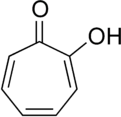
Tropolone
Tropolone is an organic compound with the formula C7H5(OH)O. It is a pale yellow solid that is soluble in organic solvents. The compound has been of interest to research chemists because of its unusual electronic structure and its role as a ligand precursor. Although not usually prepared from tropone, it can be viewed as its derivative with a hydroxyl group in the 2-position.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Hydroxy-2,4,6-cycloheptatrien-1-one | |||
| Other names
2-Hydroxytropone; Purpurocatechol | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.799 | ||
| EC Number |
| ||
| KEGG | |||
| MeSH | D014334 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C7H6O2 | |||
| Molar mass | 122.12 g/mol | ||
| Melting point | 50 to 52 °C (122 to 126 °F; 323 to 325 K) | ||
| Boiling point | 80 to 84 °C (176 to 183 °F; 353 to 357 K) (0.1 mmHg) | ||
| Acidity (pKa) | 6.89 (and -0.5 for conjugate acid) | ||
| -61·10−6 cm3/mol | |||
| Hazards | |||
| S-phrases (outdated) | S22 S24/25 | ||
| Flash point | 112 °C (234 °F; 385 K) | ||
| Related compounds | |||
Related compounds |
Hinokitiol (4-isopropyl-tropolone) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Synthesis and reactions
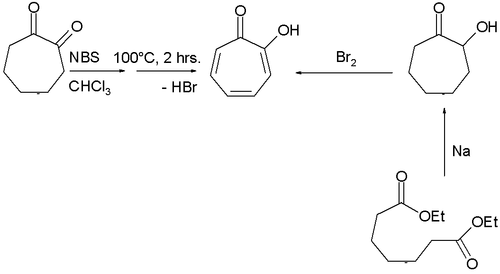
Many methods have been described for the synthesis of tropolone.[2] One involves bromination of 1,2-cycloheptanedione with N-bromosuccinimide followed by dehydrohalogenation at elevated temperatures, while another uses acyloin condensation of the ethyl ester of pimelic acid the acyloin again followed by oxidation by bromine.[3]
The compound readily undergoes O-alkylation to give cycloheptatrienyl derivatives, which in turn are versatile synthetic intermediates.[4] With metal cations, it undergoes deprotonation to give chelate complexes, e.g., Cu(O2C7H5)2.[3]
Natural occurrence
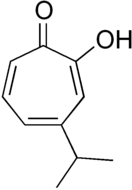
Many natural products contain the tropolone skeleton. Among the simplest, and the first to be made synthetically, were α-, β- and γ-thujaplicins, by Ralph Raphael and colleagues.[6] Others include puberulic acid, stipitatonic acid, stipitatic acid, puberulonic acid, sepedonin, and theaflavins of black tea. It arises via a polyketide pathway, which affords a phenolic intermediate that undergoes ring expansion.[4]
Biological effects
It is an inhibitor of grape polyphenol oxidase[7][8] and mushroom tyrosinase.[9]
References
- Tropolone at Sigma-Aldrich
- Minns, Richard A. (1977). "Tropolone". Org. Synth. 57: 117. doi:10.15227/orgsyn.057.0117.
- Pauson, Peter L. (1955). "Tropones and Tropolones". Chem. Rev. 55 (1): 9–136. doi:10.1021/cr50001a002.
- Pietra, F. (1973). "Seven-membered conjugated carbo- and heterocyclic compounds and their homoconjugated analogs and metal complexes. Synthesis, biosynthesis, structure, and reactivity". Chemical Reviews. 73: 293–364. doi:10.1021/cr60284a002.
- Chedgy, Russell J.; Lim, Young Woon; Breuil, Colette (2009). "Effects of leaching on fungal growth and decay of western redcedar". Canadian Journal of Microbiology. 55 (5): 578–86. doi:10.1139/W08-161. PMID 19483786.
- Cook, J.W.; Scott, A.I.; Raphael, R.A. (1951). "Tropolones. Part II. The synthesis of α-, β-, and γ-thujaplicins". J. Chem. Soc.: 695–698. doi:10.1039/JR9510000695.
- Time-dependent inhibition of grape polyphenol oxidase by tropolone. Edelmira Valero, Manuela Garcia-Moreno, Ramon Varon and Francisco Garcia-Carmona, J. Agric. Food Chem., 1991, volume 39, pp. 1043–1046, doi:10.1021/jf00006a007
- Chedgy, Russell. Secondary metabolites of Western red cedar (Thuja plicata): their biotechnological applications and role in conferring natural durability. LAP Lambert Academic Publishing, 2010, ISBN 3-8383-4661-0, ISBN 978-3-8383-4661-8
- Inhibition of mushroom tyrosinase by tropolone. Varda Kahn and Andrawis Andrawis, Phytochemistry, Volume 24, Issue 5, 1985, Pages 905-908, doi:10.1016/S0031-9422(00)83150-7