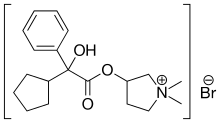
Glycopyrronium bromide
Glycopyrronium bromide is a medication of the muscarinic anticholinergic group. It does not cross the blood–brain barrier and consequently has few to no central effects. It is available in oral, intravenous, topical, and inhalated forms. It is a synthetic quaternary ammonium compound.
 | |
| Clinical data | |
|---|---|
| Trade names | Robinul, Cuvposa, Seebri, others |
| License data | |
| Pregnancy category | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.008.990 |
| Chemical and physical data | |
| Formula | C19H28BrNO3 |
| Molar mass | 398.341 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602014 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous, inhalation, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 0.6–1.2 hours |
| Excretion | 85% renal, unknown amount in the bile |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.008.990 |
| Chemical and physical data | |
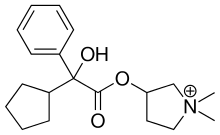
| Formula | C19H28NO3+ |
| Molar mass | 318.437 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
The cation, which is the active moiety, is called glycopyrronium (INN)[1] or glycopyrrolate (USAN).
In June 2018, glycopyrronium was approved by the FDA to treat excessive underarm sweating, becoming the first drug developed specifically to reduce excessive sweating.[2]
Medical uses
Glycopyrronium was first used in 1961 to treat peptic ulcers. Since 1975, intravenous glycopyrronium has been used before surgery to reduce salivary, tracheobronchial, and pharyngeal secretions.[3] It is also used in conjunction with neostigmine, a neuromuscular blocking reversal agent, to prevent neostigmine's muscarinic effects such as bradycardia. It can be administered to raise the heart rate in bradycardia, which often will also increase the blood pressure.
It is also used to reduce excessive saliva (sialorrhea),[4][5][6] and Ménière's disease.[7]
It has been used topically and orally to treat hyperhidrosis, in particular, gustatory hyperhidrosis.[8][9]
In inhalable form it is used to treat chronic obstructive pulmonary disease (COPD).[10][11] Doses for inhalation are much lower than oral ones, so that swallowing a dose will not have an effect.[12][13]
In end of life care, this medication will often be administered to patients that become encumbered by their bronchial secretions and are no longer able to clear their own airway through insufficient coughing reflex.
Side effects
Dry mouth, urinary retention, headaches, vomiting, diarrhea, constipation, blurry vision are possible side effects of the medication.[3] Since glycopyrronium reduces the body's sweating ability, it can even cause hyperthermia and heat stroke in hot environments. The medication also induces drowsiness, an effect exacerbated by the consumption of alcohol.
Pharmacology
Mechanism of action
Glycopyrronium competitively blocks muscarinic receptors,[3][14] thus inhibiting cholinergic transmission.
Pharmacokinetics
Glycopyrronium bromide affects the gastrointestinal tracts, liver and kidney but has a very limited effect on the brain and the central nervous system. In horse studies, after a single intravenous infusion, the observed tendencies of glycopyrronium followed a tri-exponential equation, by rapid disappearance from the blood followed by a prolonged terminal phase. Excretion was mainly in urine and in the form of an unchanged drug. Glycopyrronium has a relatively slow diffusion rate, and in a standard comparison to atropine, is more resistant to penetration through the blood-brain barrier and placenta.[15]
Research
It has been studied in asthma.[16][17]
References
- Bajaj V, Langtry JA (July 2007). "Use of oral glycopyrronium bromide in hyperhidrosis". Br. J. Dermatol. 157 (1): 118–21. doi:10.1111/j.1365-2133.2007.07884.x. PMID 17459043.
- "FDA OKs first drug made to reduce excessive sweating". AP News. Retrieved 2018-07-02.
- Chabicovsky, Monika; Winkler, Swantje; Soeberdt, Michael; Kilic, Ana; Masur, Clarissa; Abels, Christoph (May 2019). "Pharmacology, toxicology and clinical safety of glycopyrrolate". Toxicology and Applied Pharmacology. 370: 154–169. doi:10.1016/j.taap.2019.03.016.
- Mier RJ, Bachrach SJ, Lakin RC, Barker T, Childs J, Moran M (December 2000). "Treatment of sialorrhea with glycopyrrolate: A double-blind, dose-ranging study". Arch Pediatr Adolesc Med. 154 (12): 1214–8. doi:10.1001/archpedi.154.12.1214. PMID 11115305.
- Tscheng DZ (November 2002). "Sialorrhea - therapeutic drug options". Ann Pharmacother. 36 (11): 1785–90. doi:10.1345/aph.1C019. PMID 12398577.
- Olsen AK, Sjøgren P (October 1999). "Oral glycopyrrolate alleviates drooling in a patient with tongue cancer". J Pain Symptom Manage. 18 (4): 300–2. doi:10.1016/S0885-3924(99)00080-9. PMID 10534970.
- Maria, Sammartano Azia; Claudia, Cassandro; Pamela, Giordano; Andrea, Canale; Roberto, Albera (1 December 2012). "Medical therapy in Ménière's disease". Audiological Medicine. 10 (4): 171–177. doi:10.3109/1651386X.2012.718413.
- Kim WO, Kil HK, Yoon DM, Cho MJ (August 2003). "Treatment of compensatory gustatory hyperhidrosis with topical glycopyrrolate". Yonsei Med. J. 44 (4): 579–82. doi:10.3349/ymj.2003.44.4.579. PMID 12950111.
- Kim WO, Kil HK, Yoon KB, Yoon DM (May 2008). "Topical glycopyrrolate for patients with facial hyperhidrosis". Br. J. Dermatol. 158 (5): 1094–7. doi:10.1111/j.1365-2133.2008.08476.x. PMID 18294315.
- "Seebri Breezhaler EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 28 February 2020.
- "Tovanor Breezhaler EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 28 February 2020.
- "EPAR – Product information for Seebri Breezhaler" (PDF). European Medicines Agency. 28 September 2012.
- Tzelepis G, Komanapolli S, Tyler D, Vega D, Fulambarker A (January 1996). "Comparison of nebulized glycopyrrolate and metaproterenol in chronic obstructive pulmonary disease". Eur. Respir. J. 9 (1): 100–3. doi:10.1183/09031936.96.09010100. PMID 8834341.
- Haddad EB, Patel H, Keeling JE, Yacoub MH, Barnes PJ, Belvisi MG (May 1999). "Pharmacological characterization of the muscarinic receptor antagonist, glycopyrrolate, in human and guinea-pig airways". Br. J. Pharmacol. 127 (2): 413–20. doi:10.1038/sj.bjp.0702573. PMC 1566042. PMID 10385241.
- Rumpler, M.J.; Colahan, P.; Sams, R.A. (2014). "The pharmacokinetics of glycopyrrolate in Standardbred horses". J. Vet Pharmacol Ther. 37 (3): 260–8. doi:10.1111/jvp.12085. PMID 24325462.
- Hansel TT, Neighbour H, Erin EM, et al. (October 2005). "Glycopyrrolate causes prolonged bronchoprotection and bronchodilatation in patients with asthma". Chest. 128 (4): 1974–9. doi:10.1378/chest.128.4.1974. PMID 16236844. Archived from the original on 2013-04-14.
- Gilman MJ, Meyer L, Carter J, Slovis C (November 1990). "Comparison of aerosolized glycopyrrolate and metaproterenol in acute asthma". Chest. 98 (5): 1095–8. doi:10.1378/chest.98.5.1095. PMID 2225951. Archived from the original on 2013-04-14.
External links
- "Glycopyrronium bromide". Drug Information Portal. U.S. National Library of Medicine.
- "Glycopyrronium". Drug Information Portal. U.S. National Library of Medicine.