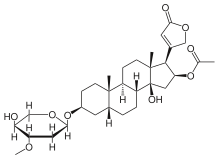
Oleandrin
Oleandrin is a toxic cardiac glycoside found in oleander (Nerium oleander L.). Along with conessine it is primarily responsible for the toxicity of the sap of oleander. Oleandrin has been used for many years in China and Russia for its properties as a cardiac glycoside, for both suicidal and therapeutic purposes as in treatment of cardiac insufficiency.
 | |
| Names | |
|---|---|
| IUPAC name
acetic acid [(3S,5R,10S,13R,14S,16S,17'R)-14-hydroxy-3-[[(2R,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyl-
2-tetrahydropyranyl]oxy]-10,13-dimethyl-17-(5-oxo-2H-furan-3-yl)-1,2,3,4,5,6,7,8,9,11,12,15,16,17- tetradecahydrocyclopenta[a]phenanthren-16-yl] ester | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.693 |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C32H48O9 | |
| Molar mass | 576.72 g/mol |
| Appearance | Oleandrin forms colourless, odourless, acicular crystals that are very bitter |
| Density | 1.261 g/ml |
| Melting point | 250.0 °C (482.0 °F; 523.1 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure and reactivity
The structure of oleandrin contains a central steroid nucleus with an unsaturated lactone ring structure on C17 and a dideoxy arabinose group on C3. In addition, the steroid ring has a substitute of an acetyloxy group on C16.[1] Oleandrin resembles very much other glycosides like ouabain and digoxin but has less effect than digoxin. It is however, just like its derivate oleandrigenin, a more potent glycoside than ouabain.[1]
Synthesis
Oleandrin and its derivate oleandrigenin are formed in the N. oleander plant. The oleandrin itself can be won out of the leaves and other parts of the plant but can also be produced in the lab by using cell cultures. Here, the oleandrin synthesis (along with other metabolites) can be stimulated in untransformed plant cell cultures with supplementation of phytohormone. However, this is not enough to produce large quantities because of early cell death. Transgenic cultures of Agrobacteria are able to synthesize great quantities of oleandrin and other metabolites of the oleander plants, fit for pharmaceutical purposes.[2] Chemical synthetic approach to the olendrigenin core structure has been developed.
Related substances
Oleandrin is, apart from its pure form, also closely related to structural similar glycosides and alkaloids, which all have more or less the same characteristics as oleandrin:
Mechanism of action
Because of its properties as a cardiac glycoside, oleandrin interferes in some essential processes within the cell, the most important of these being the inhibition of the Na-K ATPase. This protein enables the cell to exchange the cations Na+ and K+ between the intercellular and extracellular spaces by which, for instance, electronic signaling is made possible in nerve cells. Oleandrin binds to specific amino acids in the protein, causing it to lose its function. After depolarization of the cell in which Na+ flows into the cell, the Na+ cannot be transported back into the extracellular membrane, causing the sodium gradient to disappear. This gradient is the driving force for other transport proteins, such as the sodium-calcium exchanger, which plays an important role in cardiomyocytes.[3][4]
To make muscle contraction possible, a calcium influx from the extracellular fluid into the cell is crucial. After the muscle contraction, the calcium is normally pumped out of the cell and exchanged for sodium. When the sodium gradient is depleted, calcium cannot be pumped back and, as a consequence, accumulates in the cardiomyocyte.[3]
As a result of the high calcium concentration, actin and myosin filaments will bind stronger, unable to relax properly to make a new contraction possible. This may result in cardiac arrhythmias, in the worst case decreasing cardiac output and causing a shortage in oxygen supply in vital tissues.[3]
Apart from being a potent toxic compound, it may also be used in therapeutic ways. Both oleandrin and oleandrigenin, as well as their relatives, may be able to inhibit proliferation of tumor cells and stimulate their apoptosis as a result of the high concentration of intracellular calcium. In addition, it inhibits excretion of fibroblast growth factor 2 through membrane interaction and through inhibition of the Na,K-ATPase pump.[3] However, there are no results from clinical testing on humans that support any use as a cancer treatment.[5]
Symptoms
Symptoms of oleandrin poisoning can cause both gastrointestinal and cardiac effects. The gastrointestinal effects can consist of nausea, abdominal pain, and vomiting, as well as higher salivation and diarrhea (which may contain blood). After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Also, xanthopsia (yellow vision), a burning sensation of the mucous membranes of the eyes, and gastrointestinal tract and respiratory paralysis can occur.[1] Reactions to poisonings from this plant can also affect the central nervous system. These symptoms can include drowsiness, tremors, or shaking of the muscles, seizures, collapse, and even coma that can lead to death. Oleander sap can cause skin irritations, severe eye inflammation and irritation, and allergy reactions characterized by dermatitis when administered topically.[6]
Diagnosis
Diagnosis of oleandrin poisoning is mainly based on a detailed history. Whether or not a plant is ingested, description of the plant, or the plant itself, how much of it was ingested, time between ingestion and symptoms are all relevant questions. These can be asked only when the patient is not unconscious, which may happen due to cardiac symptoms, or when a third person is present. Supportive treatment should always be given to those with severe cardiac arrhythmias. History of poisoning (of a plant), ECG abnormalities that fit digoxin poisoning, and being in a region with N. oleander suggest poisoning with the plant.[7] Three methods are used for detecting oleandrin in the blood. Fluorescence polarization immunoassay is widely used. This test is slower and has a lower sensitivity than digoxin immunoassay (Digoxin III).[8] A direct analytic technique like liquid chromatography-electrospray tandem mass spectrometry is used when there are medicolegal issues.[9]
Treatment
Onset of symptoms may vary with the way of intake. Teas made of leaves or root of N. oleander give rise to a more acute onset, while eating raw leaves causes a slower onset of symptoms.[10] Management of oleandrin poisoning is done in the following steps:[11]
- Resuscitate the patient as necessary
- Take pulse, blood pressure, and oxygen saturation. Place on a cardiac monitor and take a 12-lead ECG. Insert IV cannula and give fluids IV
- Treat marked hypotension (systolic <70 mmHg) or bradycardia (<40 bpm) with bolus dose of atropine (2–3 mg). Otherwise give small bolus of atropine (0.3-0.6 mg) or and infusion (0.6 mg/h) to keep heart rate around 70-80 bpm
- Consider the administration of a single dose of activated charcoal to bind any oleandrin left in the gastrointestinal tract.
- Measure serum electrolytes and magnesium. Treat hypokalemia and hypomagnesemia until both concentrations are back in high-normal range
- AV node and/or severe sinus node block, ventricular tachycardia, and serum potassium >5.5 are indications for anti-digoxin Fab, if available. The best regimen is currently unclear. Consider giving 400 mg over 20 minutes followed by 400–800 mg over 4–8 hours by infusion.
- In the absence of anti-digoxin Fab:
- Give insulin/dextrose for potassium >5,5. Do not give calcium.
- Consider treating severe bradycardia due to AV block with temporary pacing
- Treat ventricular fibrillation with low-energy direct current cardioversion.
Previously, forced vomiting or performing gastric lavage was part of the treatment of poisoning, but is no longer recommended.[12] There is a lack of evidence that weighs efficacy versus harm.[13] Activated charcoal is still used, since it binds toxins in the gastrointestinal tract to reduce absorption. It is uncertain whether repeated administration of activated charcoal is effective, in theory interrupting enterohepatic cycling. This treatment is used for digoxin poisoning, another cardiac glycoside.[14] Supportive care like monitoring vitals and electrolyte and fluid balance is important. Patients may present hypovolemic due to vomiting and diarrhea, but severely elevated potassium can also occur.[15] Electrolyte balance is vital, since patients with low cardiac glycoside levels can still die after adequate digoxin Fab antibody treatment if they have disturbed electrolyte levels.[16] Treatment of bradyarrhythmias is by many done with intravenous isoprenaline and/or atropine.[17] In moderate cases, prolonging of the PR interval and progression to AV dissociation, cardiac pacing is used.[18] The effectiveness of all these interventions is unknown and are associated with side-effects. Therefore, consultation with a cardiologist is recommended when managing significant N. Oleander induced arrhythmias.[15] The use of anti-digoxin Fab IV has proven successful in cases of oleandrin poisoning[19] These antibodies have shown to bind in vitro with oleandrin and thus reduce the active concentration in the blood [20] A dose of 400 mg is used in digoxin poisoning, but a dose of 800 mg is recommended for oleandrin poisoning due to the lower binding affinity of the antibody to oleandrin.[21][22] Patients receiving an adequate dose of anti-digoxin Fab show a good response, resolving serious arrhythmias in two hours in fifty percent of the cases. Treated patients showed a rapid increase in heart rate and a significant decline in serum potassium levels.[22] The reason anti-digoxin Fab is sparingly used in developing countries is its high cost, even though it is such an effective treatment.[23]
Anti-cancer therapy
Oleandrin is a potential agent for anti-cancer treatment. Studies show potential in vitro effect for cancers of the colon,[24] non-small cell lung cancer,[25] leukemia,[26] pancreas,[27] melanoma [28] and prostate.[29] It may work as a cytotoxic agent, generating reactive oxygen species or inducing apoptosis, but has also shown to be synergistic with current chemotherapy. This may be due to its potential to inhibit P-glycoprotein. This transporter is responsible for phenotypes of cancer resistant to chemotherapeutic agents.[30] Although this sounds promising, in general, concentrations needed for therapeutic effect are not achievable in patient plasma. Apart from being a chemosensitizer, oleandrin has shown to be a potent radiosensitizer. Oleandrin increases caspase 3 activity in radiodamaged tumor cells and, therefore, increases radiation-induced apoptosis. It is unclear as to whether normal cells suffer the same degree of radiosensitization; however, if they do, oleandrin might not be suitable for combined treatment with radiation therapy.[31]
Metabolism
In mice, oleandrin is metabolized into oleandrigenin. Although oleandrigenin it is not formed in human plasma, it was found in the volunteers injected with oleandrin, suggesting that it is formed in other human tissues.[32] Because of its lipophilic properties, oleandrin can be easily absorbed in the gastrointestinal tract after oral dosing. The clearance is slow. The plasma concentration obtains its maximum at twenty minutes after oral intake (half-life of 2,3±0,5h, but half-life after IV administration is 0,4±0,1h).[33]
It is excreted mostly in feces, but also in urine. Because the main route of excretion is through biliary excretion into the feces, it is mainly the liver that is exposed to oleandrin. As excretion in urine is only a smaller route, the kidneys are less exposed. There is also accumulation in the cardiac tissue, which explains its potential for cardiac toxicity. In mouse studies, it also appeared that oleandrin rapidly accumulates in brain tissue, as it can pass through the blood–brain barrier. The data suggest that other components within oleander extract may enhance transport of oleandrin across the blood–brain barrier.[33]
Toxicity
Oleandrin has been reported to be lethal, but exact dosages are not fully documented. The fatal blood concentration of oleandrin has been estimated for humans to be approximately 20 ng/ml in decreased blood by extrapolation of intoxication symptoms.[34] In practice, there have been adult cases wherein 14-20 oleander leaves (of unknown oleandrin concentration) proved not to be fatal, but also a lethal case of a child that consumed only one leaf.[35]
Effects on animals
Oleandrin poisoning by eating oleander leaves can be lethal at low dosages.[36] Cases of sheep lethality have been reported to only one leaf of Oleander.[35] Symptoms present in poisoned animals include bloody diarrhea and colic, the latter especially in horses. Because the leaf itself is quite bitter, only starving animals will be likely to eat the plant. The lethal dosage for animals is estimated to be about 0.5 mg/kg.[35]
References
- Jortani, Saeed A.; Helm, R. Allen; Valdes, Roland (1996). "Inhibition of Na,K-ATPase by oleandrin and oleandrigenin, and their detection by digoxin immunoassays". Clinical Chemistry. 42 (10): 1654–8. PMID 8855150.
- Ibrahim, Amany K.; Khalifa, Sherief; Youssef, Diaa; Khan, Ikhlas; Mesbah, I; Mesbah, M (2007). "Stimulation of oleandrin production by combined Agrobacterium tumefaciens mediated transformation and fungal elecitation in Nerium oleander cell cultures". Enzyme and Microbial Technology. 41 (3): 331–66. doi:10.1016/j.enzmictec.2007.02.015.
- Timbrell, J. A. (2009). Principles of Biochemical Toxicology. New York: Informa Healthcare. pp. 349–51. ISBN 978-0-8493-7302-2.
- Yang, P.; Menter, D. G.; Cartwright, C.; Chan, D.; Dixon, S.; Suraokar, M.; Mendoza, G.; Llansa, N.; Newman, R. A. (2009). "Oleandrin-mediated inhibition of human tumor cell proliferation: Importance of Na,K-ATPase subunits as drug targets". Molecular Cancer Therapeutics. 8 (8): 2319–2328. doi:10.1158/1535-7163.MCT-08-1085. PMID 19671733.
- "More information on complementary and alternative medicine | American Cancer Society".
- Goetz, Rebecca. J.; Jordan Thomas N.; McCain, John W.; Su, Nancy Y. (1998). "Oleander". Indiana Plants Poisonous to Livestock and Pets. Cooperative Extension Service, Purdue University. Archived from the original on 2005-10-21. Retrieved 2005-10-23.
- S. Dwivedi; S. Rajpal; S. Narang (2006). "Cardiotoxic manifestations of yellow oleander (Thevetia nerifolia) poisoning and its treatment: a case report". Indian Heart Journal 58, p.450-451. Missing or empty
|url=(help) - Actor, Jeffrey K.; Reyes, Meredith; Risin, Semyon A.; Dasgupta, Amitava (2008). "Rapid Detection of Oleander Poisoning by Digoxin III, a New Digoxin Assay: Impact on Serum Digoxin Measurement". American Journal of Clinical Pathology. 129 (4): 548–553. doi:10.1309/CC6791DFF20QPCX3. PMID 18343781.
- Tor, Elizabeth R.; Filigenzi, Michael S.; Puschner, Birgit (2005). "Determination of Oleandrin in Tissues and Biological Fluids by Liquid Chromatography−Electrospray Tandem Mass Spectrometry". Journal of Agricultural and Food Chemistry. 53 (11): 4322–5. doi:10.1021/jf050201s. PMID 15913289.
- Haynes, B; Bessen, H; Wightman, W (1985). "Oleander tea: Herbal draught of death". Annals of Emergency Medicine. 14 (4): 350–3. doi:10.1016/S0196-0644(85)80103-7. PMID 4039113.
- Bandara, V; Weinstein, SA; White, J; Eddleston, M (2010). "A review of the natural history, toxinology, diagnosis and clinical management of Nerium oleander (common oleander) and Thevetia peruviana (yellow oleander) poisoning". Toxicon. 56 (3): 273–81. doi:10.1016/j.toxicon.2010.03.026. PMID 20438743.
- American Academy of Clinical Toxicology and European Association of Poisons Centres and Clinical Toxicologists (2004). "Position paper: ipecac syrup" (PDF). Clinical Toxicology Vol. 42, No. 2, p. 133–143. Retrieved 2011-03-23.
- Eddleston, Michael; Haggalla, Sapumal; Reginald, K.; Sudarshan, K.; Senthilkumaran, M.; Karalliedde, Lakshman; Ariaratnam, Ariaranee; Sheriff, M.H.Rezvi; et al. (2007). "The hazards of gastric lavage for intentional self-poisoning in a resource poor location". Clinical Toxicology. 45 (2): 136–43. doi:10.1080/15563650601006009. PMC 1941903. PMID 17364630.
- Reissell, P; Manninen, V (1982). "Effect of administration of activated charcoal and fibre on absorption, excretion and steady state blood levels of digoxin and digitoxin. Evidence for intestinal secretion of the glycosides". Acta Medica Scandinavica. Supplementum. 668: 88–90. doi:10.1111/j.0954-6820.1982.tb08527.x. PMID 6963097.
- Rajapakse, Senaka (2009). "Management of yellow oleander poisoning". Clinical Toxicology. 47 (3): 206–12. doi:10.1080/15563650902824001. PMID 19306191.
- Eddleston, M.; Warrell, DA (1999). "Management of acute yellow oleander poisoning". QJM. 92 (9): 483–5. doi:10.1093/qjmed/92.9.483. PMID 10627866.
- Peiris-John, RJ; Wickremasinghe, AR (2008). "Efficacy of activated charcoal in yellow oleander poisoning". The Ceylon Medical Journal. 53 (2): 33–5. doi:10.4038/cmj.v53i2.228. PMID 18678118.
- Eddleston, M.; Ariaratnam, C. A.; Meyer, W. P.; Perera, G.; Kularatne, A. M.; Attapattu, S.; Sheriff, M. H. R.; Warrell, D. A. (1999). "Epidemic of self-poisoning with seeds of the yellow oleander tree (Thevetia peruviana) in northern Sri Lanka". Tropical Medicine and International Health. 4 (4): 266–73. doi:10.1046/j.1365-3156.1999.00397.x. PMID 10357862.
- Camphausen, C.; Haas, N. A.; Mattke, A. C. (2005). "Successful treatment of oleander intoxication (cardiac glycosides) with digoxin-specific Fab antibody fragments in a 7-year-old child". Zeitschrift für Kardiologie. 94 (12): 817–23. doi:10.1007/s00392-005-0293-3. PMID 16382383.
- Dasgupta, A; Hart, AP (1997). "Rapid detection of oleander poisoning using fluorescence polarization immunoassay for digitoxin. Effect of treatment with digoxin-specific Fab antibody fragment (ovine)". American Journal of Clinical Pathology. 108 (4): 411–6. doi:10.1093/ajcp/108.4.411. PMID 9322594.
- Bandara, Veronika; Weinstein, Scott A.; White, Julian; Eddleston, Michael (2010). "A review of the natural history, toxinology, diagnosis and clinical management of Nerium oleander (common oleander) and Thevetia peruviana (yellow oleander) poisoning". Toxicon. 56 (3): 273–81. doi:10.1016/j.toxicon.2010.03.026. PMID 20438743.
- Eddleston, M; Rajapakse, S; Rajakanthan; Jayalath, S; Sjöström, L; Santharaj, W; Thenabadu, PN; Sheriff, MHR; Warrell, DA (2000). "Anti-digoxin Fab fragments in cardiotoxicity induced by ingestion of yellow oleander: a randomised controlled trial". The Lancet. 355 (9208): 967–72. doi:10.1016/S0140-6736(00)90014-X. PMID 10768435.
- Eddleston, Michael; Senarathna, Lalith; Mohamed, Fahim; Buckley, Nick; Juszczak, Edmund; Sheriff, MH Rezvi; Ariaratnam, Ariaranee; Rajapakse, Senaka; et al. (2003). "Deaths due to absence of an affordable antitoxin for plant poisoning". The Lancet. 362 (9389): 1041–4. doi:10.1016/S0140-6736(03)14415-7. PMID 14522536.
- Felth, J; Rickardson, L; Rosén, J; Wickström, M; Fryknäs, M; Lindskog, M; Bohlin, L; Gullbo, J (2009). "Cytotoxic effects of cardiac glycosides in colon cancer cells, alone and in combination with standard chemotherapeutic drugs". Journal of Natural Products. 72 (11): 1969–74. doi:10.1021/np900210m. PMID 19894733.
- Frese, S.; Frese-Schaper, M; Andres, AC; Miescher, D; Zumkehr, B; Schmid, RA (2006). "Cardiac Glycosides Initiate Apo2L/TRAIL-Induced Apoptosis in Non-Small Cell Lung Cancer Cells by Up-regulation of Death Receptors 4 and 5". Cancer Research. 66 (11): 5867–5874. doi:10.1158/0008-5472.CAN-05-3544. PMID 16740726.
- Turan, N; Akgün-Dar, K; Kuruca, SE; Kiliçaslan-Ayna, T; Seyhan, VG; Atasever, B; Meriçli, F; Carin, M (2006). "Cytotoxic effects of leaf, stem and root extracts of Nerium oleander on leukemia cell lines and role of the p-glycoprotein in this effect". Journal of Experimental Therapeutics & Oncology. 6 (1): 31–8. PMID 17228522.
- Newman, RA; Kondo, Y; Yokoyama, T; Dixon, S; Cartwright, C; Chan, D; Johansen, M; Yang, P (2007). "Autophagic cell death of human pancreatic tumor cells mediated by oleandrin, a lipid-soluble cardiac glycoside". Integrative Cancer Therapies. 6 (4): 354–64. doi:10.1177/1534735407309623. PMID 18048883.
- Newman, RA; Yang, P; Hittelman, WN; Lu, T; Ho, DH; Ni, D; Chan, D; Vijjeswarapu, M; et al. (2006). "Oleandrin-mediated oxidative stress in human melanoma cells". Journal of Experimental Therapeutics & Oncology. 5 (3): 167–81. PMID 16528968.
- Smith, JA; Madden, T; Vijjeswarapu, M; Newman, RA (2001). "Inhibition of export of fibroblast growth factor-2 (FGF-2) from the prostate cancer cell lines PC3 and DU145 by Anvirzel and its cardiac glycoside component, oleandrin". Biochemical pharmacology. 62 (4): 469–72. doi:10.1016/S0006-2952(01)00690-6. PMID 11448457.
- Dean, Michael (2002). "The Human ATP-Binding Cassette (ABC) Transporter Superfamily". Bethesda (MD): National Center for Biotechnology Information (US). Retrieved 2011-03-23.
- Nasu, S; Milas, L; Kawabe, S; Raju, U; Newman, R (2002). "Enhancement of radiotherapy by oleandrin is a caspase-3 dependent process". Cancer Letters. 185 (2): 145–51. doi:10.1016/S0304-3835(02)00263-X. PMID 12169388.
- Wang, Xiaomin; Plomley, Jeffry B.; Newman, Robert A.; Cisneros, Angela (2000). "LC/MS/MS Analyses of an Oleander Extract for Cancer Treatment". Analytical Chemistry. 72 (15): 3547–52. doi:10.1021/ac991425a. PMID 10952541.
- Ni, Dan; Madden, Timothy L.; Johansen, Mary; Felix, Edward; Ho, Dah H.; Newman, Robert A. (2002). "Murine pharmacokinetics and metabolism of oleandrin, a cytotoxic component of Nerium oleander". Journal of Experimental Therapeutics and Oncology. 2 (5): 278–85. doi:10.1046/j.1359-4117.2002.01052.x. PMID 12416031.
- Wasfi, I; Zorob, O; Alkatheeri, N; Alawadhi, A (2008). "A fatal case of oleandrin poisoning". Forensic Science International. 179 (2–3): e31–6. doi:10.1016/j.forsciint.2008.05.002. PMID 18602779.
- "Nerium oleander L.(PIM 366)". IPCS Inchem. 2005. Retrieved 2005-10-23.
- Soto-Blanco, B.; Fontenele-Neto, J. D.; Silva, D. M.; Reis, P. F.; Nóbrega, J. E. (2006). "Acute cattle intoxication from Nerium oleander pods". Tropical Animal Health and Production. 38 (6): 451–454. doi:10.1007/s11250-006-4400-x. PMID 17243471.
- Michalak, K.; Morawiak, M.; Wicha, J. (November 2016). "Synthetic Approach to the Core Structure of Oleandrin and Related Cardiac Glycosides with Highly Functionalized Ring D". Org. Lett., 18 (23), 6148-6151. PMID 27934370. doi:10.1021/acs.orglett.6b03157.