Thujaplicin
Thujaplicins (isopropyl cycloheptatrienolones) are a series of tropolone-related chemical substances that have been isolated from the hardwoods of the trees of Cupressaceae family.[1] These compounds are known for their antibacterial, antifungal and antioxidant properties.[2][3] They were the first natural tropolones to be made synthetically.[4]

History
Thujaplicins were discovered in the mid-1930s and purified from the heartwood of Thuja plicata Donn ex D. Don, commonly called as Western red cedar tree.[5] These compounds were also identified in the constituents of Chamaecyparis obtusa, another species from the Cupressaceae family. C. obtusa is native to East Asian countries including Japan and Taiwan, and is also known as Taiwan hinoki, from which the β-thujaplicin was first isolated in 1936 and received its name, hinokitiol. Thujaplicins were the first natural tropolones to be made synthetically, by Ralph Raphael and colleagues, and the β-thujaplicin was the first non-benzenoid aromatic compound identified, by Tetsuo Nozoe and colleagues.[4][5] The resistance of the heartwood of the tree to decay was the main reason prompting to investigate its content and identify the compounds responsible for antimicrobial properties.[4] β-thujaplicin gained more scientific interest beginning in the 2000s.[6] Later, iron-binding activity of β-thujaplicin was discovered and the molecule has been ironically nicknamed as “Iron Man molecule”,[7] because the first name of Tetsuo Nozoe can be translated into English as “Iron Man”.[6]
Occurrence and isolation
Tjujaplicins are found in the hardwoods of the trees belonging to the Cupressaceae family, including Chamaecyparis obtusa (Hinoki cypress), Thuja plicata (Western red cedar), Thujopsis dolabrata var. hondai (Hinoki asunaro), Juniperus cedrus (Canary Islands juniper), Cedrus atlantica (Atlas cedar), Cupressus lusitanica (Mexican white cedar), Chamaecyparis lawsoniana (Port Orford cedar), Chamaecyparis taiwanensis (Taiwan cypress), Chamaecyparis thyoides (Atlantic white cedar), Cupressus arizonica (Arizona cypress), Cupressus macnabiana (MacNab cypress), Cupressus macrocarpa (Monterey cypress), Juniperus chinensis (Chinese juniper), Juniperus communis (Common juniper), Juniperus californica (California juniper), Juniperus occidentalis (Western juniper), Juniperus oxycedrus (Cade), Juniperus sabina (Savin juniper), Calocedrus decurrens (California incense-cedar), Calocedrus formosana (Taiwan incense-cedar), Platycladus orientalis (Chinese thuja), Thuja occidentalis (Northern white-cedar), Thuja standishii (Japanese thuja), Tetraclinis articulata (Sandarac).[8][9][10][11]
Thujaplicins can be produced in plant cell suspension cultures,[12][13] or can be extracted from wood using solvents and ultrasonication.[14]
Chemistry
Thujaplicins belong to tropolones containing an unsaturated seven-membered carbon ring. Thujaplicins are monoterpenoids that are cyclohepta-2,4,6-trien-1-one substituted by a hydroxy group at position 2 and an isopropyl group at positions 3, 4 or 5.[15] These compounds are enols and cyclic ketones. They derive from a hydride of a cyclohepta-1,3,5-triene. Thujaplicins are soluble in organic solvents and aqueous buffers. Hinokitiol is soluble in ethanol, dimethyl sulfoxide, dimethylformamide with a solubility of 20, 30 and 12.5 mg/ml, respectively.[16]
There are three naturally occurring monocyclic tropolones described: α-thujaplicin, β-thujaplicin (hinokitiol), and γ-thujaplicin.[4]
| Compound | Chemical structure | 3D model of the molecule | IUPAC name | Chemical formula |
|---|---|---|---|---|
| α-thujaplicin[17] |  |
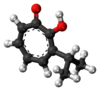 |
2-hydroxy-3-propan-2-ylcyclohepta-2,4,6-trien-1-one | C 10H 12O 2 |
| β-thujaplicin (hinokitiol)[18] |  |
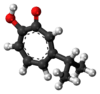 |
2-hydroxy-6-propan-2-ylcyclohepta-2,4,6-trien-1-one | C 10H 12O 2 |
| γ-thujaplicin[19] | 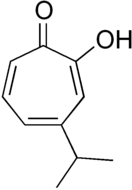 |
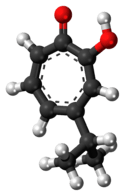 |
2-hydroxy-5-propan-2-ylcyclohepta-2,4,6-trien-1-one | C 10H 12O 2 |
Biological properties
Antibacterial and antifungal activity
Antiviral activity
Anti-inflammatory activity
Insecticidal and pesticidal activity
Thujaplicins are shown to act against Reticulitermes speratus (Japanese termites), Coptotermes formosanus (super termites), Dermatophagoides farinae (dust mites), Tyrophagus putrescentiae (mould mites), Callosobruchus chinensis (adzuki bean weevil), Lasioderma serricorne (cigarette beetle).[9][20][11]
Hinokitiol has also shown some larvicidal activities against Aedes aegypti (yellow fever mosquito) and Culex pipiens (common house mosquito), and anti-plasmodial activities against Plasmodium falciparum and Plasmodium berghei.[11]
Antioxidant activity
Chelating and ionophore activity
Thujaplicins, as other tropolones, demonstrate chelating activity by binding different metal ions.[21]
Anti-browning activity
Tropolone and thujaplicins exhibit potent suppressive activity on enzymatic browning due to inhibition of polyphenol oxidase and tyrosinase. This have been shown in experiments on different vegetables, fruits, mushrooms, plants and other agricultural products.[11] Prevention of darkening has also been elicited on seafood products.[22]
Applications
Skin care and cosmetics
Owing to their antibacterial activities against various microbes colonizing and affecting the skin, thujaplicins are used in skin care and hair growth products[23], and are especially popular in Eastern Asia.
Oral care
Hinokitiol is used in various oral care products, including toothpastes and oral sprays.[23][24]
Veterinary medicine
Due to its antifungal activity against Malassezia pachydermatis, it is used in eardrop formulations for external otitis in dogs.[25][26]
Agriculture
Considering their antifungal activity against many plant-pathogenic fungi, and pesticidal and insecticidal properties, the role of thujaplicins in agriculture is evolving, including their use in the management of different plant diseases and for controlling the postharvest decay.[9][27]
Food additive
Thujaplicins are used as food additives in Japan.[28] Due to its suppressive activity on food browning and the inhibitory activity agaist bacteria and fungi causing food spoilage (such as Clostridium perfringens, Alternaria alternata, Aspergillus niger, Botrytis cinerea, Fusobacterium species, Monilinia fructicola and Rhizopus stolonifer), hinokitiol is also used in food packaging as a shelf-life extending agent.[29][30][31]
Safety
Thujaplicins are considered to be generally very safe.
See also
References
- ERDTMAN, HOLGER; GRIPENBERG, JARL (May 1948). "Antibiotic Substances from the Heart Wood of Thuja plicata Don". Nature. 161 (4097): 719–719. doi:10.1038/161719a0.
- Chedgy, Russell J.; Lim, Young Woon; Breuil, Colette (May 2009). "Effects of leaching on fungal growth and decay of western redcedar". Canadian Journal of Microbiology. 55 (5): 578–586. doi:10.1139/W08-161.
- Chedgy, R. (2010). Secondary Metabolites of Western Red Cedar (Thuja plicata). Lambert Academic Publishing. ISBN 978-3-8383-4661-8.
- Cook, J. W.; Raphael, R. A.; Scott, A. I. (1951). "149. Tropolones. Part II. The synthesis of α-, β-, and γ-thujaplicins". J. Chem. Soc. 0 (0): 695–698. doi:10.1039/JR9510000695.
- Nakanishi, Koji (June 2013). "Tetsuo Nozoe's "Autograph Books by Chemists 1953-1994": An Essay". The Chemical Record. 13 (3): 343–352. doi:10.1002/tcr.201300007.
- "Hinokitiol". American Chemical Society.
- Service, Robert (11 May 2017). "Iron Man molecule restores balance to cells". Science. doi:10.1126/science.aal1178.
- Okabe, T; Saito, K (1994). "Antibacterial and preservative effects of natural Hinokitiol (beta-Thujaplicin) extracted from wood". Acta Agriculturae Zhejiangensis. 6 (4): 257–266.
- Morita, Yasuhiro; Matsumura, Eiko; Okabe, Toshihiro; Fukui, Toru; Shibata, Mitsunobu; Sugiura, Masaaki; Ohe, Tatsuhiko; Tsujibo, Hiroshi; Ishida, Nakao; Inamori, Yoshihiko (2004). "Biological Activity of α-Thujaplicin, the Isomer of Hinokitiol". Biological & Pharmaceutical Bulletin. 27 (6): 899–902. doi:10.1248/bpb.27.899.
- Rebia, Rina Afiani; binti Sadon, Nurul Shaheera; Tanaka, Toshihisa (22 November 2019). "Natural Antibacterial Reagents (Centella, Propolis, and Hinokitiol) Loaded into Poly[(R)-3-hydroxybutyrate-co-(R)-3-hydroxyhexanoate] Composite Nanofibers for Biomedical Applications". Nanomaterials. 9 (12): 1665. doi:10.3390/nano9121665.
- Saniewski, Marian; Horbowicz, Marcin; Kanlayanarat, Sirichai (10 September 2014). "The Biological Activities of Troponoids and Their Use in Agriculture A Review". Journal of Horticultural Research. 22 (1): 5–19. doi:10.2478/johr-2014-0001.
- Zhao, J.; Fujita, K.; Yamada, J.; Sakai, K. (1 April 2001). "Improved β-thujaplicin production in Cupressus lusitanica suspension cultures by fungal elicitor and methyl jasmonate". Applied Microbiology and Biotechnology. 55 (3): 301–305. doi:10.1007/s002530000555.
- Yamada, J.; Fujita, K.; Sakai, K. (April 2003). "Effect of major inorganic nutrients on β-thujaplicin production in a suspension culture of Cupressus lusitanica cells". Journal of Wood Science. 49 (2): 172–175. doi:10.1007/s100860300027.
- Chedgy, Russell J.; Daniels, C.R.; Kadla, John; Breuil, Colette (1 March 2007). "Screening fungi tolerant to Western red cedar (Thuja plicata Donn) extractives. Part 1. Mild extraction by ultrasonication and quantification of extractives by reverse-phase HPLC". Holzforschung. 61 (2): 190–194. doi:10.1515/HF.2007.033.
- "2,4,6-Cycloheptatrien-1-one, 2-hydroxy-3-(1-methylethyl)-". pubchem.ncbi.nlm.nih.gov. PubChem.
- "Hinokitiol - Product Information" (PDF). www.caymanchem.com. Cayman Chemical.
- "2,4,6-Cycloheptatrien-1-one, 2-hydroxy-3-(1-methylethyl)-". pubchem.ncbi.nlm.nih.gov.
- "Hinokitiol". pubchem.ncbi.nlm.nih.gov.
- "gamma-Thujaplicin". pubchem.ncbi.nlm.nih.gov.
- INAMORI, Yoshihiko; SAKAGAMI, Yoshikazu; MORITA, Yasuhiro; SHIBATA, Mistunobu; SUGIURA, Masaaki; KUMEDA, Yuko; OKABE, Toshihiro; TSUJIBO, Hiroshi; ISHIDA, Nakao (2000). "Antifungal Activity of Hinokitiol-Related Compounds on Wood-Rotting Fungi and Their Insecticidal Activities". Biological & Pharmaceutical Bulletin. 23 (8): 995–997. doi:10.1248/bpb.23.995.
- Pietra, Francesco (August 1973). "Seven-membered conjugated carbo- and heterocyclic compounds and their homoconjugated analogs and metal complexes. Synthesis, biosynthesis, structure, and reactivity". Chemical Reviews. 73 (4): 293–364. doi:10.1021/cr60284a002.
- Aladaileh, Saleem; Rodney, Peters; Nair, Sham V.; Raftos, David A. (December 2007). "Characterization of phenoloxidase activity in Sydney rock oysters (Saccostrea glomerata)". Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology. 148 (4): 470–480. doi:10.1016/j.cbpb.2007.07.089.
- "Hinokitiol | 499-44-5". www.chemicalbook.com.
- Suzuki, Joichiro; Tokiwa, Tamami; Mochizuki, Maho; Ebisawa, Masato; Nagano, Takatoshi; Yuasa, Mohei; Kanazashi, Mikimoto; Gomi, Kazuhiro; Arai, Takashi (2008). "Effects of a newly designed toothbrush for the application of periodontal disease treatment medicine (HinoporonTM) on the plaque removal and the improvement of gingivitis". Nihon Shishubyo Gakkai Kaishi (Journal of the Japanese Society of Periodontology). 50 (1): 30–38. doi:10.2329/perio.50.030.
- NAKANO, Yasuyuki; MATSUO, Saburo; TANI, Hiroyuki; SASAI, Kazumi; BABA, Eiichiroh (2006). "Therapeutic Effects of β-Thujaplicin Eardrops on Canine Malassezia-Related Otitis Externa". Journal of Veterinary Medical Science. 68 (4): 373–374. doi:10.1292/jvms.68.373.
- NAKANO, Yasuyuki; WADA, Makoto; TANI, Hiroyuki; SASAI, Kazumi; BABA, Eiichiroh (2005). "Effects of β-Thujaplicin on Anti-Malassezia pachydermatis Remedy for Canine Otitis Externa". Journal of Veterinary Medical Science. 67 (12): 1243–1247. doi:10.1292/jvms.67.1243.
- Aharoni, Y.; Copel, A.; Fallik, E. (June 1993). "Hinokitiol (β‐thujaplicin), for postharvest decay control on 'Galia' melons". New Zealand Journal of Crop and Horticultural Science. 21 (2): 165–169. doi:10.1080/01140671.1993.9513763.
- "The Japan Food chemical Research Faundation". www.ffcr.or.jp.
- L. Brody, Aaron; Strupinsky, E. P.; Kline, Lauri R. (2001). Active Packaging for Food Applications (1 ed.). CRC Press. ISBN 9780367397289.
- MITSUBOSHI, SAORI; OBITSU, RIE; MURAMATSU, KANAKO; FURUBE, KENTARO; YOSHITAKE, SHIGEHIRO; KIUCHI, KAN (2007). "Growth Inhibitory Effect of Shelf Life Extending Agents on Bacillus subtilis IAM 1026". Biocontrol Science. 12 (2): 71–75. doi:10.4265/bio.12.71.
- Vanitha, Thiraviam; Thammawong, Manasikan; Umehara, Hitomi; Nakamura, Nobutaka; Shiina, Takeo (3 September 2019). "Effect of hinokitiol impregnated sheets on shelf life and quality of "KEK‐1" tomatoes during storage". Packaging Technology and Science. 32 (12): 641–648. doi:10.1002/pts.2479.