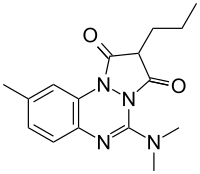
Azapropazone
Azapropazone is a nonsteroidal anti-inflammatory drug (NSAID). It is manufactured by Goldshield under the tradename Rheumox.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Rheumox |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 20 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.033.543 |
| Chemical and physical data | |
| Formula | C16H20N4O2 |
| Molar mass | 300.362 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
It was available in the UK as a prescription-only drug, with restrictions due to certain contra-indications and side-effects.[2] Azopropazone has now been discontinued in the British National Formulary.
Azapropazone has a half-life of approximately 20 hours in humans and is not extensively metabolized.[3]
References
- "Rheumox Capsules". South Africa Electronic Package Inserts.
- "Azapropazone". Patient UK. Archived from the original on 12 April 2009.
- Jones CJ (1976). "The pharmacology and pharmacokinetics of azapropazone - a review". Current Medical Research and Opinion. 4 (1): 3–16. doi:10.1185/03007997609109361. PMID 770078.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.