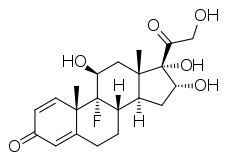
Triamcinolone
Triamcinolone is a glucocorticoid used to treat certain skin diseases, allergies, and rheumatic disorders among others.[2] It is also used to prevent worsening of asthma and COPD.[2] It can be taken in various ways including by mouth, injection into a muscle, and inhalation.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Kenalog, Nasacort, others |
| Other names | Click show to see
(8S,9R,10S,11S,13S,14S,16R,17S)-9-fluoro-11,16,17-trihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-3-one; (1R,2S,10S,11S,13R,14S,15S,17S)-1-fluoro-13,14,17-trihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyltetracyclo[8.7.0.02,7.011,15]heptadeca-3,6-dien-5-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601122 |
| License data | |
| Pregnancy category | |
| Routes of administration | Oral, topical, IM, intra-articular, intrasynovial |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 68% |
| Metabolism | Liver |
| Elimination half-life | 88 minutes |
| Excretion | Fecal and Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.290 |
| Chemical and physical data | |
| Formula | C21H27FO6 |
| Molar mass | 394.439 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects with long term use include osteoporosis, cataracts, thrush, and muscle weakness.[2] Serious side effects may include psychosis, increased risk of infections, adrenal suppression, and bronchospasm.[2] Use in pregnancy is generally safe.[3] It works by decreasing inflammation and immune system activity.[2]
Triamcinolone was patented in 1956 and came into medical use in 1958.[4] It is available as a generic medication.[5] In the United States the wholesale cost is about US$0.06 per gram for the cream.[6] In 2017, it was the 103rd most commonly prescribed medication in the United States, with more than seven million prescriptions.[7][8]
Medical uses
Triamcinolone is used to treat a number of different medical conditions, such as eczema, lichen sclerosus, psoriasis, arthritis, allergies, ulcerative colitis, lupus, sympathetic ophthalmia, temporal arteritis, uveitis, ocular inflammation, keloids, urushiol-induced contact dermatitis, aphthous ulcers (usually as triamcinolone acetonide), central retinal vein occlusion, visualization during vitrectomy and the prevention of asthma attacks.[9][10][11]
The derivative triamcinolone acetonide is the active ingredient in various topical skin preparations (cream, lotion, ointment, aerosol spray) designed to treat skin conditions such as rash, inflammation, redness, or intense itching due to eczema[12] and dermatitis.[13]
Side effects
Side effects of triamcinolone include sore throat, nosebleeds, increased coughing, headache, and runny nose. White patches in the throat or nose indicate a serious side effect. Symptoms of an allergic reaction include rash, itch, swelling, severe dizziness, trouble breathing.[14]
Chemistry
Triamcinolone is a synthetic pregnane corticosteroid and derivative of cortisol (hydrocortisone) and is also known as 1-dehydro-9α-fluoro-16α-hydroxyhydrocortisone or 9α-fluoro-16α-hydroxyprednisolone as well as 9α-fluoro-11β,16α,17α,21-tetrahydroxypregna-1,4-diene-3,20-dione.[15][16]
Society and culture
In 2010, TEVA and Perrigo launched the first generic inhalable triamcinolone.[17]
According to Chang et al. (2014), "Triamcinolone acetonide (TA) is classified as an S9 glucocorticoid in the 2014 Prohibited List published by the World Anti-Doping Agency, which caused it to be prohibited in international athletic competition when administered orally, intravenously, intramuscularly or rectally".[18]
Brand names
Trade names for triamcinolone include Aristocort (Sandoz, now Novartis), Azmacort (KOS), Kenacort (Bristol-Myers Squibb), Kenalog (Bristol-Myers Squibb), Nincort, Ratio-Triacomb, Triaderm (Schering-Plough), Trianex, Tricort (Cadila), Tricortone, Trilone, Tristoject, Volon A.
 Aristocort (triamcinolone) topical cream
Aristocort (triamcinolone) topical cream Kenalog (triamcinolone) IM injection
Kenalog (triamcinolone) IM injection
See also
- Glucocorticoid (a chart comparing various glucocorticoids)
- Triamcinolone acetonide
References
- British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1172. ISBN 9780857113382.
- "Triamcinolone Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- "Triamcinolone Use During Pregnancy". Drugs.com. Retrieved 3 March 2019.
- Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 486. ISBN 978-3-527-60749-5.
- Vallerand, April Hazard (2018). Davis's Drug Guide for Nurses. F.A. Davis. p. 365. ISBN 978-0-8036-7000-6.
- "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Retrieved 3 March 2019.
- "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- "Triamcinolone - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
- Triamcinolone – Drugs.com
- Triamcinolone Inhalation – Drugs.com
- Alcon Receives FDA Approval of Triesence Injectable Triamcinolone Suspension for Use in Eye Surgery – Drugs.com
- Chong M, Fonacier L (December 2016). "Treatment of Eczema: Corticosteroids and Beyond". Clinical Reviews in Allergy & Immunology. 51 (3): 249–262. doi:10.1007/s12016-015-8486-7. PMID 25869743.
- Eichenfield LF, Tom WL, Berger TG, Krol A, Paller AS, Schwarzenberger K, et al. (July 2014). "Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies". Journal of the American Academy of Dermatology. 71 (1): 116–32. doi:10.1016/j.jaad.2014.03.023. PMC 4326095. PMID 24813302.
Topical corticosteroids (TCS) are used in the management of AD in both adults and children and are the mainstay of anti-inflammatory therapy.
- "Drugs and Treatments – Nasacort AQ Nasl – Patient Handout". WebMD. Retrieved 2008-03-24.
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 1228–. ISBN 978-1-4757-2085-3.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 1054–. ISBN 978-3-88763-075-1.
- Perrigo Announces Launch Of Generic Version Of Nasacort AQ – CBS Detroit
- Chang CW, Huang TY, Tseng YC, Chang-Chien GP, Lin SF, Hsu MC (November 2014). "Positive doping results caused by the single-dose local injection of triamcinolone acetonide". Forensic Science International. 244: 1–6. doi:10.1016/j.forsciint.2014.07.024. PMID 25126738.
External links
- "Triamcinolone". Drug Information Portal. U.S. National Library of Medicine.
- "Triamcinolone Topical". MedlinePlus.
- "Triamcinolone Nasal Spray". MedlinePlus.