Amanullin
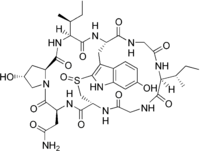
Amanullin is a cyclic peptide. It is an amatoxin, all of which are found in several members of the mushroom genus Amanita. The oral LD50 of amanullin is approximately 20 mg/kg in mice; however, it is non-toxic in humans.
 | |
 | |
| Names | |
|---|---|
| Other names
3-Isoleucine-alpha-amanitin | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C39H54N10O12S | |
| Molar mass | 886.86 g/mol |
| Appearance | Colorless, crystalline solid |
| Soluble | |
| Solubility in ethanol, methanol | Soluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Toxicology
Like other amatoxins, amanullin is an inhibitor of RNA polymerase II. Amanullin has a species dependent and specific attraction to the enzyme RNA polymerase II. Upon ingestion, it binds to the RNA polymerase II enzyme, effectively causing cytolysis of hepatocytes (liver cells).[1]
gollark: I was mostly looking for suggestions for stuff to add to make this a bit less, I don't know, empty.
gollark: All hail compact machines.
gollark: Well, not really, it's a cool simpler assembly computerthing.
gollark: Oh, and RFtools and TIS-3D.
gollark: I have thermal whatever, nuclearcraft, psi, botania, opencomputers, opensecurity, tinkers' construct, compact machines.
See also
References
- Cochet-Meilhac M, Chambon P (June 1974). "Animal DNA-dependent RNA polymerases. 11. Mechanism of the inhibition of RNA polymerases B by amatoxins". Biochim. Biophys. Acta. 353 (2): 160–84. doi:10.1016/0005-2787(74)90182-8. PMID 4601749.
External links
- Amatoxins REVISED
- Poisonous Mushrooms (German)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
