Glibenclamide
Glibenclamide, also known as glyburide, is a medication used to treat diabetes mellitus type 2.[1] It is recommended that it be taken together with diet and exercise.[1] It may be used with other antidiabetic medication.[1] It is not recommended for use by itself in diabetes mellitus type 1.[1] It is taken by mouth.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Diabeta, Flycron, others[1] |
| Other names | Glyburide (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684058 |
| License data | |
| Pregnancy category | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | Extensive |
| Metabolism | Liver hydroxylation (CYP2C9-mediated) |
| Elimination half-life | 10 hours |
| Excretion | Kidney and biliary |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.030.505 |
| Chemical and physical data | |
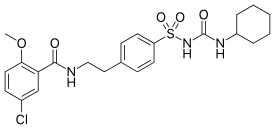
| Formula | C23H28ClN3O5S |
| Molar mass | 494.00 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 169 to 170 °C (336 to 338 °F) |
| |
| |
| (verify) | |
Common side effects include nausea and heartburn.[1] Serious side effects may include angioedema and low blood sugar.[1] It is generally not recommended during pregnancy but can be used during breastfeeding.[2] It is in the sulfonylureas class of medications and works by increasing the release of insulin from the pancreas.[1]
Glibenclamide was discovered in 1969 and approved for medical use in the United States in 1984.[3][1] It is available as a generic medication.[2] In 2017, it was the 174th most commonly prescribed medication in the United States, with more than three million prescriptions.[4][5]
Medical uses
It is used in the treatment of type 2 diabetes.
It is not as good as either metformin or insulin in those who have gestational diabetes.[6]
Side effects
Frequently reported side effects include: nausea, heartburn, weight gain, and bloating.[7] The medication is also a major cause of medication-induced hypoglycemia. The risk is greater than with other sulfonylureas.[8] Cholestatic jaundice is noted.
Glibenclamide may be not recommended in those with G6PD deficiency, as it may cause acute hemolysis.[9]
Pregnancy and breastfeeding
It is generally not recommended during pregnancy but can be used during breastfeeding.[2]
Mechanism of action
The medication works by binding to and inhibiting the ATP-sensitive potassium channels (KATP) inhibitory regulatory subunit sulfonylurea receptor 1 (SUR1)[10] in pancreatic beta cells. This inhibition causes cell membrane depolarization, opening voltage-dependent calcium channels. This results in an increase in intracellular calcium in the pancreatic beta cell and subsequent stimulation of insulin release.
After a cerebral ischemic insult, the blood–brain barrier is broken and glibenclamide can reach the central nervous system. Glibenclamide has been shown to bind more efficiently to the ischemic hemisphere.[11] Moreover, under ischemic conditions SUR1, the regulatory subunit of the KATP- and the NCCa-ATP-channels, is expressed in neurons, astrocytes, oligodendrocytes, endothelial cells[12] and by reactive microglia.[11]
History
It was developed in 1966 in a cooperative study between Boehringer Mannheim (now part of Roche) and Hoechst (now part of Sanofi-Aventis).[13]
Trade names
Glibenclamide is available as a generic, is manufactured by many pharmaceutical companies and is sold in doses of 1.25, 2.5 and 5 mg under many brand names including Gliben-J, Daonil, Diabeta, Euglucon, Gilemal, Glidanil, Glybovin, Glynase, Maninil, Micronase and Semi-Daonil. It is also available in a fixed-dose combination drug with metformin that is sold under various trade names, e.g. Bagomet Plus, Benimet, Glibomet, Gluconorm, Glucored, Glucovance, Metglib and many others.
References
- "Glyburide Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 18 March 2019.
- British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 692. ISBN 9780857113382.
- Diabetes in Clinical Practice: Questions and Answers from Case Studies. John Wiley & Sons. 2007. p. 342. ISBN 9780470059135.
- "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- "Glyburide - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
- Balsells, M; García-Patterson, A; Solà, I; Roqué, M; Gich, I; Corcoy, R (21 January 2015). "Glibenclamide, metformin, and insulin for the treatment of gestational diabetes: a systematic review and meta-analysis". BMJ (Clinical Research Ed.). 350: h102. doi:10.1136/bmj.h102. PMC 4301599. PMID 25609400.
- "Glyburide: MedlinePlus Drug Information". medlineplus.gov. Retrieved 29 October 2019.
- Gangji, A. S.; Cukierman, T.; Gerstein, H. C.; Goldsmith, C. H.; Clase, C. M. (1 February 2007). "A Systematic Review and Meta-Analysis of Hypoglycemia and Cardiovascular Events: A comparison of glyburide with other secretagogues and with insulin". Diabetes Care. 30 (2): 389–394. doi:10.2337/dc06-1789. PMID 17259518.
- Meloni G, Meloni T (January 1996). "Glyburide-induced acute haemolysis in a G6PD-deficient patient with NIDDM". Br. J. Haematol. 92 (1): 159–60. doi:10.1046/j.1365-2141.1996.275810.x. PMID 8562390.
- Serrano-Martín X, Payares G, Mendoza-León A (December 2006). "Glibenclamide, a blocker of K+(ATP) channels, shows antileishmanial activity in experimental murine cutaneous leishmaniasis". Antimicrob. Agents Chemother. 50 (12): 4214–6. doi:10.1128/AAC.00617-06. PMC 1693980. PMID 17015627.
- Ortega FJ, Gimeno-Bayon J, Espinosa-Parrilla JF, Carrasco JL, Batlle M, Pugliese M, Mahy N, Rodríguez MJ (May 2012). "ATP-dependent potassium channel blockade strengthens microglial neuroprotection after hypoxia-ischemia in rats" (PDF). Exp. Neurol. 235 (1): 282–96. doi:10.1016/j.expneurol.2012.02.010. hdl:2445/34278. PMID 22387180.
- Simard JM, Woo SK, Schwartzbauer GT, Gerzanich V (September 2012). "Sulfonylurea receptor 1 in central nervous system injury: a focused review". J. Cereb. Blood Flow Metab. 32 (9): 1699–717. doi:10.1038/jcbfm.2012.91. PMC 3434627. PMID 22714048.
- Marble A (1971). "Glibenclamide, a new sulphonylurea: whither oral hypoglycaemic agents?". Drugs. 1 (2): 109–15. doi:10.2165/00003495-197101020-00001. PMID 4999930.