate Rubidium sulfate Caesium sulfate |
Related compounds |
Potassium hydrogen sulfate Potassium sulfite Potassium bisulfite Potassium persulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
History
Potassium sulfate (K2SO4) has been known since early in the 14th century. It was studied by Glauber, Boyle, and Tachenius. In the 17th century, it was named arcanuni or sal duplicatum, as it was a combination of an acid salt with an alkaline salt. It was also known as vitriolic tartar and Glaser's salt or sal polychrestum Glaseri after the pharmaceutical chemist Christopher Glaser who prepared it and used medicinally.[4][5]
Known as arcanum duplicatum ("double secret") or panacea duplicata in pre-modern medicine, it was prepared from the residue (caput mortuum) left over from the production of aqua fortis (nitric acid, HNO3) from nitre (potassium nitrate, KNO3) and oil of vitriol (sulphuric acid, H2SO4) via Glauber's process:
- 2 KNO3 + H2SO4 → 2 HNO3 + K2SO4
The residue was dissolved in hot water, filtered, and evaporated to a cuticle. It was then left to crystallise. It was used as a diuretic and sudorific.[6]
According to Chambers's Cyclopedia, the recipe was purchased for five hundred thalers by Charles Frederick, Duke of Holstein-Gottorp. Schroder, the duke's physician, wrote wonders of its great uses in hypochondriacal cases, continued and intermitting fevers, stone, scurvy, etc.[6]
Natural resources
The mineral form of potassium sulfate, arcanite, is relatively rare. Natural resources of potassium sulfate are minerals abundant in the Stassfurt salt. These are cocrystallizations of potassium sulfate and sulfates of magnesium calcium and sodium.
Relevant minerals are:
- Kainite, KMg(SO4)·Cl·3H2O
- Schönite (now known as picromerite), K2SO4·MgSO4·6H2O
- Leonite, K2SO4·MgSO4·4H2O
- Langbeinite, K2Mg2(SO4)3
- Aphthitalite (previously known as glaserite), K3Na(SO4)2
- Polyhalite, K2SO4·MgSO4·2CaSO4·2H2O
The potassium sulfate can be separated from some of these minerals, like kainite, because the corresponding salt is less soluble in water.
Kieserite, MgSO4·H2O, can be combined with a solution of potassium chloride to produce potassium sulfate.
Production
Approximately 1.5 million tons were produced in 1985, typically by the reaction of potassium chloride with sulfuric acid, analogous to the Mannheim process for producing sodium sulfate.[7] The process involves intermediate formation of potassium bisulfate, an exothermic reaction that occurs at room temperature:
- KCl + H2SO4 → HCl + KHSO4
The second step of the process is endothermic, requiring energy input:
- KCl + KHSO4 → HCl + K2SO4
Structure and properties
Two crystalline forms are known. Orthorhombic β-K2SO4 is the common form, but it converts to α-K2SO4 above 583 °C.[7] These structures are complex, although the sulfate adopts the typical tetrahedral geometry.[8]
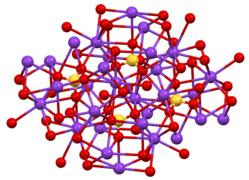 Structure of β-K2SO4.
Structure of β-K2SO4.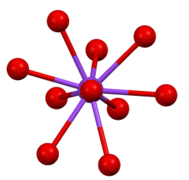 Coordination sphere of one of two types of K+ site.
Coordination sphere of one of two types of K+ site.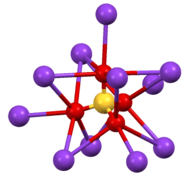 SO4 environment in β-K2SO4.
SO4 environment in β-K2SO4.
It does not form a hydrate, unlike sodium sulfate. The salt crystallizes as double six-sided pyramids, classified as rhombic. They are transparent, very hard and have a bitter, salty taste. The salt is soluble in water, but insoluble in solutions of potassium hydroxide (sp. gr. 1.35), or in absolute ethanol.
When potassium sulfate is heated in water and subject to swirling in a beaker, the crystals form a multi-arm spiral structure when allowed to settle.[9]
Uses
The dominant use of potassium sulfate is as a fertilizer. K2SO4 does not contain chloride, which can be harmful to some crops. Potassium sulfate is preferred for these crops, which include tobacco and some fruits and vegetables. Crops that are less sensitive may still require potassium sulfate for optimal growth if the soil accumulates chloride from irrigation water.[10]
The crude salt is also used occasionally in the manufacture of glass. Potassium sulfate is also used as a flash reducer in artillery propellant charges. It reduces muzzle flash, flareback and blast overpressure.
It is sometimes used as an alternative blast media similar to soda in soda blasting as it is harder and similarly water-soluble.[11]
Potassium sulfate can also be used in pyrotechnics in combination with potassium nitrate to generate a purple flame.
Reactions
Acidification
Potassium hydrogen sulfate (also known as potassium bisulfate), KHSO4, is readily produced by reacting K2SO4 with sulfuric acid. It forms rhombic pyramids, which melt at 197 °C (387 °F). It dissolves in three parts of water at 0 °C (32 °F). The solution behaves much as if its two congeners, K2SO4 and H2SO4, were present side by side of each other uncombined; an excess of ethanol the precipitates normal sulfate (with little bisulfate) with excess acid remaining.
The behavior of the fused dry salt is similar when heated to several hundred degrees; it acts on silicates, titanates, etc., the same way as sulfuric acid that is heated beyond its natural boiling point does. Hence it is frequently used in analytical chemistry as a disintegrating agent. For information about other salts that contain sulfate, see sulfate.
References
- Patnaik, Pradyot (2002). Handbook of Inorganic Chemicals. McGraw-Hill. ISBN 978-0-07-049439-8.
- Windholtz, M; Budavari, S, eds. (1983). The Merck Index. Rahway, New Jersey: Merck & Co.
- Chambers, Michael. "Potassium sulfate RN: 7778-80-5". ChemIDplus. United States National Library of Medicine.
- De Milt, Clara (1942). "Christopher Glaser". Journal of Chemical Education. 19 (2): 53. doi:10.1021/ed019p53.
- van Klooster, H. S. (1959). "Three centuries of Rochelle salt". Journal of Chemical Education. 36 (7): 314. doi:10.1021/ed036p314.
-

- Schultz, H.; Bauer, G.; Schachl, E.; Hagedorn, F.; Schmittinger, P. (2005). "Potassium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_039. ISBN 3527306730.
- Gaultier, M.; Pannetier, G. (1968). "Structure cristalline de la forme 'basse température' du sulfate de potassium K2SO4-beta" [Crystal structure of the "low temperature" β-form of potassium sulfate]. Bulletin de la Société Chimique de France (in French). 1: 105–112.
- Thomas, S. (2017). "Potassium sulfate forms a spiral structure when dissolved in solution". Russian Journal of Physical Chemistry B. 11: 195–198. doi:10.1134/S1990793117010328.
- United Nations Industrial Development Organization; International Fertilizer Development Center (1998). Fertilizer manual (3rd ed.). Dordrecht, Netherlands: Kluwer Academic. ISBN 978-0-7923-5032-3.
- "Super K (Potassium Sulphate)". American Surface Prep. Archived from the original on 9 December 2014. Retrieved 7 December 2014.