3-Methoxytyramine
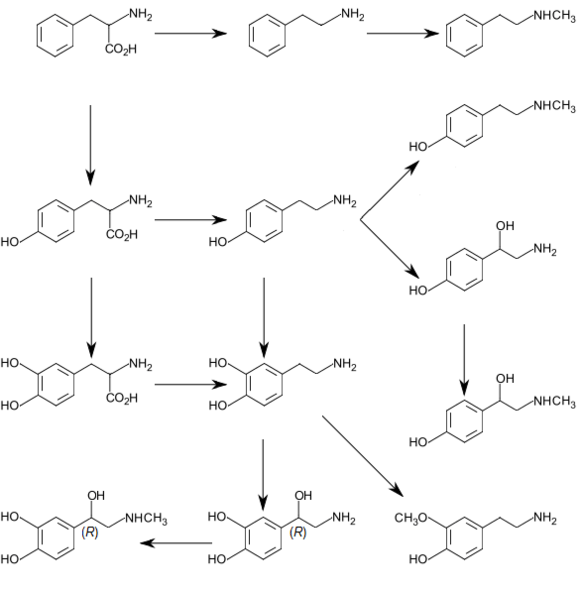
3-Methoxytyramine (3-MT), also known as 3-methoxy-4-hydroxyphenethylamine, is a human trace amine that occurs as a metabolite of the neurotransmitter dopamine.[1] It is formed by the introduction of a methyl group to dopamine by the enzyme catechol-O-methyl transferase (COMT). 3-MT can be further metabolized by the enzyme monoamine oxidase (MAO) to form homovanillic acid (HVA), which is then typically excreted in the urine.
 | |
 | |
| Names | |
|---|---|
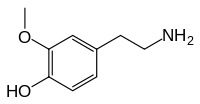
| IUPAC name
4-(2-aminoethyl)-2-methoxyphenol | |
| Other names
3-O-methyldopamine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.122.789 |
| MeSH | 3-methoxytyramine |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H13NO2 | |
| Molar mass | 167.21 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Originally thought to be physiologically inactive, 3-MT has recently been shown to act as an agonist of human TAAR1.[1][2]
Occurrence
3-Methoxytyramine occurs naturally in the prickly pear cactus (genus Opuntia),[3] and is in general widespread throughout the Cactaceae.[4] It has also been found in crown gall tumors on Nicotiana sp.[5]
In humans, 3-methoxytyramine is a trace amine that occurs as a metabolite of dopamine.[1]
See also
References
- Khan MZ, Nawaz W (October 2016). "The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system". Biomed. Pharmacother. 83: 439–449. doi:10.1016/j.biopha.2016.07.002. PMID 27424325.
- Sotnikova TD, Beaulieu JM, Espinoza S, et al. (2010). "The dopamine metabolite 3-methoxytyramine is a neuromodulator". PLOS ONE. 5 (10): e13452. doi:10.1371/journal.pone.0013452. PMC 2956650. PMID 20976142.
- Neuwinger, Hans Dieter (1996). "Cactaceae". African ethnobotany: poisons and drugs: chemistry, pharmacology, toxicology. CRC Press. p. 271. ISBN 978-3-8261-0077-2. Retrieved on June 12, 2009 through Google Book Search.
- Smith T. A. (1977). "Phenethylamine and related compounds in plants". Phytochemistry. 16: 9–18. doi:10.1016/0031-9422(77)83004-5.
- Mitchell S. D.; Firmin J. L.; Gray D. O. (1984). "Enhanced 3-methoxytyramine levels in crown gall tumours and other undifferentiated plant tissues". Biochem. J. 221: 891–5. doi:10.1042/bj2210891. PMC 1144120. PMID 6477503.
- Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacology & Therapeutics. 125 (3): 363–375. doi:10.1016/j.pharmthera.2009.11.005. PMID 19948186.
- Lindemann L, Hoener MC (May 2005). "A renaissance in trace amines inspired by a novel GPCR family". Trends in Pharmacological Sciences. 26 (5): 274–281. doi:10.1016/j.tips.2005.03.007. PMID 15860375.
- Wang X, Li J, Dong G, Yue J (February 2014). "The endogenous substrates of brain CYP2D". European Journal of Pharmacology. 724: 211–218. doi:10.1016/j.ejphar.2013.12.025. PMID 24374199.