Diabetes
Diabetes mellitus (DM), commonly known as diabetes, is a group of metabolic disorders characterized by a high blood sugar level over a prolonged period of time.[11] Symptoms often include frequent urination, increased thirst, and increased appetite.[2] If left untreated, diabetes can cause many complications.[2] Acute complications can include diabetic ketoacidosis, hyperosmolar hyperglycemic state, or death.[3] Serious long-term complications include cardiovascular disease, stroke, chronic kidney disease, foot ulcers, damage to the nerves, damage to the eyes and cognitive impairment.[2][5]
| Diabetes mellitus | |
|---|---|
 | |
| Universal blue circle symbol for diabetes.[1] | |
| Pronunciation |
|
| Specialty | Endocrinology |
| Symptoms | Frequent urination, increased thirst, increased hunger[2] |
| Complications | Diabetic ketoacidosis, hyperosmolar hyperglycemic state, heart disease, stroke, chronic kidney failure, foot ulcers, cognitive impairment, gastroparesis[2][3][4][5] |
| Risk factors | Type 1: Family history[6] Type 2: Obesity, lack of exercise, genetics[2][7] |
| Diagnostic method | High blood sugar[2] |
| Treatment | Healthy diet, physical exercise[2] |
| Medication | Insulin, anti-diabetic medication like metformin[2][8][9] |
| Frequency | 463 million (8.8%)[10] |
| Deaths | 4.2 million (2019)[10] |
Diabetes is due to either the pancreas not producing enough insulin, or the cells of the body not responding properly to the insulin produced.[12] There are three main types of diabetes mellitus:[2]
- Type 1 diabetes results from the pancreas's failure to produce enough insulin due to loss of beta cells.[2] This form was previously referred to as "insulin-dependent diabetes mellitus" (IDDM) or "juvenile diabetes".[2] The loss of beta cells is caused by an autoimmune response.[13] The cause of this autoimmune response is unknown.[2]
- Type 2 diabetes begins with insulin resistance, a condition in which cells fail to respond to insulin properly.[2] As the disease progresses, a lack of insulin may also develop.[14] This form was previously referred to as "non insulin-dependent diabetes mellitus" (NIDDM) or "adult-onset diabetes".[2] The most common cause is a combination of excessive body weight and insufficient exercise.[2]
- Gestational diabetes is the third main form, and occurs when pregnant women without a previous history of diabetes develop high blood sugar levels.[2]
Type 1 diabetes must be managed with insulin injections.[2] Prevention and treatment of type 2 diabetes involves maintaining a healthy diet, regular physical exercise, a normal body weight, and avoiding use of tobacco.[2] Type 2 diabetes may be treated with medications such as insulin sensitizers with or without insulin.[15] Control of blood pressure and maintaining proper foot and eye care are important for people with the disease.[2] Insulin and some oral medications can cause low blood sugar.[16] Weight loss surgery in those with obesity is sometimes an effective measure in those with type 2 diabetes.[17] Gestational diabetes usually resolves after the birth of the baby.[18]
As of 2019, an estimated 463 million people had diabetes worldwide (8.8% of the adult population), with type 2 diabetes making up about 90% of the cases.[10] Rates are similar in women and men.[19] Trends suggest that rates will continue to rise.[10] Diabetes at least doubles a person's risk of early death.[2] In 2019, diabetes resulted in approximately 4.2 million deaths.[10] It is the 7th leading cause of death globally.[20][21] The global economic cost of diabetes related health expenditure in 2017 was estimated at US$727 billion.[10] In the United States, diabetes cost nearly US$327 billion in 2017.[22] Average medical expenditures among people with diabetes are about 2.3 times higher.[23]
Signs and symptoms
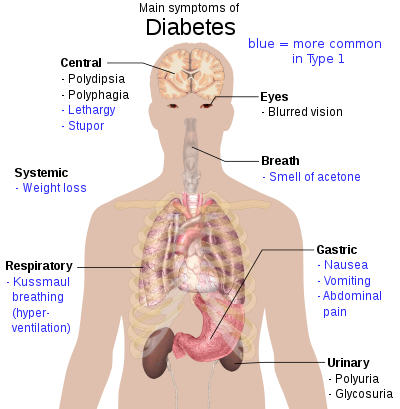
The classic symptoms of untreated diabetes are unintended weight loss, polyuria (increased urination), polydipsia (increased thirst), and polyphagia (increased hunger).[24] Symptoms may develop rapidly (weeks or months) in type 1 diabetes, while they usually develop much more slowly and may be subtle or absent in type 2 diabetes.[25]
Several other signs and symptoms can mark the onset of diabetes although they are not specific to the disease. In addition to the known ones above, they include blurred vision, headache, fatigue, slow healing of cuts, and itchy skin. Prolonged high blood glucose can cause glucose absorption in the lens of the eye, which leads to changes in its shape, resulting in vision changes. Long-term vision loss can also be caused by diabetic retinopathy. A number of skin rashes that can occur in diabetes are collectively known as diabetic dermadromes.[26]
Diabetic emergencies
People (usually with type 1 diabetes) may also experience episodes of diabetic ketoacidosis (DKA), a metabolic disturbance characterized by nausea, vomiting and abdominal pain, the smell of acetone on the breath, deep breathing known as Kussmaul breathing, and in severe cases a decreased level of consciousness.[27] A rare but equally severe possibility is hyperosmolar hyperglycemic state (HHS), which is more common in type 2 diabetes and is mainly the result of dehydration.[27]
Treatment-related low blood sugar (hypoglycemia) is common in people with type 1 and also type 2 diabetes depending on the medication being used. Most cases are mild and are not considered medical emergencies. Effects can range from feelings of unease, sweating, trembling, and increased appetite in mild cases to more serious effects such as confusion, changes in behavior such as aggressiveness, seizures, unconsciousness, and (rarely) permanent brain damage or death in severe cases.[28][29] Rapid breathing, sweating, and cold, pale skin are characteristic of low blood sugar but not definitive.[30] Mild to moderate cases are self-treated by eating or drinking something high in sugar. Severe cases can lead to unconsciousness and must be treated with intravenous glucose or injections with glucagon.[31]
Complications
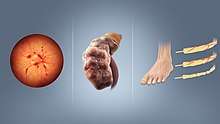
All forms of diabetes increase the risk of long-term complications. These typically develop after many years (10–20) but may be the first symptom in those who have otherwise not received a diagnosis before that time.
The major long-term complications relate to damage to blood vessels. Diabetes doubles the risk of cardiovascular disease[32] and about 75% of deaths in people with diabetes are due to coronary artery disease.[33] Other macrovascular diseases include stroke, and peripheral artery disease.
The primary complications of diabetes due to damage in small blood vessels include damage to the eyes, kidneys, and nerves.[34] Damage to the eyes, known as diabetic retinopathy, is caused by damage to the blood vessels in the retina of the eye, and can result in gradual vision loss and eventual blindness.[34] Diabetes also increases the risk of having glaucoma, cataracts, and other eye problems. It is recommended that people with diabetes visit an eye doctor once a year.[35] Damage to the kidneys, known as diabetic nephropathy, can lead to tissue scarring, urine protein loss, and eventually chronic kidney disease, sometimes requiring dialysis or kidney transplantation.[34] Damage to the nerves of the body, known as diabetic neuropathy, is the most common complication of diabetes.[34] The symptoms can include numbness, tingling, pain, and altered pain sensation, which can lead to damage to the skin. Diabetes-related foot problems (such as diabetic foot ulcers) may occur, and can be difficult to treat, occasionally requiring amputation. Additionally, proximal diabetic neuropathy causes painful muscle atrophy and weakness.
There is a link between cognitive deficit and diabetes. Compared to those without diabetes, those with the disease have a 1.2 to 1.5-fold greater rate of decline in cognitive function.[36] Having diabetes, especially when on insulin, increases the risk of falls in older people.[37]
Causes
| Feature | Type 1 diabetes | Type 2 diabetes |
|---|---|---|
| Onset | Sudden | Gradual |
| Age at onset | Mostly in children | Mostly in adults |
| Body size | Thin or normal[39] | Often obese |
| Ketoacidosis | Common | Rare |
| Autoantibodies | Usually present | Absent |
| Endogenous insulin | Low or absent | Normal, decreased or increased |
| Concordance in identical twins |
50% | 90% |
| Prevalence | ~10% | ~90% |
Diabetes mellitus is classified into four broad categories: type 1 diabetes, type 2 diabetes, gestational diabetes, and "other specific types".[12] The "other specific types" are a collection of a few dozen individual causes.[12] Diabetes is a more variable disease than once thought and people may have combinations of forms.[40] The term "diabetes", without qualification, refers to diabetes mellitus.[41]
Type 1
Type 1 diabetes is characterized by loss of the insulin-producing beta cells of the pancreatic islets, leading to insulin deficiency. This type can be further classified as immune-mediated or idiopathic. The majority of type 1 diabetes is of the immune-mediated nature, in which a T cell-mediated autoimmune attack leads to the loss of beta cells and thus insulin.[42] It causes approximately 10% of diabetes mellitus cases in North America and Europe. Most affected people are otherwise healthy and of a healthy weight when onset occurs. Sensitivity and responsiveness to insulin are usually normal, especially in the early stages. Although it has been called "juvenile diabetes" due to the frequent onset in children, the majority of individuals living with type 1 diabetes are now adults.[6]
"Brittle" diabetes, also known as unstable diabetes or labile diabetes, is a term that was traditionally used to describe the dramatic and recurrent swings in glucose levels, often occurring for no apparent reason in insulin-dependent diabetes. This term, however, has no biologic basis and should not be used.[43] Still, type 1 diabetes can be accompanied by irregular and unpredictable high blood sugar levels, and the potential for diabetic ketoacidosis or serious low blood sugar levels. Other complications include an impaired counterregulatory response to low blood sugar, infection, gastroparesis (which leads to erratic absorption of dietary carbohydrates), and endocrinopathies (e.g., Addison's disease).[43] These phenomena are believed to occur no more frequently than in 1% to 2% of persons with type 1 diabetes.[44]

Type 1 diabetes is partly inherited, with multiple genes, including certain HLA genotypes, known to influence the risk of diabetes. In genetically susceptible people, the onset of diabetes can be triggered by one or more environmental factors,[45] such as a viral infection or diet. Several viruses have been implicated, but to date there is no stringent evidence to support this hypothesis in humans.[45][46] Among dietary factors, data suggest that gliadin (a protein present in gluten) may play a role in the development of type 1 diabetes, but the mechanism is not fully understood.[47][48]
Type 1 diabetes can occur at any age, and a significant proportion is diagnosed during adulthood. Latent autoimmune diabetes of adults (LADA) is the diagnostic term applied when type 1 diabetes develops in adults; it has a slower onset than the same condition in children. Given this difference, some use the unofficial term "type 1.5 diabetes" for this condition. Adults with LADA are frequently initially misdiagnosed as having type 2 diabetes, based on age rather than cause[49]
Type 2

Type 2 diabetes is characterized by insulin resistance, which may be combined with relatively reduced insulin secretion.[12] The defective responsiveness of body tissues to insulin is believed to involve the insulin receptor. However, the specific defects are not known. Diabetes mellitus cases due to a known defect are classified separately. Type 2 diabetes is the most common type of diabetes mellitus.[2] Many people with type 2 diabetes have evidence of prediabetes (impaired fasting glucose and/or impaired glucose tolerance) before meeting the criteria for type 2 diabetes.[50] The progression of prediabetes to overt type 2 diabetes can be slowed or reversed by lifestyle changes or medications that improve insulin sensitivity or reduce the liver's glucose production.[51]
Type 2 diabetes is primarily due to lifestyle factors and genetics.[52] A number of lifestyle factors are known to be important to the development of type 2 diabetes, including obesity (defined by a body mass index of greater than 30), lack of physical activity, poor diet, stress, and urbanization.[38] Excess body fat is associated with 30% of cases in people of Chinese and Japanese descent, 60–80% of cases in those of European and African descent, and 100% of Pima Indians and Pacific Islanders.[12] Even those who are not obese may have a high waist–hip ratio.[12]
Dietary factors such as sugar-sweetened drinks is associated with an increased risk.[53][54] The type of fats in the diet is also important, with saturated fat and trans fats increasing the risk and polyunsaturated and monounsaturated fat decreasing the risk.[52] Eating white rice excessively may increase the risk of diabetes, especially in Chinese and Japanese people.[55] Lack of physical activity may increase the risk of diabetes in some people.[56]
Adverse childhood experiences (ACEs), including abuse, neglect, and household difficulties, increase the likelihood of type 2 diabetes later in life by 32%, with neglect having the strongest effect.[57]
Gestational diabetes
Gestational diabetes resembles type 2 diabetes in several respects, involving a combination of relatively inadequate insulin secretion and responsiveness. It occurs in about 2–10% of all pregnancies and may improve or disappear after delivery.[58] It is recommended that all pregnant women get tested starting around 24–28 weeks gestation.[59] It is most often diagnosed in the second or third trimester because of the increase in insulin-antagonist hormone levels that occurs at this time.[59] However, after pregnancy approximately 5–10% of women with gestational diabetes are found to have another form of diabetes, most commonly type 2.[58] Gestational diabetes is fully treatable, but requires careful medical supervision throughout the pregnancy. Management may include dietary changes, blood glucose monitoring, and in some cases, insulin may be required[60]
Though it may be transient, untreated gestational diabetes can damage the health of the fetus or mother. Risks to the baby include macrosomia (high birth weight), congenital heart and central nervous system abnormalities, and skeletal muscle malformations. Increased levels of insulin in a fetus's blood may inhibit fetal surfactant production and cause infant respiratory distress syndrome. A high blood bilirubin level may result from red blood cell destruction. In severe cases, perinatal death may occur, most commonly as a result of poor placental perfusion due to vascular impairment. Labor induction may be indicated with decreased placental function. A caesarean section may be performed if there is marked fetal distress or an increased risk of injury associated with macrosomia, such as shoulder dystocia.[61]
Other types
Maturity onset diabetes of the young (MODY) is a rare autosomal dominant inherited form of diabetes, due to one of several single-gene mutations causing defects in insulin production.[62] It is significantly less common than the three main types, constituting 1-2% of all cases. The name of this disease refers to early hypotheses as to its nature. Being due to a defective gene, this disease varies in age at presentation and in severity according to the specific gene defect; thus there are at least 13 subtypes of MODY. People with MODY often can control it without using insulin.[63]
Some cases of diabetes are caused by the body's tissue receptors not responding to insulin (even when insulin levels are normal, which is what separates it from type 2 diabetes); this form is very uncommon. Genetic mutations (autosomal or mitochondrial) can lead to defects in beta cell function. Abnormal insulin action may also have been genetically determined in some cases. Any disease that causes extensive damage to the pancreas may lead to diabetes (for example, chronic pancreatitis and cystic fibrosis). Diseases associated with excessive secretion of insulin-antagonistic hormones can cause diabetes (which is typically resolved once the hormone excess is removed). Many drugs impair insulin secretion and some toxins damage pancreatic beta cells, whereas others increase insulin resistance (especially glucocorticoids which can provoke "steroid diabetes"). The ICD-10 (1992) diagnostic entity, malnutrition-related diabetes mellitus (MRDM or MMDM, ICD-10 code E12), was deprecated by the World Health Organization (WHO) when the current taxonomy was introduced in 1999.[64]
The following is a list of disorders that may increase the risk of diabetes:[65]
|
|
Pathophysiology

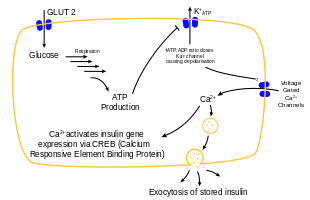
Insulin is the principal hormone that regulates the uptake of glucose from the blood into most cells of the body, especially liver, adipose tissue and muscle, except smooth muscle, in which insulin acts via the IGF-1. Therefore, deficiency of insulin or the insensitivity of its receptors play a central role in all forms of diabetes mellitus.[67]
The body obtains glucose from three main sources: the intestinal absorption of food; the breakdown of glycogen (glycogenolysis), the storage form of glucose found in the liver; and gluconeogenesis, the generation of glucose from non-carbohydrate substrates in the body.[68] Insulin plays a critical role in regulating glucose levels in the body. Insulin can inhibit the breakdown of glycogen or the process of gluconeogenesis, it can stimulate the transport of glucose into fat and muscle cells, and it can stimulate the storage of glucose in the form of glycogen.[68]
Insulin is released into the blood by beta cells (β-cells), found in the islets of Langerhans in the pancreas, in response to rising levels of blood glucose, typically after eating. Insulin is used by about two-thirds of the body's cells to absorb glucose from the blood for use as fuel, for conversion to other needed molecules, or for storage. Lower glucose levels result in decreased insulin release from the beta cells and in the breakdown of glycogen to glucose. This process is mainly controlled by the hormone glucagon, which acts in the opposite manner to insulin.[69]
If the amount of insulin available is insufficient, or if cells respond poorly to the effects of insulin (insulin resistance), or if the insulin itself is defective, then glucose is not absorbed properly by the body cells that require it, and is not stored appropriately in the liver and muscles. The net effect is persistently high levels of blood glucose, poor protein synthesis, and other metabolic derangements, such as metabolic acidosis in cases of complete insulin deficiency.[68]
When glucose concentration in the blood remains high over time, the kidneys reach a threshold of reabsorption, and the body excretes glucose in the urine (glycosuria).[70] This increases the osmotic pressure of the urine and inhibits reabsorption of water by the kidney, resulting in increased urine production (polyuria) and increased fluid loss. Lost blood volume is replaced osmotically from water in body cells and other body compartments, causing dehydration and increased thirst (polydipsia).[68] In addition, intracellular glucose deficiency stimulates appetite leading to excessive food intake (polyphagia).[71]
Diagnosis
| Condition | 2-hour glucose | Fasting glucose | HbA1c | |||
|---|---|---|---|---|---|---|
| Unit | mmol/L | mg/dL | mmol/L | mg/dL | mmol/mol | DCCT % |
| Normal | < 7.8 | < 140 | < 6.1 | < 110 | < 42 | < 6.0 |
| Impaired fasting glycaemia | < 7.8 | < 140 | 6.1–7.0 | 110–126 | 42–46 | 6.0–6.4 |
| Impaired glucose tolerance | ≥ 7.8 | ≥ 140 | < 7.0 | < 126 | 42–46 | 6.0–6.4 |
| Diabetes mellitus | ≥ 11.1 | ≥ 200 | ≥ 7.0 | ≥ 126 | ≥ 48 | ≥ 6.5 |
Diabetes mellitus is characterized by recurrent or persistent high blood sugar, and is diagnosed by demonstrating any one of the following:[64]
- Fasting plasma glucose level ≥ 7.0 mmol/L (126 mg/dL)
- Plasma glucose ≥ 11.1 mmol/L (200 mg/dL) two hours after a 75 gram oral glucose load as in a glucose tolerance test (OGTT)
- Symptoms of high blood sugar and casual plasma glucose ≥ 11.1 mmol/L (200 mg/dL)
- Glycated hemoglobin (HbA1C) ≥ 48 mmol/mol (≥ 6.5 DCCT %).[74]
A positive result, in the absence of unequivocal high blood sugar, should be confirmed by a repeat of any of the above methods on a different day. It is preferable to measure a fasting glucose level because of the ease of measurement and the considerable time commitment of formal glucose tolerance testing, which takes two hours to complete and offers no prognostic advantage over the fasting test.[75] According to the current definition, two fasting glucose measurements above 7.0 mmol/L (126 mg/dL) is considered diagnostic for diabetes mellitus.
Per the WHO, people with fasting glucose levels from 6.1 to 6.9 mmol/L (110 to 125 mg/dL) are considered to have impaired fasting glucose.[76] People with plasma glucose at or above 7.8 mmol/L (140 mg/dL), but not over 11.1 mmol/L (200 mg/dL), two hours after a 75 gram oral glucose load are considered to have impaired glucose tolerance. Of these two prediabetic states, the latter in particular is a major risk factor for progression to full-blown diabetes mellitus, as well as cardiovascular disease.[77] The American Diabetes Association (ADA) since 2003 uses a slightly different range for impaired fasting glucose of 5.6 to 6.9 mmol/L (100 to 125 mg/dL).[78]
Glycated hemoglobin is better than fasting glucose for determining risks of cardiovascular disease and death from any cause.[79]
Prevention
There is no known preventive measure for type 1 diabetes.[2] Type 2 diabetes—which accounts for 85–90% of all cases worldwide—can often be prevented or delayed by maintaining a normal body weight, engaging in physical activity, and eating a healthy diet.[2] Higher levels of physical activity (more than 90 minutes per day) reduce the risk of diabetes by 28%.[80] Dietary changes known to be effective in helping to prevent diabetes include maintaining a diet rich in whole grains and fiber, and choosing good fats, such as the polyunsaturated fats found in nuts, vegetable oils, and fish.[81] Limiting sugary beverages and eating less red meat and other sources of saturated fat can also help prevent diabetes.[81] Tobacco smoking is also associated with an increased risk of diabetes and its complications, so smoking cessation can be an important preventive measure as well.[82]
The relationship between type 2 diabetes and the main modifiable risk factors (excess weight, unhealthy diet, physical inactivity and tobacco use) is similar in all regions of the world. There is growing evidence that the underlying determinants of diabetes are a reflection of the major forces driving social, economic and cultural change: globalization, urbanization, population aging, and the general health policy environment.[83]
Management
Diabetes management concentrates on keeping blood sugar levels as close to normal, without causing low blood sugar. This can usually be accomplished with dietary changes, exercise, weight loss, and use of appropriate medications (insulin, oral medications).
Learning about the disease and actively participating in the treatment is important, since complications are far less common and less severe in people who have well-managed blood sugar levels.[84][85] Per the American College of Physicians, the goal of treatment is an HbA1C level of 7-8%.[86] Attention is also paid to other health problems that may accelerate the negative effects of diabetes. These include smoking, high blood pressure, metabolic syndrome obesity, and lack of regular exercise.[87] Specialized footwear is widely used to reduce the risk of ulcers in at-risk diabetic feet although evidence for the efficacy of this remains equivocal.[88]
Lifestyle
People with diabetes can benefit from education about the disease and treatment, dietary changes, and exercise, with the goal of keeping both short-term and long-term blood glucose levels within acceptable bounds. In addition, given the associated higher risks of cardiovascular disease, lifestyle modifications are recommended to control blood pressure.[89][90]
Weight loss can prevent progression from prediabetes to diabetes type 2, decrease the risk of cardiovascular disease, or result in a partial remission in people with diabetes.[91][92] No single dietary pattern is best for all people with diabetes.[93] Healthy dietary patterns, such as the Mediterranean diet, low-carbohydrate diet, or DASH diet are often recommended, although evidence does not support one over the others.[91][92] According to the ADA, "reducing overall carbohydrate intake for individuals with diabetes has demonstrated the most evidence for improving glycemia", and for individuals with type 2 diabetes who can not meet the glycemic targets or where reducing anti-glycemic medications is a priority, low or very-low carbohydrate diets are a viable approach.[92] For overweight people with type 2 diabetes, any diet that achieves weight loss is effective.[93][94]
Medications
Glucose control
Most medications used to treat diabetes act by lowering blood sugar levels through different mechanisms. There is broad consensus that when people with diabetes maintain tight glucose control – keeping the glucose levels in their blood within normal ranges – they experience fewer complications, such as kidney problems or eye problems.[95][96] There is however debate as to whether this is appropriate and cost effective for people later in life in whom the risk of hypoglycemia may be more significant.[97]
There are a number of different classes of anti-diabetic medications. Type 1 diabetes requires treatment with insulin, typically with a combination of regular and NPH insulin, or synthetic insulin analogs. Type 2 diabetes may also be treated with insulin at later stages. Some medications for type 2 diabetes are taken by mouth, such as metformin, while others are only administered by injection, such as GLP-1 agonists.
Metformin is generally recommended as a first-line treatment for type 2 diabetes, as there is good evidence that it decreases mortality.[8] It works by decreasing the liver's production of glucose.[98] Several other groups of drugs, mostly given by mouth, may also decrease blood sugar in type 2 diabetes. These include agents that increase insulin release (sulfonylureas), agents that decrease absorption of sugar from the intestines (acarbose), agents that inhibit the enzyme dipeptidyl peptidase-4 (DPP-4) that inactivates incretins such as GLP-1 and GIP (sitagliptin), agents that make the body more sensitive to insulin (thiazolidinedione) and agents that increase the excretion of glucose in the urine (SGLT2 inhibitors).[98] When insulin is used in type 2 diabetes, a long-acting formulation is usually added initially, while continuing oral medications.[8] Doses of insulin are then increased until glucose targets are reached.[8][99]
Blood pressure lowering
Cardiovascular disease is a serious complication associated with diabetes, and many international guidelines recommend blood pressure treatment targets that are lower than 140/90 mmHg for people with diabetes.[100] However, there is only limited evidence regarding what the lower targets should be. A 2016 systematic review found potential harm to treating to targets lower than 140 mmHg,[101] and a subsequent systematic review in 2019 found no evidence of additional benefit from blood pressure lowering to between 130 - 140mmHg, although there was an increased risk of adverse events.[102]
2015 American Diabetes Association recommendations are that people with diabetes and albuminuria should receive an inhibitor of the renin-angiotensin system to reduce the risks of progression to end-stage renal disease, cardiovascular events, and death.[103] There is some evidence that angiotensin converting enzyme inhibitors (ACEIs) are superior to other inhibitors of the renin-angiotensin system such as angiotensin receptor blockers (ARBs),[104] or aliskiren in preventing cardiovascular disease.[105] Although a more recent review found similar effects of ACEIs and ARBs on major cardiovascular and renal outcomes.[106] There is no evidence that combining ACEIs and ARBs provides additional benefits.[106]
Aspirin
The use of aspirin to prevent cardiovascular disease in diabetes is controversial.[103] Aspirin is recommended in people at high risk of cardiovascular disease, however routine use of aspirin has not been found to improve outcomes in uncomplicated diabetes.[107] 2015 American Diabetes Association recommendations for aspirin use (based on expert consensus or clinical experience) are that low-dose aspirin use is reasonable in adults with diabetes who are at intermediate risk of cardiovascular disease (10-year cardiovascular disease risk, 5–10%).[103]
Surgery
Weight loss surgery in those with obesity and type 2 diabetes is often an effective measure.[17] Many are able to maintain normal blood sugar levels with little or no medications following surgery[108] and long-term mortality is decreased.[109] There is, however, a short-term mortality risk of less than 1% from the surgery.[110] The body mass index cutoffs for when surgery is appropriate are not yet clear.[109] It is recommended that this option be considered in those who are unable to get both their weight and blood sugar under control.[111]
A pancreas transplant is occasionally considered for people with type 1 diabetes who have severe complications of their disease, including end stage kidney disease requiring kidney transplantation.[112]
Support
In countries using a general practitioner system, such as the United Kingdom, care may take place mainly outside hospitals, with hospital-based specialist care used only in case of complications, difficult blood sugar control, or research projects. In other circumstances, general practitioners and specialists share care in a team approach. Home telehealth support can be an effective management technique.[113]
Epidemiology
_Gradient_Map.png)
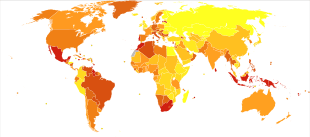
In 2017, 425 million people had diabetes worldwide,[114] up from an estimated 382 million people in 2013[115] and from 108 million in 1980.[116] Accounting for the shifting age structure of the global population, the prevalence of diabetes is 8.8% among adults, nearly double the rate of 4.7% in 1980.[114][116] Type 2 makes up about 90% of the cases.[38][19] Some data indicate rates are roughly equal in women and men,[19] but male excess in diabetes has been found in many populations with higher type 2 incidence, possibly due to sex-related differences in insulin sensitivity, consequences of obesity and regional body fat deposition, and other contributing factors such as high blood pressure, tobacco smoking, and alcohol intake.[117][118]
The WHO estimates that diabetes resulted in 1.5 million deaths in 2012, making it the 8th leading cause of death.[15][116] However another 2.2 million deaths worldwide were attributable to high blood glucose and the increased risks of cardiovascular disease and other associated complications (e.g. kidney failure), which often lead to premature death and are often listed as the underlying cause on death certificates rather than diabetes.[116][119] For example, in 2017, the International Diabetes Federation (IDF) estimated that diabetes resulted in 4.0 million deaths worldwide,[114] using modeling to estimate the total number of deaths that could be directly or indirectly attributed to diabetes.[114]
Diabetes occurs throughout the world but is more common (especially type 2) in more developed countries. The greatest increase in rates has however been seen in low- and middle-income countries,[116] where more than 80% of diabetic deaths occur.[120] The fastest prevalence increase is expected to occur in Asia and Africa, where most people with diabetes will probably live in 2030.[121] The increase in rates in developing countries follows the trend of urbanization and lifestyle changes, including increasingly sedentary lifestyles, less physically demanding work and the global nutrition transition, marked by increased intake of foods that are high energy-dense but nutrient-poor (often high in sugar and saturated fats, sometimes referred to as the "Western-style" diet).[116][121] The global number of diabetes cases might increase by 48% between 2017 and 2045.[114]
History
Diabetes was one of the first diseases described,[122] with an Egyptian manuscript from c. 1500 BCE mentioning "too great emptying of the urine."[123] The Ebers papyrus includes a recommendation for a drink to take in such cases.[124] The first described cases are believed to have been type 1 diabetes.[123] Indian physicians around the same time identified the disease and classified it as madhumeha or "honey urine", noting the urine would attract ants.[123][124]
The term "diabetes" or "to pass through" was first used in 230 BCE by the Greek Apollonius of Memphis.[123] The disease was considered rare during the time of the Roman empire, with Galen commenting he had only seen two cases during his career.[123] This is possibly due to the diet and lifestyle of the ancients, or because the clinical symptoms were observed during the advanced stage of the disease. Galen named the disease "diarrhea of the urine" (diarrhea urinosa).[125]
The earliest surviving work with a detailed reference to diabetes is that of Aretaeus of Cappadocia (2nd or early 3rd century CE). He described the symptoms and the course of the disease, which he attributed to the moisture and coldness, reflecting the beliefs of the "Pneumatic School". He hypothesized a correlation between diabetes and other diseases, and he discussed differential diagnosis from the snakebite, which also provokes excessive thirst. His work remained unknown in the West until 1552, when the first Latin edition was published in Venice.[125]
Two types of diabetes were identified as separate conditions for the first time by the Indian physicians Sushruta and Charaka in 400–500 CE with one type being associated with youth and another type with being overweight.[123] The term "mellitus" or "from honey" was added by the Briton John Rolle in the late 1700s to separate the condition from diabetes insipidus, which is also associated with frequent urination.[123] Effective treatment was not developed until the early part of the 20th century when Canadians Frederick Banting and Charles Herbert Best isolated and purified insulin in 1921 and 1922.[123] This was followed by the development of the long-acting insulin NPH in the 1940s.[123]
Etymology
The word diabetes (/ˌdaɪ.əˈbiːtiːz/ or /ˌdaɪ.əˈbiːtɪs/) comes from Latin diabētēs, which in turn comes from Ancient Greek διαβήτης (diabētēs), which literally means "a passer through; a siphon".[126] Ancient Greek physician Aretaeus of Cappadocia (fl. 1st century CE) used that word, with the intended meaning "excessive discharge of urine", as the name for the disease.[127][128] Ultimately, the word comes from Greek διαβαίνειν (diabainein), meaning "to pass through,"[126] which is composed of δια- (dia-), meaning "through" and βαίνειν (bainein), meaning "to go".[127] The word "diabetes" is first recorded in English, in the form diabete, in a medical text written around 1425.
The word mellitus (/məˈlaɪtəs/ or /ˈmɛlɪtəs/) comes from the classical Latin word mellītus, meaning "mellite"[129] (i.e. sweetened with honey;[129] honey-sweet[130]). The Latin word comes from mell-, which comes from mel, meaning "honey";[129][130] sweetness;[130] pleasant thing,[130] and the suffix -ītus,[129] whose meaning is the same as that of the English suffix "-ite".[131] It was Thomas Willis who in 1675 added "mellitus" to the word "diabetes" as a designation for the disease, when he noticed the urine of a person with diabetes had a sweet taste (glycosuria). This sweet taste had been noticed in urine by the ancient Greeks, Chinese, Egyptians, Indians, and Persians.
Society and culture
The 1989 "St. Vincent Declaration"[132][133] was the result of international efforts to improve the care accorded to those with diabetes. Doing so is important not only in terms of quality of life and life expectancy but also economically – expenses due to diabetes have been shown to be a major drain on health – and productivity-related resources for healthcare systems and governments.
Several countries established more and less successful national diabetes programmes to improve treatment of the disease.[134]
People with diabetes who have neuropathic symptoms such as numbness or tingling in feet or hands are twice as likely to be unemployed as those without the symptoms.[135]
In 2010, diabetes-related emergency room (ER) visit rates in the United States were higher among people from the lowest income communities (526 per 10,000 population) than from the highest income communities (236 per 10,000 population). Approximately 9.4% of diabetes-related ER visits were for the uninsured.[136]
Naming
The term "type 1 diabetes" has replaced several former terms, including childhood-onset diabetes, juvenile diabetes, and insulin-dependent diabetes mellitus (IDDM). Likewise, the term "type 2 diabetes" has replaced several former terms, including adult-onset diabetes, obesity-related diabetes, and noninsulin-dependent diabetes mellitus (NIDDM). Beyond these two types, there is no agreed-upon standard nomenclature.
Diabetes mellitus is also occasionally known as "sugar diabetes" to differentiate it from diabetes insipidus.[137]
Other animals
In animals, diabetes is most commonly encountered in dogs and cats. Middle-aged animals are most commonly affected. Female dogs are twice as likely to be affected as males, while according to some sources, male cats are more prone than females. In both species, all breeds may be affected, but some small dog breeds are particularly likely to develop diabetes, such as Miniature Poodles.[138]
Feline diabetes is strikingly similar to human type 2 diabetes. The Burmese breed, along with the Russian Blue, Abyssinian, and Norwegian Forest cat breeds, showed an increased risk of DM than other breeds. There is an association between overweight and an increased risk of feline diabetes.[139]
The symptoms may relate to fluid loss and polyuria, but the course may also be insidious. Diabetic animals are more prone to infections. The long-term complications recognized in humans are much rarer in animals. The principles of treatment (weight loss, oral antidiabetics, subcutaneous insulin) and management of emergencies (e.g. ketoacidosis) are similar to those in humans.[138]
Research
Inhalable insulin has been developed. The original products were withdrawn due to side effects. Afrezza, under development by the pharmaceuticals company MannKind Corporation, was approved by the United States Food and Drug Administration (FDA) for general sale in June 2014.[140] An advantage to inhaled insulin is that it may be more convenient and easy to use.[141]
Transdermal insulin in the form of a cream has been developed and trials are being conducted on people with type 2 diabetes.[142][143]
Major clinical trials
The Diabetes Control and Complications Trial (DCCT) was a clinical study conducted by the United States National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) that was published in the New England Journal of Medicine in 1993. Test subjects all had type 1 diabetes and were randomized to a tight glycemic arm and a control arm with the standard of care at the time; people were followed for an average of seven years, and people in the treatment had dramatically lower rates of diabetic complications. It was as a landmark study at the time, and significantly changed the management of all forms of diabetes.[97][144][145]
The United Kingdom Prospective Diabetes Study (UKPDS) was a clinical study conducted by Z that was published in The Lancet in 1998. Around 3,800 people with type 2 diabetes were followed for an average of ten years, and were treated with tight glucose control or the standard of care, and again the treatment arm had far better outcomes. This confirmed the importance of tight glucose control, as well as blood pressure control, for people with this condition.[97][146][147]
References
- "Diabetes Blue Circle Symbol". International Diabetes Federation. 17 March 2006. Archived from the original on 5 August 2007.
- "Diabetes Fact sheet N°312". WHO. October 2013. Archived from the original on 26 August 2013. Retrieved 25 March 2014.
- Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN (July 2009). "Hyperglycemic crises in adult patients with diabetes". Diabetes Care. 32 (7): 1335–43. doi:10.2337/dc09-9032. PMC 2699725. PMID 19564476.
- Krishnasamy S, Abell TL (July 2018). "Diabetic Gastroparesis: Principles and Current Trends in Management". Diabetes Therapy. 9 (Suppl 1): 1–42. doi:10.1007/s13300-018-0454-9. PMC 6028327. PMID 29934758.
- Saedi, E; Gheini, MR; Faiz, F; Arami, MA (15 September 2016). "Diabetes mellitus and cognitive impairments". World Journal of Diabetes. 7 (17): 412–22. doi:10.4239/wjd.v7.i17.412. PMC 5027005. PMID 27660698.
- Chiang JL, Kirkman MS, Laffel LM, Peters AL (July 2014). "Type 1 diabetes through the life span: a position statement of the American Diabetes Association". Diabetes Care. 37 (7): 2034–54. doi:10.2337/dc14-1140. PMC 5865481. PMID 24935775.
- "Causes of Diabetes". National Institute of Diabetes and Digestive and Kidney Diseases. June 2014. Archived from the original on 2 February 2016. Retrieved 10 February 2016.
- Ripsin, CM; Kang, H; Urban, RJ (January 2009). "Management of blood glucose in type 2 diabetes mellitus" (PDF). American Family Physician. 79 (1): 29–36. PMID 19145963. Archived (PDF) from the original on 2013-05-05.
- Brutsaert, Erika F. (February 2017). "Drug Treatment of Diabetes Mellitus". Retrieved 12 October 2018.
- "IDF DIABETES ATLAS Ninth Edition 2019" (PDF). www.diabetesatlas.org. Retrieved 18 May 2020.
- "About diabetes". World Health Organization. Archived from the original on 31 March 2014. Retrieved 4 April 2014.
- Shoback DG, Gardner D, eds. (2011). "Chapter 17". Greenspan's basic & clinical endocrinology (9th ed.). New York: McGraw-Hill Medical. ISBN 978-0-07-162243-1.
- Norman A, Henry H (2015). Hormones. Elsevier. pp. 136–137. ISBN 9780123694447.
- RSSDI textbook of diabetes mellitus (Revised 2nd ed.). Jaypee Brothers Medical Publishers. 2012. p. 235. ISBN 978-93-5025-489-9. Archived from the original on 14 October 2015.
- "The top 10 causes of death Fact sheet N°310". World Health Organization. October 2013. Archived from the original on 30 May 2017.
- Rippe RS, Irwin JM, eds. (2010). Manual of intensive care medicine (5th ed.). Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 549. ISBN 978-0-7817-9992-8.
- Picot J, Jones J, Colquitt JL, Gospodarevskaya E, Loveman E, Baxter L, Clegg AJ (September 2009). "The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: a systematic review and economic evaluation". Health Technology Assessment. 13 (41): 1–190, 215–357, iii–iv. doi:10.3310/hta13410. hdl:10536/DRO/DU:30064294. PMID 19726018.
- Cash, Jill (2014). Family Practice Guidelines (3rd ed.). Springer. p. 396. ISBN 978-0-8261-6875-7. Archived from the original on 31 October 2015.
- Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. (December 2012). "Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2163–96. doi:10.1016/S0140-6736(12)61729-2. PMC 6350784. PMID 23245607.
- "What is Diabetes?". Centers for Disease Control and Prevention. 11 March 2020. Retrieved 18 May 2020.
- "The top 10 causes of death". www.who.int. Retrieved 18 May 2020.
- American Diabetes Association (2018-03-22). "Economic Costs of Diabetes in the U.S. in 2017". Diabetes Care. 41 (5): 917–928. doi:10.2337/dci18-0007. ISSN 0149-5992. PMC 5911784. PMID 29567642.
- "Deaths and Cost | Data & Statistics | Diabetes | CDC". cdc.gov. 20 February 2019. Retrieved 2 July 2019.
- Cooke DW, Plotnick L (November 2008). "Type 1 diabetes mellitus in pediatrics". Pediatrics in Review. 29 (11): 374–84, quiz 385. doi:10.1542/pir.29-11-374. PMID 18977856. S2CID 20528207.
- "WHO | Diabetes mellitus". WHO. Retrieved 2019-03-23.
- Rockefeller, J.D. (2015). Diabetes: Symptoms, Causes, Treatment and Prevention. ISBN 978-1-5146-0305-5.
- Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN (July 2009). "Hyperglycemic crises in adult patients with diabetes". Diabetes Care. 32 (7): 1335–43. doi:10.2337/dc09-9032. PMC 2699725. PMID 19564476. Archived from the original on 2016-06-25.
- Kenny C (April 2014). "When hypoglycemia is not obvious: diagnosing and treating under-recognized and undisclosed hypoglycemia". Primary Care Diabetes. 8 (1): 3–11. doi:10.1016/j.pcd.2013.09.002. PMID 24100231.
- Verrotti A, Scaparrotta A, Olivieri C, Chiarelli F (December 2012). "Seizures and type 1 diabetes mellitus: current state of knowledge". European Journal of Endocrinology. 167 (6): 749–58. doi:10.1530/EJE-12-0699. PMID 22956556.
- "Symptoms of Low Blood Sugar". WebMD. Archived from the original on 18 June 2016. Retrieved 29 June 2016.
- "Glucagon–Injection side effects, medical uses, and drug interactions". MedicineNet. Retrieved 2018-02-05.
- Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, Di Angelantonio E, Ingelsson E, Lawlor DA, Selvin E, Stampfer M, Stehouwer CD, Lewington S, Pennells L, Thompson A, Sattar N, White IR, Ray KK, Danesh J (June 2010). "Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies". Lancet. 375 (9733): 2215–22. doi:10.1016/S0140-6736(10)60484-9. PMC 2904878. PMID 20609967.
- O'Gara PT, Kushner FG, Ascheim DD, Casey DE, Chung MK, de Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA, Granger CB, Krumholz HM, Linderbaum JA, Morrow DA, Newby LK, Ornato JP, Ou N, Radford MJ, Tamis-Holland JE, Tommaso CL, Tracy CM, Woo YJ, Zhao DX, Anderson JL, Jacobs AK, Halperin JL, Albert NM, Brindis RG, Creager MA, DeMets D, Guyton RA, Hochman JS, Kovacs RJ, Kushner FG, Ohman EM, Stevenson WG, Yancy CW (January 2013). "2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines". Circulation. 127 (4): e362–425. doi:10.1161/CIR.0b013e3182742cf6. PMID 23247304.
- "Diabetes Programme". World Health Organization. Archived from the original on 26 April 2014. Retrieved 22 April 2014.
- "Diabetes – eye care: MedlinePlus Medical Encyclopedia". medlineplus.gov. Retrieved 2018-03-27.
- Cukierman, T (8 Nov 2005). "Cognitive decline and dementia in diabetes – systematic overview of prospective observational studies". Diabetologia. 48 (12): 2460–69. doi:10.1007/s00125-005-0023-4. PMID 16283246.
- Yang Y, Hu X, Zhang Q, Zou R (November 2016). "Diabetes mellitus and risk of falls in older adults: a systematic review and meta-analysis". Age and Ageing. 45 (6): 761–67. doi:10.1093/ageing/afw140. PMID 27515679.
- Williams textbook of endocrinology (12th ed.). Elsevier/Saunders. 2011. pp. 1371–1435. ISBN 978-1-4377-0324-5.
- Lambert P, Bingley PJ (2002). "What is Type 1 Diabetes?". Medicine. 30: 1–5. doi:10.1383/medc.30.1.1.28264.
- Tuomi T, Santoro N, Caprio S, Cai M, Weng J, Groop L (March 2014). "The many faces of diabetes: a disease with increasing heterogeneity". Lancet. 383 (9922): 1084–94. doi:10.1016/S0140-6736(13)62219-9. PMID 24315621.
- "Definition of Diabetes mellitus". MedicineNet. Retrieved 2019-11-04.
- Rother KI (April 2007). "Diabetes treatment--bridging the divide". The New England Journal of Medicine. 356 (15): 1499–501. doi:10.1056/NEJMp078030. PMC 4152979. PMID 17429082.
- "Diabetes Mellitus (DM): Diabetes Mellitus and Disorders of Carbohydrate Metabolism: Merck Manual Professional". Merck Publishing. April 2010. Archived from the original on 2010-07-28. Retrieved 2010-07-30.
- Dorner M, Pinget M, Brogard JM (May 1977). "[Essential labile diabetes (author's transl)]". MMW, Munchener Medizinische Wochenschrift (in German). 119 (19): 671–74. PMID 406527.
- Petzold A, Solimena M, Knoch KP (October 2015). "Mechanisms of Beta Cell Dysfunction Associated With Viral Infection". Current Diabetes Reports (Review). 15 (10): 73. doi:10.1007/s11892-015-0654-x. PMC 4539350. PMID 26280364.
So far, none of the hypotheses accounting for virus-induced beta cell autoimmunity has been supported by stringent evidence in humans, and the involvement of several mechanisms rather than just one is also plausible.
- Butalia S, Kaplan GG, Khokhar B, Rabi DM (December 2016). "Environmental Risk Factors and Type 1 Diabetes: Past, Present, and Future". Canadian Journal of Diabetes (Review). 40 (6): 586–93. doi:10.1016/j.jcjd.2016.05.002. PMID 27545597.
- Serena G, Camhi S, Sturgeon C, Yan S, Fasano A (August 2015). "The Role of Gluten in Celiac Disease and Type 1 Diabetes". Nutrients. 7 (9): 7143–62. doi:10.3390/nu7095329. PMC 4586524. PMID 26343710.
- Visser J, Rozing J, Sapone A, Lammers K, Fasano A (May 2009). "Tight junctions, intestinal permeability, and autoimmunity: celiac disease and type 1 diabetes paradigms". Annals of the New York Academy of Sciences. 1165 (1): 195–205. Bibcode:2009NYASA1165..195V. doi:10.1111/j.1749-6632.2009.04037.x. PMC 2886850. PMID 19538307.
- Laugesen E, Østergaard JA, Leslie RD (July 2015). "Latent autoimmune diabetes of the adult: current knowledge and uncertainty". Diabetic Medicine. 32 (7): 843–52. doi:10.1111/dme.12700. PMC 4676295. PMID 25601320.
- American Diabetes Association (January 2017). "2. Classification and Diagnosis of Diabetes". Diabetes Care. 40 (Suppl 1): S11–S24. doi:10.2337/dc17-S005. PMID 27979889.
- Carris NW, Magness RR, Labovitz AJ (February 2019). "Prevention of Diabetes Mellitus in Patients With Prediabetes". The American Journal of Cardiology. 123 (3): 507–512. doi:10.1016/j.amjcard.2018.10.032. PMC 6350898. PMID 30528418.
- Risérus U, Willett WC, Hu FB (January 2009). "Dietary fats and prevention of type 2 diabetes". Progress in Lipid Research. 48 (1): 44–51. doi:10.1016/j.plipres.2008.10.002. PMC 2654180. PMID 19032965.
- Malik VS, Popkin BM, Bray GA, Després JP, Hu FB (March 2010). "Sugar-sweetened beverages, obesity, type 2 diabetes mellitus, and cardiovascular disease risk". Circulation. 121 (11): 1356–64. doi:10.1161/CIRCULATIONAHA.109.876185. PMC 2862465. PMID 20308626.
- Malik VS, Popkin BM, Bray GA, Després JP, Willett WC, Hu FB (November 2010). "Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes: a meta-analysis". Diabetes Care. 33 (11): 2477–83. doi:10.2337/dc10-1079. PMC 2963518. PMID 20693348.
- Hu EA, Pan A, Malik V, Sun Q (March 2012). "White rice consumption and risk of type 2 diabetes: meta-analysis and systematic review". BMJ. 344: e1454. doi:10.1136/bmj.e1454. PMC 3307808. PMID 22422870.
- Lee IM, Shiroma EJ, Lobelo F, Puska P, Blair SN, Katzmarzyk PT (July 2012). "Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy". Lancet. 380 (9838): 219–29. doi:10.1016/S0140-6736(12)61031-9. PMC 3645500. PMID 22818936.
- Huang, Hao; Yan, Peipei; Shan, Zhilei; Chen, Sijing; Li, Moying; Luo, Cheng; Gao, Hui; Hao, Liping; Liu, Liegang (2015-11-01). "Adverse childhood experiences and risk of type 2 diabetes: A systematic review and meta-analysis". Metabolism - Clinical and Experimental. 64 (11): 1408–1418. doi:10.1016/j.metabol.2015.08.019. ISSN 0026-0495. PMID 26404480.
- "National Diabetes Clearinghouse (NDIC): National Diabetes Statistics 2011". U.S. Department of Health and Human Services. Archived from the original on 17 April 2014. Retrieved 22 April 2014.
- Soldavini, Jessica (November 2019). "Krause's Food & The Nutrition Care Process". Journal of Nutrition Education and Behavior. 51 (10): 1225. doi:10.1016/j.jneb.2019.06.022. ISSN 1499-4046.
- "Managing & Treating Gestational Diabetes | NIDDK". National Institute of Diabetes and Digestive and Kidney Diseases. Retrieved 2019-05-06.
- National Collaborating Centre for Women's and Children's Health (February 2015). "Intrapartum care". Diabetes in Pregnancy: Management of diabetes and its complications from preconception to the postnatal period. National Institute for Health and Care Excellence (UK).
- "Monogenic Forms of Diabetes". National institute of diabetes and digestive and kidney diseases. US NIH. Archived from the original on 12 March 2017. Retrieved 12 March 2017.
- Thanabalasingham G, Owen KR (October 2011). "Diagnosis and management of maturity onset diabetes of the young (MODY)". BMJ. 343 (oct19 3): d6044. doi:10.1136/bmj.d6044. PMID 22012810.
- "Definition, Diagnosis and Classification of Diabetes Mellitus and its Complications" (PDF). World Health Organization. 1999. Archived (PDF) from the original on 2003-03-08.
- Unless otherwise specified, reference is: Table 20-5 in Mitchell, Richard Sheppard; Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson (2007). Robbins Basic Pathology (8th ed.). Philadelphia: Saunders. ISBN 978-1-4160-2973-1.
- Sattar N, Preiss D, Murray HM, Welsh P, Buckley BM, de Craen AJ, Seshasai SR, McMurray JJ, Freeman DJ, Jukema JW, Macfarlane PW, Packard CJ, Stott DJ, Westendorp RG, Shepherd J, Davis BR, Pressel SL, Marchioli R, Marfisi RM, Maggioni AP, Tavazzi L, Tognoni G, Kjekshus J, Pedersen TR, Cook TJ, Gotto AM, Clearfield MB, Downs JR, Nakamura H, Ohashi Y, Mizuno K, Ray KK, Ford I (February 2010). "Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials". Lancet. 375 (9716): 735–42. doi:10.1016/S0140-6736(09)61965-6. PMID 20167359.
- "Insulin Basics". American Diabetes Association. Archived from the original on 14 February 2014. Retrieved 24 April 2014.
- Shoback DG, Gardner D, eds. (2011). Greenspan's basic & clinical endocrinology (9th ed.). McGraw-Hill Medical. ISBN 978-0-07-162243-1.
- Barrett KE, et al. (2012). Ganong's review of medical physiology (24th ed.). McGraw-Hill Medical. ISBN 978-0-07-178003-2.
- Murray RK, et al. (2012). Harper's illustrated biochemistry (29th ed.). McGraw-Hill Medical. ISBN 978-0-07-176576-3.
- Mogotlane, Sophie (2013). Juta's Complete Textbook of Medical Surgical Nursing. Cape Town: Juta. p. 839.
- Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia: Report of a WHO/IDF consultation (PDF). Geneva: World Health Organization. 2006. p. 21. ISBN 978-92-4-159493-6.
- Vijan, S (March 2010). "Type 2 diabetes". Annals of Internal Medicine. 152 (5): ITC31-15. doi:10.7326/0003-4819-152-5-201003020-01003. PMID 20194231.
- ""Diabetes Care" January 2010". Diabetes Care. 33: S3. 2009. doi:10.2337/dc10-S003. PMC 2797388. PMID 20042773. Archived from the original on 13 January 2010. Retrieved 29 January 2010.
- Saydah SH, Miret M, Sung J, Varas C, Gause D, Brancati FL (August 2001). "Post-challenge hyperglycemia and mortality in a national sample of U.S. adults". Diabetes Care. 24 (8): 1397–402. doi:10.2337/diacare.24.8.1397. PMID 11473076.
- Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia : report of a WHO/IDF consultation (PDF). World Health Organization. 2006. p. 21. ISBN 978-92-4-159493-6. Archived (PDF) from the original on 11 May 2012.
- Santaguida PL, Balion C, Hunt D, Morrison K, Gerstein H, Raina P, Booker L, Yazdi H (2005). "Diagnosis, Prognosis, and Treatment of Impaired Glucose Tolerance and Impaired Fasting Glucose". Evidence Report/Technology Assessment (Summary). Agency for Healthcare Research and Quality (128): 1–11. PMC 4780988. PMID 16194123. Archived from the original on 16 September 2008. Retrieved 20 July 2008.
- Bartoli E, Fra GP, Carnevale Schianca GP (February 2011). "The oral glucose tolerance test (OGTT) revisited". European Journal of Internal Medicine. 22 (1): 8–12. doi:10.1016/j.ejim.2010.07.008. PMID 21238885.
- Selvin E, Steffes MW, Zhu H, Matsushita K, Wagenknecht L, Pankow J, Coresh J, Brancati FL (March 2010). "Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults". The New England Journal of Medicine. 362 (9): 800–11. CiteSeerX 10.1.1.589.1658. doi:10.1056/NEJMoa0908359. PMC 2872990. PMID 20200384.
- Kyu HH, Bachman VF, Alexander LT, Mumford JE, Afshin A, Estep K, Veerman JL, Delwiche K, Iannarone ML, Moyer ML, Cercy K, Vos T, Murray CJ, Forouzanfar MH (August 2016). "Physical activity and risk of breast cancer, colon cancer, diabetes, ischemic heart disease, and ischemic stroke events: systematic review and dose-response meta-analysis for the Global Burden of Disease Study 2013". BMJ. 354: i3857. doi:10.1136/bmj.i3857. PMC 4979358. PMID 27510511.
- "Simple Steps to Preventing Diabetes". The Nutrition Source. Harvard T.H. Chan School of Public Health. Archived from the original on 25 April 2014.
- Willi C, Bodenmann P, Ghali WA, Faris PD, Cornuz J (December 2007). "Active smoking and the risk of type 2 diabetes: a systematic review and meta-analysis". JAMA. 298 (22): 2654–64. doi:10.1001/jama.298.22.2654. PMID 18073361.
- "Chronic diseases and their common risk factors" (PDF). World Health Organization. 2005. Archived (PDF) from the original on 2016-10-17. Retrieved 30 August 2016.
- Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, Raskin P, Zinman B (December 2005). "Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes". The New England Journal of Medicine. 353 (25): 2643–53. doi:10.1056/NEJMoa052187. PMC 2637991. PMID 16371630.
- "The effect of intensive diabetes therapy on the development and progression of neuropathy. The Diabetes Control and Complications Trial Research Group". Annals of Internal Medicine. 122 (8): 561–68. April 1995. doi:10.7326/0003-4819-122-8-199504150-00001. PMID 7887548.
- Qaseem A, Wilt TJ, Kansagara D, Horwitch C, Barry MJ, Forciea MA (April 2018). "Hemoglobin A1c Targets for Glycemic Control With Pharmacologic Therapy for Nonpregnant Adults With Type 2 Diabetes Mellitus: A Guidance Statement Update From the American College of Physicians". Annals of Internal Medicine. 168 (8): 569–576. doi:10.7326/M17-0939. PMID 29507945.
- National Institute for Health and Clinical Excellence. Clinical guideline 66: Type 2 diabetes. London, 2008.
- Cavanagh PR (2004). "Therapeutic footwear for people with diabetes". Diabetes/Metabolism Research and Reviews. 20 Suppl 1 (Suppl 1): S51–55. doi:10.1002/dmrr.435. PMID 15150815.
- Haw JS, Galaviz KI, Straus AN, Kowalski AJ, Magee MJ, Weber MB, Wei J, Narayan KM, Ali MK (December 2017). "Long-term Sustainability of Diabetes Prevention Approaches: A Systematic Review and Meta-analysis of Randomized Clinical Trials". JAMA Internal Medicine. 177 (12): 1808–17. doi:10.1001/jamainternmed.2017.6040. PMC 5820728. PMID 29114778.
- Mottalib A, Kasetty M, Mar JY, Elseaidy T, Ashrafzadeh S, Hamdy O (August 2017). "Weight Management in Patients with Type 1 Diabetes and Obesity". Current Diabetes Reports. 17 (10): 92. doi:10.1007/s11892-017-0918-8. PMC 5569154. PMID 28836234.
- American Diabetes, Association. (January 2019). "5. Lifestyle Management: Standards of Medical Care in Diabetes-2019". Diabetes Care. 42 (Suppl 1): S46–S60. doi:10.2337/dc19-S005. PMID 30559231.
- Evert AB, Dennison M, Gardner CD, Garvey WT, Lau KH, MacLeod J, et al. (May 2019). "Nutrition Therapy for Adults With Diabetes or Prediabetes: A Consensus Report". Diabetes Care (Professional society guidelines). 42 (5): 731–754. doi:10.2337/dci19-0014. PMC 7011201. PMID 31000505.
- Emadian A, Andrews RC, England CY, Wallace V, Thompson JL (November 2015). "The effect of macronutrients on glycaemic control: a systematic review of dietary randomised controlled trials in overweight and obese adults with type 2 diabetes in which there was no difference in weight loss between treatment groups". The British Journal of Nutrition. 114 (10): 1656–66. doi:10.1017/S0007114515003475. PMC 4657029. PMID 26411958.
- Grams J, Garvey WT (June 2015). "Weight Loss and the Prevention and Treatment of Type 2 Diabetes Using Lifestyle Therapy, Pharmacotherapy, and Bariatric Surgery: Mechanisms of Action". Current Obesity Reports. 4 (2): 287–302. doi:10.1007/s13679-015-0155-x. PMID 26627223.
- Rosberger, DF (December 2013). "Diabetic retinopathy: current concepts and emerging therapy". Endocrinology and Metabolism Clinics of North America. 42 (4): 721–45. doi:10.1016/j.ecl.2013.08.001. PMID 24286948.
- MacIsaac, RJ; Jerums, G; Ekinci, EI (March 2018). "Glycemic Control as Primary Prevention for Diabetic Kidney Disease". Advances in Chronic Kidney Disease. 25 (2): 141–148. doi:10.1053/j.ackd.2017.11.003. PMID 29580578.
- Pozzilli, P; Strollo, R; Bonora, E (March 2014). "One size does not fit all glycemic targets for type 2 diabetes". Journal of Diabetes Investigation. 5 (2): 134–41. doi:10.1111/jdi.12206. PMC 4023573. PMID 24843750.
- Krentz, AJ; Bailey, CJ (2005). "Oral antidiabetic agents: current role in type 2 diabetes mellitus". Drugs. 65 (3): 385–411. doi:10.2165/00003495-200565030-00005. PMID 15669880.
- Consumer Reports; American College of Physicians (April 2012), "Choosing a type 2 diabetes drug – Why the best first choice is often the oldest drug" (PDF), High Value Care, Consumer Reports, archived (PDF) from the original on July 2, 2014, retrieved August 14, 2012
- Mitchell, Sharon; Malanda, Belma; Damasceno, Albertino; Eckel, Robert H.; Gaita, Dan; Kotseva, Kornelia; Januzzi, James L.; Mensah, George; Plutzky, Jorge; Prystupiuk, Maksym; Ryden, Lars (September 2019). "A Roadmap on the Prevention of Cardiovascular Disease Among People Living With Diabetes". Global Heart. 14 (3): 215–240. doi:10.1016/j.gheart.2019.07.009. ISSN 2211-8179. PMID 31451236.
- Brunström M, Carlberg B (February 2016). "Effect of antihypertensive treatment at different blood pressure levels in patients with diabetes mellitus: systematic review and meta-analyses". BMJ. 352: i717. doi:10.1136/bmj.i717. PMC 4770818. PMID 26920333.
- Brunström, Mattias; Carlberg, Bo (2019-09-30). "Benefits and harms of lower blood pressure treatment targets: systematic review and meta-analysis of randomised placebo-controlled trials". BMJ Open. 9 (9): e026686. doi:10.1136/bmjopen-2018-026686. ISSN 2044-6055. PMC 6773352. PMID 31575567.
- Fox, Caroline S.; Golden, Sherita Hill; Anderson, Cheryl; Bray, George A.; Burke, Lora E.; Boer, Ian H. de; Deedwania, Prakash; Eckel, Robert H.; Ershow, Abby G.; Fradkin, Judith; Inzucchi, Silvio E. (2015-09-01). "Update on Prevention of Cardiovascular Disease in Adults With Type 2 Diabetes Mellitus in Light of Recent Evidence: A Scientific Statement From the American Heart Association and the American Diabetes Association". Diabetes Care. 38 (9): 1777–1803. doi:10.2337/dci15-0012. ISSN 0149-5992. PMC 4876675. PMID 26246459.
- Cheng J, Zhang W, Zhang X, Han F, Li X, He X, Li Q, Chen J (May 2014). "Effect of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers on all-cause mortality, cardiovascular deaths, and cardiovascular events in patients with diabetes mellitus: a meta-analysis". JAMA Internal Medicine. 174 (5): 773–85. doi:10.1001/jamainternmed.2014.348. PMID 24687000.
- Zheng, Sean L.; Roddick, Alistair J.; Ayis, Salma (September 2017). "Effects of aliskiren on mortality, cardiovascular outcomes and adverse events in patients with diabetes and cardiovascular disease or risk: A systematic review and meta-analysis of 13,395 patients". Diabetes & Vascular Disease Research. 14 (5): 400–406. doi:10.1177/1479164117715854. ISSN 1752-8984. PMC 5600262. PMID 28844155.
- Catalá-López, Ferrán; Macías Saint-Gerons, Diego; González-Bermejo, Diana; Rosano, Giuseppe M.; Davis, Barry R.; Ridao, Manuel; Zaragoza, Abel; Montero-Corominas, Dolores; Tobías, Aurelio; de la Fuente-Honrubia, César; Tabarés-Seisdedos, Rafael (March 2016). "Cardiovascular and Renal Outcomes of Renin-Angiotensin System Blockade in Adult Patients with Diabetes Mellitus: A Systematic Review with Network Meta-Analyses". PLOS Medicine. 13 (3): e1001971. doi:10.1371/journal.pmed.1001971. ISSN 1549-1676. PMC 4783064. PMID 26954482.
- Pignone M, Alberts MJ, Colwell JA, Cushman M, Inzucchi SE, Mukherjee D, Rosenson RS, Williams CD, Wilson PW, Kirkman MS (June 2010). "Aspirin for primary prevention of cardiovascular events in people with diabetes: a position statement of the American Diabetes Association, a scientific statement of the American Heart Association, and an expert consensus document of the American College of Cardiology Foundation". Diabetes Care. 33 (6): 1395–402. doi:10.2337/dc10-0555. PMC 2875463. PMID 20508233.
- Frachetti KJ, Goldfine AB (April 2009). "Bariatric surgery for diabetes management". Current Opinion in Endocrinology, Diabetes and Obesity. 16 (2): 119–24. doi:10.1097/MED.0b013e32832912e7. PMID 19276974.
- Schulman AP, del Genio F, Sinha N, Rubino F (September–October 2009). ""Metabolic" surgery for treatment of type 2 diabetes mellitus". Endocrine Practice. 15 (6): 624–31. doi:10.4158/EP09170.RAR. PMID 19625245.
- Colucci RA (January 2011). "Bariatric surgery in patients with type 2 diabetes: a viable option". Postgraduate Medicine. 123 (1): 24–33. doi:10.3810/pgm.2011.01.2242. PMID 21293081.
- Dixon JB, le Roux CW, Rubino F, Zimmet P (June 2012). "Bariatric surgery for type 2 diabetes". Lancet. 379 (9833): 2300–11. doi:10.1016/S0140-6736(12)60401-2. PMID 22683132.
- "Pancreas Transplantation". American Diabetes Association. Archived from the original on 13 April 2014. Retrieved 9 April 2014.
- Polisena J, Tran K, Cimon K, Hutton B, McGill S, Palmer K (October 2009). "Home telehealth for diabetes management: a systematic review and meta-analysis". Diabetes, Obesity & Metabolism. 11 (10): 913–30. doi:10.1111/j.1463-1326.2009.01057.x. PMID 19531058.
- Elflein, John (Dec 10, 2019). Estimated number diabetics worldwide.CS1 maint: date and year (link)
- Shi Y, Hu FB (June 2014). "The global implications of diabetes and cancer". Lancet. 383 (9933): 1947–48. doi:10.1016/S0140-6736(14)60886-2. PMID 24910221.
- "Global Report on Diabetes" (PDF). World Health Organization. 2016. Retrieved 20 September 2018.
- Gale EA, Gillespie KM (January 2001). "Diabetes and gender". Diabetologia. 44 (1): 3–15. doi:10.1007/s001250051573. PMID 11206408.
- Meisinger C, Thorand B, Schneider A, et al. (2002). "Sex differences in risk factors for incident type 2 Diabetes Mellitus: The MONICA Augsburg Cohort Study". JAMA Internal Medicine. 162 (1): 82–89. doi:10.1001/archinte.162.1.82. PMID 11784224.
- Public Health Agency of Canada, Diabetes in Canada: Facts and figures from a public health perspective. Ottawa, 2011.
- Mathers CD, Loncar D (November 2006). "Projections of global mortality and burden of disease from 2002 to 2030". PLOS Medicine. 3 (11): e442. doi:10.1371/journal.pmed.0030442. PMC 1664601. PMID 17132052.
- Wild S, Roglic G, Green A, Sicree R, King H (May 2004). "Global prevalence of diabetes: estimates for the year 2000 and projections for 2030". Diabetes Care. 27 (5): 1047–53. doi:10.2337/diacare.27.5.1047. PMID 15111519.
- Ripoll, Brian C. Leutholtz, Ignacio (2011-04-25). Exercise and disease management (2nd ed.). Boca Raton: CRC Press. p. 25. ISBN 978-1-4398-2759-8. Archived from the original on 2016-04-03.
- Poretsky, Leonid, ed. (2009). Principles of diabetes mellitus (2nd ed.). New York: Springer. p. 3. ISBN 978-0-387-09840-1. Archived from the original on 2016-04-04.
- Roberts, Jacob (2015). "Sickening sweet". Distillations. Vol. 1 no. 4. pp. 12–15. Retrieved 20 March 2018.
- Laios K, Karamanou M, Saridaki Z, Androutsos G (2012). "Aretaeus of Cappadocia and the first description of diabetes" (PDF). Hormones. 11 (1): 109–13. doi:10.1007/BF03401545. PMID 22450352. Archived (PDF) from the original on 2017-01-04.
- Oxford English Dictionary. diabetes. Retrieved 2011-06-10.
- Harper, Douglas (2001–2010). "Online Etymology Dictionary. diabetes.". Archived from the original on 2012-01-13. Retrieved 2011-06-10.
- Aretaeus, De causis et signis acutorum morborum (lib. 2), Κεφ. β. περὶ Διαβήτεω (Chapter 2, On Diabetes, Greek original) Archived 2014-07-02 at the Wayback Machine, on Perseus
- Oxford English Dictionary. mellite. Retrieved 2011-06-10.
- "MyEtimology. mellitus.". Archived from the original on 2011-03-16. Retrieved 2011-06-10.
- Oxford English Dictionary. -ite. Retrieved 2011-06-10.
- Theodore H. Tulchinsky, Elena A. Varavikova (2008). The New Public Health, Second Edition. New York: Academic Press. p. 200. ISBN 978-0-12-370890-8.
- Piwernetz K, Home PD, Snorgaard O, Antsiferov M, Staehr-Johansen K, Krans M (May 1993). "Monitoring the targets of the St Vincent Declaration and the implementation of quality management in diabetes care: the DIABCARE initiative. The DIABCARE Monitoring Group of the St Vincent Declaration Steering Committee". Diabetic Medicine. 10 (4): 371–77. doi:10.1111/j.1464-5491.1993.tb00083.x. PMID 8508624.
- Dubois H, Bankauskaite V (2005). "Type 2 diabetes programmes in Europe" (PDF). Euro Observer. 7 (2): 5–6. Archived (PDF) from the original on 2012-10-24.
- Stewart WF, Ricci JA, Chee E, Hirsch AG, Brandenburg NA (June 2007). "Lost productive time and costs due to diabetes and diabetic neuropathic pain in the US workforce". Journal of Occupational and Environmental Medicine. 49 (6): 672–79. doi:10.1097/JOM.0b013e318065b83a. PMID 17563611.
- Washington R.E.; Andrews R.M.; Mutter R.L. (November 2013). "Emergency Department Visits for Adults with Diabetes, 2010". HCUP Statistical Brief #167. Rockville MD: Agency for Healthcare Research and Quality. Archived from the original on 2013-12-03.
- Parker, Katrina (2008). Living with diabetes. New York: Facts On File. p. 143. ISBN 978-1-4381-2108-6.
- "Diabetes mellitus". Merck Veterinary Manual, 9th edition (online version). 2005. Archived from the original on 2011-09-27. Retrieved 2011-10-23.
- Öhlund, Malin. Feline diabetes mellitus Aspects on epidemiology and pathogenesis (PDF). Acta Universitatis agriculturae Sueciae. ISBN 978-91-7760-067-1.
- "Press Announcement". Journal of Polymer Science B Polymer Physics. 8 (10): 1845. 1970. Bibcode:1970JPoSB...8.1845.. doi:10.1002/pol.1970.160081020. Archived from the original on 3 March 2016. Retrieved 11 February 2016.
- "Inhaled Insulin Clears Hurdle Toward F.D.A. Approval". New York Times. Archived from the original on 7 April 2014. Retrieved 12 April 2014.
- in-PharmaTechnologist.com. "World's first transdermal insulin shows promise". Archived from the original on 2015-05-01. Retrieved 2016-07-03.
- "Phosphagenics Initiates Trial of Transdermal Insulin Gel". fdanews.com. Archived from the original on 2016-08-18. Retrieved 2016-07-03.
- American Diabetes Association (1 January 2003). "American Diabetes Association Statement: Implications of the Diabetes Control and Complications Trial". Diabetes Care. 26 (Supplement 1): S25–S27. doi:10.2337/diacare.26.2007.S25. PMID 12502616.
- The Diabetes Control; Complications Trial Research Group. (1993). "The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus". N Engl J Med. 329 (14): 977–86. doi:10.1056/NEJM199309303291401. PMID 8366922.
- "Position statement: UKPDS – Implications for the care of people with Type 2 diabetes". Diabetes UK. January 1999. Archived from the original on 2 March 2009.
- "Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33)". The Lancet. 352 (9131): 837–53. 1998. doi:10.1016/S0140-6736(98)07019-6. PMID 9742976.
External links
| Classification | |
|---|---|
| External resources |
- Diabetes at Curlie
- American Diabetes Association
- IDF Diabetes Atlas
- National Diabetes Education Program
- ADA's Standards of Medical Care in Diabetes 2019
- Polonsky KS (October 2012). "The past 200 years in diabetes". The New England Journal of Medicine. 367 (14): 1332–40. doi:10.1056/NEJMra1110560. PMID 23034021. S2CID 9456681.
- "Diabetes". MedlinePlus. U.S. National Library of Medicine.