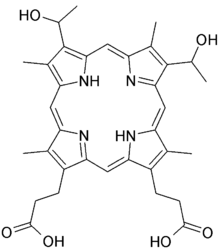
Hematoporphyrin
Hematoporphyrin (Photodyn, Sensibion) is a porphyrin prepared from hemin. It is a derivative of protoporphyrin IX, where the two vinyl groups have been hydrated (converted to alcohols). It is a deeply colored solid that is usually encountered as a solution. Its chemical structure was determined in 1900.[1]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| ECHA InfoCard | 100.034.939 |
| Chemical and physical data | |
| Formula | C34H38N4O6 |
| Molar mass | 598.700 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 172.5 °C (342.5 °F) |
| |
| |
| (verify) | |
It is used as a photosensitizer in photodynamic therapy. Acetylation of hematoporphyrin followed by hydrolysis of the product of that reaction affords a mixture called hematoporphyrin derivative (HPD), which is also used in photodynamic therapy.[2]
Hematoporphyrin has also been used as an antidepressant and antipsychotic since the 1920s.[3][4]
References
- Luzgina VN, Filippovich EI, Evstigneeva RP (May 1977). "Hematoporphyrin IX". Pharmaceutical Chemistry Journal. 11 (5): 613–20. doi:10.1007/BF00780815 (inactive 2020-07-12).
- Kessel D (June 1984). "Hematoporphyrin and HPD: photophysics, photochemistry and phototherapy". Photochemistry and Photobiology. 39 (6): 851–9. doi:10.1111/j.1751-1097.1984.tb08871.x. PMID 6235529.
- O'Neil MJ (2001). The Merck index: an encyclopedia of chemicals, drugs, and biologicals. Rahway, NJ: Merck Research Laboratories. ISBN 0-911910-13-1.
- Strecker EA, Palmer HP, Braceland FJ (May 1934). "Hematoporphyrin as a Therapeutic Agent in the Psychoses". American Journal of Psychiatry. 90 (6): 1157–1173. doi:10.1176/ajp.90.6.1157.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.