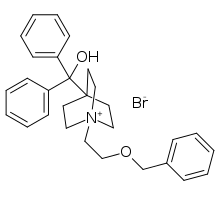
Umeclidinium bromide
Umeclidinium bromide (trade name Incruse Ellipta) is a long-acting muscarinic antagonist approved for the maintenance treatment of chronic obstructive pulmonary disease (COPD).[1] It is also approved for this indication in combination with vilanterol (as umeclidinium bromide/vilanterol)[2][3] and also as a triple-therapy combination as fluticasone furoate/umeclidinium bromide/vilanterol.[4]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Incruse Ellipta |
| Other names | GSK573719A |
| License data | |
| Pregnancy category |
|
| Routes of administration | Inhalation (DPI) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | ~89%[1] |
| Metabolism | Hepatic (CYP2D6) |
| Elimination half-life | 11 hours |
| Excretion | Feces (58%) and urine (22%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.166.375 |
| Chemical and physical data | |
| Formula | C29H34BrNO2 |
| Molar mass | 508.500 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
References
- "Incruse Ellipta (umeclidinium inhalation powder) for Oral Inhalation Use. Full Prescribing Information" (PDF). GlaxoSmithKline, Research Triangle Park, NC 27709. Retrieved 22 February 2016.
- Feldman GJ, Edin A (December 2013). "The combination of umeclidinium bromide and vilanterol in the management of chronic obstructive pulmonary disease: current evidence and future prospects". Therapeutic Advances in Respiratory Disease. 7 (6): 311–9. doi:10.1177/1753465813499789. PMID 24004659.
- "FDA Approves Umeclidinium and Vilanterol Combo for COPD". Medscape. December 18, 2013.
- "TRELEGY ELLIPTA Package Insert" (PDF). GlaxoSmithKline. September 2017. Retrieved 11 July 2020.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.