Scandium oxide
Scandium(III) oxide or scandia is a inorganic compound with formula Sc2O3. It is one of several oxides of rare earth elements with a high melting point. It is used in the preparation of other scandium compounds as well as in high-temperature systems (for its resistance to heat and thermal shock), electronic ceramics, and glass composition (as a helper material).
 | |
| Names | |
|---|---|
| IUPAC name
Scandium(III) oxide | |
| Other names
Scandia, scandium sesquioxide | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.031.844 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Sc2O3 | |
| Molar mass | 137.910 g/mol |
| Appearance | White powder |
| Density | 3.86 g/cm3 |
| Melting point | 2,485 °C (4,505 °F; 2,758 K) |
| insoluble in water | |
| Solubility | soluble in hot acids (reacts) |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
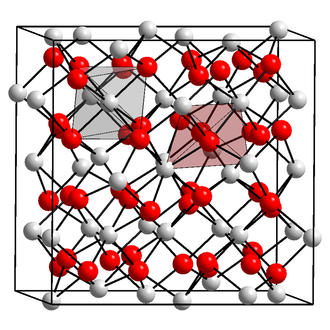
Structure and physical properties
Scandium(III) oxide adopts a cubic crystal structure (point group: tetrahedral (Th), space group: Ia3) containing 6-coordinate metal centres.[1] Powder diffraction analysis shows Sc−O bond distances of 2.159–2.071 Å.[2]
Scandium oxide is an insulator with a band gap of 6.0 eV.[3]
Production
Scandium oxide is the primary form of refined scandium produced by the mining industry. Scandium-rich ores, such as thortveitite (Sc,Y)2(Si2O7) and kolbeckite ScPO4·2H2O are rare, however trace amounts of scandium are present in many other minerals. Scandium oxide is therefore predominately produced as a by-product from the extraction of other elements.
Reactions
Scandium oxide is the primary form of refined scandium produced by the mining industry, making it the start point for all scandium chemistry.
Scandium oxide reacts with most acids upon heating, to produce the expected hydrated product. For example, heating in excess aqueous HCl produces hydrated ScCl3·nH2O. This can be rendered anhydrous by evaporation to dryness in the presence of NH4Cl, with the mixture then being purified by removal of NH4Cl by sublimation at 300-500 °C.[4] The presence of NH4Cl is required, as the hydrated ScCl3·nH2O would otherwise form a mixed oxychloride upon drying.
Likewise, it is converted into hydrated scandium(III) triflate (Sc(OTf)3·nH2O) by a reaction with triflic acid.[5]
Metallic scandium is produced industrially by the reduction of scandium oxide; this proceeds via conversion to scandium fluoride followed by a reduction with metallic calcium. This process is in some ways similar to the Kroll process for the production of metallic titanium.
Scandium oxide forms scandate salts with alkalis, unlike its higher homologues yttrium oxide and lanthanum oxide (but like lutetium oxide), for example forming K3Sc(OH)6 with KOH. In this, scandium oxide shows more similarity with aluminium oxide.
Natural occurrence
Natural scandia, although impure, occurs as mineral kangite.[6]
References
- Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- Knop, Osvald; Hartley, Jean M. (15 April 1968). "Refinement of the crystal structure of scandium oxide". Canadian Journal of Chemistry. 46 (8): 1446–1450. doi:10.1139/v68-236.
- Emeline, A. V.; Kataeva, G. V.; Ryabchuk, V. K.; Serpone, N. (1 October 1999). "Photostimulated Generation of Defects and Surface Reactions on a Series of Wide Band Gap Metal-Oxide Solids". The Journal of Physical Chemistry B. 103 (43): 9190–9199. doi:10.1021/jp990664z.
- Stotz, Robert W.; Melson, Gordon A. (1 July 1972). "Preparation and mechanism of formation of anhydrous scandium(III) chloride and bromide". Inorganic Chemistry. 11 (7): 1720–1721. doi:10.1021/ic50113a058.
- McCleverty, J.A. and Meyer, T.J., Comprehensive Coordination Chemistry II, 2003, Elsevier Science, ISBN 0-08-043748-6, Vol. 3, p. 99 ["Refluxing scandium oxide with triflic acid leads to the isolation of hydrated scandium triflate"]
- Mindat, http://www.mindat.org/min-42879.html
