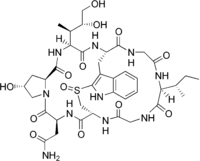
Amaninamide
Amaninamide is a cyclic peptide. It is one of the amatoxins, all of which are found in several members of the mushroom genera Amanita, Lepiota and Galerina. It differs from alpha-amanitin in lacking the hydroxyl group on tryptophan. This alters its UV absorption spectrum but not its toxicity.
 | |
 | |
| Names | |
|---|---|
| Other names
4-(2-mercapto-L-tryptophan)-alpha-Amanitin | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C39H54N10O13S | |
| Molar mass | 902.97 g/mol |
| Appearance | Colorless, crystalline solid |
| Soluble | |
| Solubility in ethanol, methanol | Soluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Toxicology
Like other amatoxins, amaninamide is an inhibitor of RNA polymerase II. Upon ingestion, it binds to the RNA polymerase II enzyme which completely prevents mRNA synthesis, effectively causing cytolysis of hepatocytes (liver cells) and kidney cells.[1]
gollark: > Tell me why exactly you think someone would go to the effort of grafting and setting up a real company, only to con people... because, conning people... gets you money?
gollark: burden of proof™
gollark: Nuclear is very cool and needs to be used more.
gollark: As far as I know it's something like ~~0.5% efficiency~~ (correction: wikipedia says ~5%) and the main advantage of photosynthesis is just that it produces convenient storable chemical energy as output.
gollark: I have a fun diagram too!
See also
References
- M. Cochet-Meillhac; Chambon P. (1974). "Animal DNA-dependent RNA polymerases. 11. Mechanism of the inhibition of RNA polymerases B by amatoxins". Biochim Biophys Acta. 353 (2): 160–184. doi:10.1016/0005-2787(74)90182-8. PMID 4601749.
External links
- Amatoxins REVISED
- Poisonous Mushrooms (German)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
