Dementia
Dementia is a broad category of brain diseases that cause a long-term and often gradual decrease in the ability to think and remember that is severe enough to affect daily functioning.[2] Other common symptoms include emotional problems, difficulties with language, and a decrease in motivation.[2][3] Consciousness is usually not affected.[11] A diagnosis of dementia requires a change from a person's usual mental functioning and a greater decline than one would expect due to aging.[2][12] These diseases have a significant effect on caregivers.[2]
| Dementia | |
|---|---|
| Other names | Senility,[1] senile dementia |
 | |
| Painting of a man diagnosed with dementia | |
| Specialty | Neurology, psychiatry |
| Symptoms | Decreased ability to think and remember, emotional problems, problems with language, decreased motivation[2][3] |
| Usual onset | Gradual[2] |
| Duration | Long term[2] |
| Causes | Alzheimer's disease, vascular dementia, Lewy body dementia, frontotemporal dementia[2][3] |
| Diagnostic method | Cognitive testing (mini mental state examination)[3][4] |
| Differential diagnosis | Delirium[5] |
| Prevention | Early education, prevent high blood pressure, prevent obesity, no smoking, social engagement[6] |
| Treatment | Supportive care[2] |
| Medication | Cholinesterase inhibitors (small benefit)[7][8] |
| Frequency | 46 million (2015)[9] |
| Deaths | 1.9 million (2015)[10] |
The most common type of dementia is Alzheimer's disease, which makes up 50% to 70% of cases.[2][3] Other common types include vascular dementia (25%), dementia with Lewy bodies (15%), and frontotemporal dementia.[2][3] Less common causes include normal pressure hydrocephalus, Parkinson's disease dementia, syphilis, HIV, and Creutzfeldt–Jakob disease.[13] More than one type of dementia may exist in the same person.[2] A small proportion of cases run in families.[14] In the DSM-5, dementia was reclassified as a neurocognitive disorder, with degrees of severity.[15] Diagnosis is usually based on history of the illness and cognitive testing with medical imaging and blood tests used to rule out other possible causes.[4] The mini mental state examination is one commonly used cognitive test.[3] Efforts to prevent dementia include trying to decrease risk factors such as high blood pressure, smoking, diabetes, and obesity.[2] Screening the general population for the disorder is not recommended.[16]
There is no known cure for dementia.[2] Cholinesterase inhibitors such as donepezil are often used and may be beneficial in mild to moderate disorder.[7][17][18] Overall benefit, however, may be minor.[7][8] There are many measures that can improve the quality of life of people with dementia and their caregivers.[2] Cognitive and behavioral interventions may be appropriate.[2] Educating and providing emotional support to the caregiver is important.[2] Exercise programs may be beneficial with respect to activities of daily living and potentially improve outcomes.[19] Treatment of behavioral problems with antipsychotics is common but not usually recommended, due to the limited benefit and the side effects, including an increased risk of death.[20][21]
Globally, dementia affected about 46 million people in 2015.[9] About 10% of people develop the disorder at some point in their lives.[14] It becomes more common with age.[22] About 3% of people between the ages of 65–74 have dementia, 19% between 75 and 84, and nearly half of those over 85 years of age.[23] In 2013 dementia resulted in about 1.7 million deaths, up from 0.8 million in 1990.[24] As more people are living longer, dementia is becoming more common.[22] For people of a specific age, however, it may be becoming less frequent, at least in the developed world, due to a decrease in risk factors.[22] It is one of the most common causes of disability among the old.[3] It is believed to result in economic costs of US$604 billion a year.[2] People with dementia are often physically or chemically restrained to a greater degree than necessary, raising issues of human rights.[2] Social stigma against those affected is common.[3]
Signs and symptoms


The symptoms of dementia vary across types and stages of the diagnosis.[25] The most commonly affected areas include memory, visual-spatial perception and orientation, language, attention and problem solving. Most types of dementia are slow and progressive. By the time signs of the disorder are apparent, deterioration in the brain has been happening for a long time.
Neuropsychiatric symptoms that may be present are termed Behavioural and psychological symptoms of dementia (BPSD). These can include problems with:[26]
- Balance
- Tremor
- Speech and language
- Eating/swallowing
- Memory (believing that a memory has already happened when it has not, thinking an old memory is a new one, combining two memories, or confusing the people in a memory)
- Wandering or restlessness
- Visual perception[27]
Behavioral and psychological symptoms of dementia almost always occur in all types of dementia and may manifest as:[28][29]
- Agitation
- Depression
- Anxiety
- Abnormal motor behavior
- Elated mood
- Irritability
- Apathy
- Disinhibition and impulsivity
- Delusions (often believing people are stealing from them) or hallucinations
- Changes in sleep or appetite.
When people with dementia are put in circumstances beyond their abilities, they may experience a sudden change to crying or anger (a "catastrophic reaction").[30]
Psychosis (often delusions of persecution) and agitation/aggression also often accompany dementia.[31]
Stages
Mild cognitive impairment
In the first stages of dementia, signs and symptoms may be subtle. Often, the early signs become apparent when looking back in time. The earliest stage of dementia is called mild cognitive impairment (MCI). 70% of those diagnosed with MCI later progress to dementia.[12] In MCI, changes in the person's brain have been happening for a long time, but symptoms are just beginning to appear. These problems, however, are not severe enough to affect daily function. If/once they do, the diagnosis becomes dementia. A person with MCI scores between 27 and 30 on the Mini-Mental State Examination (MMSE), which is a normal score. They may have some memory trouble and trouble finding words, but they solve everyday problems and competently handle their life affairs.[32]
Early stages
In the early stage of dementia, symptoms become noticeable to others. In addition, the symptoms begin to interfere with daily activities. MMSE scores are between 20 and 25. The symptoms are dependent on the type of dementia. More complicated chores and tasks around the house or at work become more difficult. The person can usually still take care of themselves but may forget things like taking pills or doing laundry and may need prompting or reminders.[33]
The symptoms of early dementia usually include memory difficulty, but can also include some word-finding problems (anomia) and problems with planning and organizational skills (executive function).[34] One very good way of assessing a person's impairment is by asking if they are still able to handle their finances independently. This is often one of the first things to become problematic. Other signs might be getting lost in new places, repeating things, personality changes,[35] social withdrawal and difficulties at work.
When evaluating dementia, it is important to consider how the person functioned five or ten years earlier. It is also important to consider education level when assessing function. For example, an accountant who can no longer balance a checkbook would be more concerning than a person who had not finished high school or had never taken care of their own finances.[12]
In Alzheimer's dementia the most prominent early symptom is memory difficulty. Others include word-finding problems and getting lost. In other types of dementia, such as dementia with Lewy bodies and fronto-temporal dementia, personality changes and difficulty with organization and planning may be the first signs.
Middle stages
As dementia progresses, initial symptoms generally worsen. The rate of decline is different for each person. MMSE between scores of 6–17 signal moderate dementia . For example, people with moderate Alzheimer's dementia lose almost all new information. People with dementia may be severely impaired in solving problems, and their social judgment is usually also impaired. They cannot usually function outside their own home, and generally should not be left alone. They may be able to do simple chores around the house but not much else, and begin to require assistance for personal care and hygiene beyond simple reminders.[12]
Late stages
People with late-stage dementia typically turn increasingly inward and need assistance with most or all of their personal care. Persons with dementia in the late stages usually need 24-hour supervision to ensure personal safety, as well as to ensure meeting basic needs. If left unsupervised, a person with late-stage dementia may wander or fall, may not recognize common dangers such as a hot stove, or may not realize that they need to use the bathroom. They may become incontinent.[32]
Changes in eating frequently occur. People with late-stage dementia often eat pureed diets, thickened liquids, and require assistance in eating, to prolong their lives, to cause them to retain weight, to reduce choking risk and to make eating easier.[36] The person's appetite may decline to the point that the person does not want to eat at all. They may not want to get out of bed, or may need assistance doing so. Commonly, the person no longer recognizes familiar faces. They may have significant changes in sleeping habits or have trouble sleeping at all.[12]
Causes
Reversible causes
Causes of easily reversible dementia include hypothyroidism, vitamin B12 deficiency, Lyme disease, and neurosyphilis. All people with memory difficulty should be checked for hypothyroidism and B12 deficiency. For Lyme disease and neurosyphilis, testing should be done if risk factors are present. Because risk factors[37] are often difficult to determine, testing for neurosyphilis and Lyme disease, as well as other mentioned factors, may be undertaken as a matter of course where dementia is suspected.[12]:31–32 Hearing loss may also be associated with dementia.[38] Hearing aids may have some benefit.
Alzheimer's
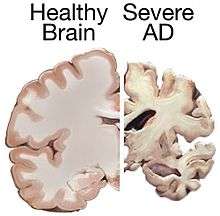
Alzheimer's disease accounts for 50% to 70% of cases of dementia.[2][3] The most common symptoms of Alzheimer's disease are short-term memory loss and word-finding difficulties. Trouble with visual-spatial processing (for example, they may begin to get lost often), reasoning, judgment and insight fail. Insight refers to whether or not the person realizes they have memory problems.
Common early symptoms of Alzheimer's include repetition, getting lost, difficulties tracking bills, problems with cooking especially new or complicated meals, forgetting to take medication and word-finding problems.
The part of the brain most affected by Alzheimer's is the hippocampus. [39] Other parts that show atrophy (shrinking) include the temporal and parietal lobes.[12] Although this pattern suggests Alzheimer's, the brain shrinkage in Alzheimer's disease is variable, and a brain scan is not sufficient for diagnosis. The relationship between anesthesia and AD is unclear.[40]
Vascular dementia
Vascular dementia accounts for at least 20% of dementia cases, making it the second most common type.[41] It is caused by disease or injury affecting the blood supply to the brain, typically involving a series of mini-strokes. The symptoms of this dementia depend on where in the brain the strokes occurred and whether the vessels are large or small.[12] Multiple injuries can cause progressive dementia over time, while a single injury located in an area critical for cognition such as the hippocampus, or thalamus, can lead to sudden cognitive decline.[41]
Brain scans may show evidence of multiple strokes of different sizes in various locations. People with vascular dementia tend to have risk factors for disease of the blood vessels, such as tobacco use, high blood pressure, atrial fibrillation, high cholesterol or diabetes, or other signs of vascular disease such as a previous heart attack or angina.
Dementia with Lewy bodies
Dementia with Lewy bodies (DLB) has the primary symptoms of visual hallucinations and "Parkinsonism". Parkinsonism is the symptoms of Parkinson's disease, which include tremor, rigid muscles, and a face without emotion. The visual hallucinations in DLB are generally vivid hallucinations of people or animals and they often occur when someone is about to fall asleep or wake up. Other prominent symptoms include problems with attention, organization, problem solving and planning (executive function) and difficulty with visual-spatial function.[12]
Again, imaging studies cannot determine the diagnosis of DLB, but some changes are particularly common. A person with DLB often shows occipital hypoperfusion on a SPECT scan or occipital hypometabolism on a PET scan. Generally, a diagnosis of DLB is straightforward and typically does not require a brain scan.[12]
Frontotemporal dementia
Frontotemporal dementias (FTDs) are characterized by drastic personality changes and language difficulties. In all FTDs, the person has a relatively early social withdrawal and early lack of insight. Memory problems are not a main feature.[12][42]
There are six main types of FTD. The first has major symptoms in personality and behavior. This is called behavioral variant FTD (bv-FTD) and is the most common. In bv-FTD, the person shows a change in personal hygiene, becomes rigid in their thinking, and rarely acknowledges problems; they are socially withdrawn, and often have a drastic increase in appetite. They may become socially inappropriate. For example, they may make inappropriate sexual comments, or may begin using pornography openly. One of the most common signs is apathy, or not caring about anything. Apathy, however, is a common symptom in many dementias.[12]
Two types of FTD feature aphasia (language problems) as the main symptom. One type is called semantic variant primary progressive aphasia (SV-PPA). The main feature of this is the loss of the meaning of words. It may begin with difficulty naming things. The person eventually may lose the meaning of objects as well. For example, a drawing of a bird, dog, and an airplane in someone with FTD may all appear almost the same.[12] In a classic test for this, a patient is shown a picture of a pyramid and below it a picture of both a palm tree and a pine tree. The person is asked to say which one goes best with the pyramid. In SV-PPA the person cannot answer that question. The other type is called non-fluent agrammatic variant primary progressive aphasia (NFA-PPA). This is mainly a problem with producing speech. They have trouble finding the right words, but mostly they have a difficulty coordinating the muscles they need to speak. Eventually, someone with NFA-PPA only uses one-syllable words or may become totally mute.
Progressive supranuclear palsy (PSP) is a form of FTD that is characterized by problems with eye movements. Generally the problems begin with difficulty moving the eyes up or down (vertical gaze palsy). Since difficulty moving the eyes upward can sometimes happen in normal aging, problems with downward eye movements are the key in PSP. Other key symptoms include falling backward, balance problems, slow movements, rigid muscles, irritability, apathy, social withdrawal and depression. The person may have certain "frontal lobe" signs such as perseveration, a grasp reflex and utilization behavior (the need to use an object once you see it). People with PSP often have progressive difficulty eating and swallowing, and eventually with talking. Because of the rigidity and slow movements, PSP is sometimes misdiagnosed as Parkinson's disease. On scans the midbrain of people with PSP is generally shrunken (atrophied), but no other common brain abnormalities are visible.
Corticobasal degeneration (CBD) is a rare form of FTD that is characterized by many different types of neurological problems that progressively worsen. This is because the disorder affects the brain in many different places, but at different rates. One common sign is difficulty with using only one limb. One symptom that is rare in any other condition is the "alien limb". The alien limb is a limb that seems to have a mind of its own, it moves without conscious control of the person's brain. Other common symptoms include jerky movements of one or more limbs (myoclonus), symptoms that are different in different limbs (asymmetric), difficulty with speech from inability to move the mouth muscles in a coordinated way, numbness and tingling of the limbs and neglecting one side of vision or senses. In neglect, a person ignores the opposite side of the body from the one that has the problem. For example, a person may not feel pain on one side, or may only draw half of a picture when asked. In addition, the person's affected limbs may be rigid or have muscle contractions causing dystonia (strange repetitive movements).[12] The brain area most often affected in corticobasal degeneration is the posterior frontal lobe and parietal lobe, although many other parts can be affected.[12]
Finally, FT dementia associated with MND (FTD-MND) includes the symptoms of FTD (behavior, language and movement problems) co-occurring with amyotrophic lateral sclerosis (death of motor neurons).
Rapidly progressive
Creutzfeldt-Jakob disease typically causes a dementia that worsens over weeks to months, and is caused by prions. The common causes of slowly progressive dementia also sometimes present with rapid progression: Alzheimer's disease, dementia with Lewy bodies, frontotemporal lobar degeneration (including corticobasal degeneration and progressive supranuclear palsy).
Encephalopathy or delirium may develop relatively slowly and resemble dementia. Possible causes include brain infection (viral encephalitis, subacute sclerosing panencephalitis, Whipple's disease) or inflammation (limbic encephalitis, Hashimoto's encephalopathy, cerebral vasculitis); tumors such as lymphoma or glioma; drug toxicity (e.g., anticonvulsant drugs); metabolic causes such as liver failure or kidney failure; chronic subdural hematoma; and repeated brain trauma (chronic traumatic encephalopathy, a condition associated with contact sports).
Immunologically mediated
Chronic inflammatory conditions that may affect the brain and cognition include Behçet's disease, multiple sclerosis, sarcoidosis, Sjögren's syndrome, systemic lupus erythematosus, celiac disease, and non-celiac gluten sensitivity.[43][44] These types of dementias can rapidly progress, but usually have a good response to early treatment. This consists of immunomodulators or steroid administration, or in certain cases, the elimination of the causative agent.[44] A 2019 review found no association between celiac disease and dementia overall but a potential association with vascular dementia.[45] A 2018 review found a link between celiac disease or non-celiac gluten sensitivity and cognitive impairment and that celiac disease may be associated with Alzheimer's disease, vascular dementia, and frontotemporal dementia.[46] A strict gluten-free diet started early may protect against dementia associated with gluten-related disorders.[45][46]
Other conditions
Many other medical and neurological conditions include dementia only late in the illness. For example, a proportion of patients with Parkinson's disease develop dementia, though widely varying figures are quoted for this proportion.[47] When dementia occurs in Parkinson's disease, the underlying cause may be dementia with Lewy bodies or Alzheimer's disease, or both.[48] Cognitive impairment also occurs in the Parkinson-plus syndromes of progressive supranuclear palsy and corticobasal degeneration (and the same underlying pathology may cause the clinical syndromes of frontotemporal lobar degeneration). Although the acute porphyrias may cause episodes of confusion and psychiatric disturbance, dementia is a rare feature of these rare diseases. Limbic-predominant age-related TDP-43 encephalopathy (LATE) is a type of dementia that primarily affects people in their 80s or 90s and in which TDP-43 protein deposits in the limbic portion of the brain.[49]
Aside from those mentioned above, heritable conditions that can cause dementia (alongside other symptoms) include:[50]
- Alexander disease
- Canavan disease
- Cerebrotendinous xanthomatosis
- Dentatorubral-pallidoluysian atrophy
- Epilepsy
- Fatal familial insomnia
- Fragile X-associated tremor/ataxia syndrome
- Glutaric aciduria type 1
- Krabbe's disease
- Maple syrup urine disease
- Niemann–Pick disease type C
- Neuronal ceroid lipofuscinosis
- Neuroacanthocytosis
- Organic acidemias
- Pelizaeus–Merzbacher disease
- Sanfilippo syndrome type B
- Spinocerebellar ataxia type 2
- Urea cycle disorders
Mild cognitive impairment
Mild cognitive impairment means that the person exhibits memory or thinking difficulties, but those difficulties are not severe enough for a diagnosis of dementia.[51] They should score between 25–30 on the MMSE.[12] Around 70% of people with MCI go on to develop some form of dementia.[12] MCI is generally divided into two categories. The first is primarily memory loss (amnestic MCI). The second is anything else (non-amnestic MCI). People with primarily memory problems typically develop Alzheimer's disease. People with the other type of MCI may develop other types of dementia.
Diagnosis of MCI is often difficult, as cognitive testing may be normal. Often, more in-depth neuropsychological testing is necessary to make the diagnosis. The most commonly used criteria are called the Peterson criteria and include:
- Memory or other cognitive (thought-processing) complaint by the person or a person who knows the patient well.
- A memory or other cognitive problem as compared to a person of the same age and level of education.
- Symptoms not severe enough to affect daily function.
- Absence of dementia.
Fixed cognitive impairment
Various types of brain injury may cause irreversible cognitive impairment that remains stable over time. Traumatic brain injury may cause generalized damage to the white matter of the brain (diffuse axonal injury), or more localized damage (as may also accompany neurosurgery). A temporary reduction in the brain's blood supply or oxygen may lead to hypoxic-ischemic injury. Strokes (ischemic stroke, or intracerebral, subarachnoid, subdural or extradural hemorrhage) or infections (meningitis or encephalitis) affecting the brain, prolonged epileptic seizures, and acute hydrocephalus may also have long-term effects on cognition. Excessive alcohol use may cause alcohol dementia, Wernicke's encephalopathy, or Korsakoff's psychosis.
Slowly progressive
Dementia that begins gradually and worsens over several years is usually caused by neurodegenerative disease—that is, by conditions that affect only or primarily brain neurons and cause gradual but irreversible loss of function. Less commonly, a non-degenerative condition may have secondary effects on brain cells, which may or may not be reversible if the condition is treated.
Causes of dementia depend on the age when symptoms begin. In the elderly population, a large majority of dementia cases are caused by Alzheimer's disease, vascular dementia, or dementia with Lewy bodies.[52][53][54] Hypothyroidism sometimes causes slowly progressive cognitive impairment as the main symptom, which may be fully reversible with treatment. Normal pressure hydrocephalus, though relatively rare, is important to recognize since treatment may prevent progression and improve other symptoms of the condition. However, significant cognitive improvement is unusual.
Dementia is much less common under 65 years of age. Alzheimer's disease is still the most frequent cause, but inherited forms of the disorder account for a higher proportion of cases in this age group. Frontotemporal lobar degeneration and Huntington's disease account for most of the remaining cases.[55] Vascular dementia also occurs, but this in turn may be due to underlying conditions (including antiphospholipid syndrome, CADASIL, MELAS, homocystinuria, moyamoya, and Binswanger's disease). People who receive frequent head trauma, such as boxers or football players, are at risk of chronic traumatic encephalopathy[56] (also called dementia pugilistica in boxers).
In young adults (up to 40 years of age) who were previously of normal intelligence, it is very rare to develop dementia without other features of neurological disease, or without features of disease elsewhere in the body. Most cases of progressive cognitive disturbance in this age group are caused by psychiatric illness, alcohol or other drugs, or metabolic disturbance. However, certain genetic disorders can cause true neurodegenerative dementia at this age. These include familial Alzheimer's disease, SCA17 (dominant inheritance); adrenoleukodystrophy (X-linked); Gaucher's disease type 3, metachromatic leukodystrophy, Niemann-Pick disease type C, pantothenate kinase-associated neurodegeneration, Tay–Sachs disease, and Wilson's disease (all recessive). Wilson's disease is particularly important since cognition can improve with treatment.
At all ages, a substantial proportion of patients who complain of memory difficulty or other cognitive symptoms have depression rather than a neurodegenerative disease. Vitamin deficiencies and chronic infections may also occur at any age; they usually cause other symptoms before dementia occurs, but occasionally mimic degenerative dementia. These include deficiencies of vitamin B12, folate, or niacin, and infective causes including cryptococcal meningitis, AIDS, Lyme disease, progressive multifocal leukoencephalopathy, subacute sclerosing panencephalitis, syphilis, and Whipple's disease.
Limbic-predominant age-related TDP-43 encephalopathy
Limbic-predominant age-related TDP-43 encephalopathy (LATE) is a type of dementia similar to Alzheimer disease which was proposed in 2019.[57] Usually older people are affected.[57]
Hearing loss
Hearing loss is linked with dementia with a greater degree of hearing loss tied to a higher risk.[38] One hypothesis is that as hearing loss increases, cognitive resources are redistributed to auditory perception to the detriment of other cognitive processes.[38] Another hypothesis is that hearing loss leads to social isolation which negatively affect the cognitive functions.[38]
Mixed dementia
About 10% of people with dementia have what is known as mixed dementia, which is usually a combination of Alzheimer's disease and another type of dementia such as frontotemporal dementia or vascular dementia.[58][59]
Diagnosis
Symptoms are similar across dementia types and it is difficult to diagnose by symptoms alone. Diagnosis may be aided by brain scanning techniques. In many cases, the diagnosis requires a brain biopsy to become final, but this is rarely recommended (though it can be performed at autopsy). In those who are getting older, general screening for cognitive impairment using cognitive testing or early diagnosis of dementia has not been shown to improve outcomes.[60] However, screening exams are useful in 65+ persons with memory complaints.[12]
Normally, symptoms must be present for at least six months to support a diagnosis.[61] Cognitive dysfunction of shorter duration is called delirium. Delirium can be easily confused with dementia due to similar symptoms. Delirium is characterized by a sudden onset, fluctuating course, a short duration (often lasting from hours to weeks), and is primarily related to a somatic (or medical) disturbance. In comparison, dementia has typically a long, slow onset (except in the cases of a stroke or trauma), slow decline of mental functioning, as well as a longer trajectory (from months to years).[62]
Some mental illnesses, including depression and psychosis, may produce symptoms that must be differentiated from both delirium and dementia.[63] Therefore, any dementia evaluation should include a depression screening such as the Neuropsychiatric Inventory or the Geriatric Depression Scale.[12] Physicians used to think that people with memory complaints had depression and not dementia (because they thought that those with dementia are generally unaware of their memory problems). This is called pseudodementia. However, in recent years researchers have realized that many older people with memory complaints in fact have MCI, the earliest stage of dementia. Depression should always remain high on the list of possibilities, however, for an elderly person with memory trouble.
Changes in thinking, hearing and vision are associated with normal ageing and can cause problems when diagnosing dementia due to the similarities.[64]
Cognitive testing
| Test | Sensitivity | Specificity | Reference |
| MMSE | 71%–92% | 56%–96% | [65] |
| 3MS | 83%–93.5% | 85%–90% | [66] |
| AMTS | 73%–100% | 71%–100% | [66] |
Various brief tests (5–15 minutes) have reasonable reliability to screen for dementia. While many tests have been studied,[67][68][69] presently the mini mental state examination (MMSE) is the best studied and most commonly used. The MMSE is a useful tool for helping to diagnose dementia if the results are interpreted along with an assessment of a person's personality, their ability to perform activities of daily living, and their behaviour.[70] Other cognitive tests include the abbreviated mental test score (AMTS), the, Modified Mini-Mental State Examination (3MS),[71] the Cognitive Abilities Screening Instrument (CASI),[72] the Trail-making test,[73] and the clock drawing test.[74] The MoCA (Montreal Cognitive Assessment) is a reliable screening test and is available online for free in 35 different languages.[12] The MoCA has also been shown somewhat better at detecting mild cognitive impairment than the MMSE.[75][76] The AD-8 – a screening questionnaire used to assess changes in function related to cognitive decline – is potentially useful, but is not diagnostic, is variable, and has risk of bias.[77] Brief cognitive tests may be affected by factors such as age, education and ethnicity.[78]
Another approach to screening for dementia is to ask an informant (relative or other supporter) to fill out a questionnaire about the person's everyday cognitive functioning. Informant questionnaires provide complementary information to brief cognitive tests. Probably the best known questionnaire of this sort is the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE).[79] Evidence is insufficient to determine how accurate the IQCODE is for diagnosing or predicting dementia.[80] The Alzheimer's Disease Caregiver Questionnaire is another tool. It is about 90% accurate for Alzheimer's when by a caregiver.[12] The General Practitioner Assessment Of Cognition combines both a patient assessment and an informant interview. It was specifically designed for use in the primary care setting.
Clinical neuropsychologists provide diagnostic consultation following administration of a full battery of cognitive testing, often lasting several hours, to determine functional patterns of decline associated with varying types of dementia. Tests of memory, executive function, processing speed, attention and language skills are relevant, as well as tests of emotional and psychological adjustment. These tests assist with ruling out other etiologies and determining relative cognitive decline over time or from estimates of prior cognitive abilities.
Instead of using “mild or early stage”, “middle stage”, and “late stage” dementia as descriptors, numeric scales allow more detailed descriptions. These scales include: Global Deterioration Scale for Assessment of Primary Degenerative Dementia (GDS or Reisberg Scale),[81] Functional Assessment Staging Test (FAST),[82] and Clinical Dementia Rating (CDR).
Laboratory tests
Routine blood tests are usually performed to rule out treatable causes. These tests include vitamin B12, folic acid, thyroid-stimulating hormone (TSH), C-reactive protein, full blood count, electrolytes, calcium, renal function, and liver enzymes. Abnormalities may suggest vitamin deficiency, infection, or other problems that commonly cause confusion or disorientation in the elderly.
Imaging
A CT scan or magnetic resonance imaging (MRI scan) is commonly performed, although these tests do not pick up diffuse metabolic changes associated with dementia in a person who shows no gross neurological problems (such as paralysis or weakness) on a neurological exam. CT or MRI may suggest normal pressure hydrocephalus, a potentially reversible cause of dementia, and can yield information relevant to other types of dementia, such as infarction (stroke) that would point at a vascular type of dementia.
The functional neuroimaging modalities of SPECT and PET are more useful in assessing long-standing cognitive dysfunction, since they have shown similar ability to diagnose dementia as a clinical exam and cognitive testing.[83] The ability of SPECT to differentiate the vascular cause (i.e., multi-infarct dementia) from Alzheimer's disease dementias, appears superior to differentiation by clinical exam.[84]
Recent research has established the value of PET imaging using carbon-11 Pittsburgh Compound B as a radiotracer (PIB-PET) in predictive diagnosis, particularly Alzheimer's disease. Studies reported that PIB-PET was 86% accurate in predicting which patients with mild cognitive impairment would develop Alzheimer's disease within two years. In another study, carried out using 66 patients, PET studies using either PIB or another radiotracer, carbon-11 dihydrotetrabenazine (DTBZ), led to more accurate diagnosis for more than one-fourth of patients with mild cognitive impairment or mild dementia.[85]
Prevention
Various factors can decrease the risk of dementia.[6] As a group they may be able to prevent a third of cases. The group includes early education, treating high blood pressure, preventing obesity, preventing hearing loss, treating depression, physical activity, preventing diabetes, not smoking, and social connection.[6][86] The decreased risk with a healthy lifestyle is seen even in those with a high genetic risk.[87] A 2018 review however concluded that no medications have good evidence of a preventive effect, including blood pressure medications.[88] A 2020 review found a decrease in the risk of dementia or cognitive problems from 7.5% to 7.0% with blood pressure lowering medications.[89]
Among otherwise healthy older people, computerized cognitive training may, in the short term, improve memory.[90] However it is not known whether it prevents dementia.[91][92] Exercise has poor evidence of preventing dementia.[93][94] In those with normal mental function evidence for medications is poor.[95] The same applies to supplements.[96]
The early introduction of a strict gluten-free diet in people with celiac disease or non-celiac gluten sensitivity before cognitive impairment begins potentially has a protective effect.[45]
Management
Except for the treatable types listed above, no cure has been developed. Cholinesterase inhibitors are often used early in the disorder course; however, benefit is generally small.[8][97] Treatments other than medication appear to be better for agitation and aggression than medication.[98] Cognitive and behavioral interventions may be appropriate. Some evidence suggests that education and support for the person with dementia, as well as caregivers and family members, improves outcomes.[99] Exercise programs are beneficial with respect to activities of daily living and potentially improve dementia.[19]
Psychological therapies
Psychological therapies for dementia include some limited evidence for reminiscence therapy (namely, some positive effects in the areas of quality of life, cognition, communication and mood – the first three particularly in care home settings),[100] some benefit for cognitive reframing for caretakers,[101] unclear evidence for validation therapy[102] and tentative evidence for mental exercises, such as cognitive stimulation programs for people with mild to moderate dementia.[103] Reminiscence therapy can improve quality of life, cognition, communication and possibly mood in people with dementia in some circumstances, although all of these benefits may be small.[100]
Adult daycare centers as well as special care units in nursing homes often provide specialized care for dementia patients. Adult daycare centers offer supervision, recreation, meals, and limited health care to participants, as well as providing respite for caregivers. In addition, home care can provide one-on-one support and care in the home allowing for more individualized attention that is needed as the disorder progresses. Psychiatric nurses can make a distinctive contribution to people's mental health.[104]
Since dementia impairs normal communication due to changes in receptive and expressive language, as well as the ability to plan and problem solve, agitated behaviour is often a form of communication for the person with dementia. Actively searching for a potential cause, such as pain, physical illness, or overstimulation can be helpful in reducing agitation.[105] Additionally, using an "ABC analysis of behaviour" can be a useful tool for understanding behavior in people with dementia. It involves looking at the antecedents (A), behavior (B), and consequences (C) associated with an event to help define the problem and prevent further incidents that may arise if the person's needs are misunderstood.[106] The strongest evidence for non-pharmacological therapies for the management of changed behaviours in dementia is for using such approaches.[107] Low quality evidence suggests that regular (at least five sessions of) music therapy may help institutionalized residents. It may reduce depressive symptoms and improve overall behaviour. It may also supply a beneficial effect on emotional well-being and quality of life, as well as reduce anxiety.[108] In 2003, The Alzheimer’s Society established 'Singing for the Brain' (SftB) a project based on pilot studies which suggested that the activity encouraged participation and facilitated the learning of new songs. The sessions combine aspects of reminiscence therapy and music.[109]
Some London hospitals found that using color, designs, pictures and lights helped people with dementia adjust to being at the hospital. These adjustments to the layout of the dementia wings at these hospitals helped patients by preventing confusion.[110]
Medications
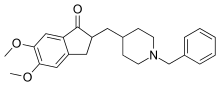
No medications have been shown to prevent or cure dementia.[111] Medications may be used to treat the behavioural and cognitive symptoms, but have no effect on the underlying disease process.[12][112]
Acetylcholinesterase inhibitors, such as donepezil, may be useful for Alzheimer disease[113] and dementia in Parkinson's, DLB, or vascular dementia.[112] The quality of the evidence is poor[114] and the benefit is small.[8] No difference has been shown between the agents in this family.[17] In a minority of people side effects include a slow heart rate and fainting.[115]
As assessment for an underlying cause of the behavior is needed before prescribing antipsychotic medication for symptoms of dementia.[116] Antipsychotic drugs should be used to treat dementia only if non-drug therapies have not worked, and the person's actions threaten themselves or others.[117][118][119][120] Aggressive behavior changes are sometimes the result of other solvable problems, that could make treatment with antipsychotics unnecessary.[117] Because people with dementia can be aggressive, resistant to their treatment, and otherwise disruptive, sometimes antipsychotic drugs are considered as a therapy in response.[117] These drugs have risky adverse effects, including increasing the person's chance of stroke and death.[117] Given these adverse events and small benefit antipsychotics should be avoided whenever possible.[107] Generally, stopping antipsychotics for people with dementia does not cause problems, even in those who have been on them a long time.[121]
N-methyl-D-aspartate (NMDA) receptor blockers such as memantine may be of benefit but the evidence is less conclusive than for AChEIs.[122] Due to their differing mechanisms of action memantine and acetylcholinesterase inhibitors can be used in combination however the benefit is slight.[123][124]
While depression is frequently associated with dementia, selective serotonin reuptake inhibitors (SSRIs) do not appear to affect outcomes.[125][126] The SSRIs sertraline and citalopram have been demonstrated to reduce symptoms of agitation, compared to placebo.[127]
The use of medications to alleviate sleep disturbances that people with dementia often experience has not been well researched, even for medications that are commonly prescribed.[128] In 2012 the American Geriatrics Society recommended that benzodiazepines such as diazepam, and non-benzodiazepine hypnotics, be avoided for people with dementia due to the risks of increased cognitive impairment and falls.[129] Additionally, little evidence supports the effectiveness of benzodiazepines in this population.[128][130] No clear evidence shows that melatonin or ramelteon improves sleep for people with dementia due to Alzheimer's.[128] Limited evidence suggests that a low dose of trazodone may improve sleep, however more research is needed.[128]
No solid evidence indicates that folate or vitamin B12 improves outcomes in those with cognitive problems.[131] Statins have no benefit in dementia.[132] Medications for other health conditions may need to be managed differently for a person who has a dementia diagnosis. It is unclear whether blood pressure medication and dementia are linked. People may experience an increase in cardiovascular-related events if these medications are withdrawn.[133]
The Medication Appropriateness Tool for Comorbid Health Conditions in Dementia (MATCH-D) criteria can help identify ways that a diagnosis of dementia changes medication management for other health conditions.[134] These criteria were developed because people with dementia live with an average of five other chronic diseases, which are often managed with medications.
Pain
As people age, they experience more health problems, and most health problems associated with aging carry a substantial burden of pain; therefore, between 25% and 50% of older adults experience persistent pain. Seniors with dementia experience the same prevalence of conditions likely to cause pain as seniors without dementia.[135] Pain is often overlooked in older adults and, when screened for, is often poorly assessed, especially among those with dementia, since they become incapable of informing others of their pain.[135][136] Beyond the issue of humane care, unrelieved pain has functional implications. Persistent pain can lead to decreased ambulation, depressed mood, sleep disturbances, impaired appetite, and exacerbation of cognitive impairment[136] and pain-related interference with activity is a factor contributing to falls in the elderly.[135][137]
Although persistent pain in people with dementia is difficult to communicate, diagnose, and treat, failure to address persistent pain has profound functional, psychosocial and quality of life implications for this vulnerable population. Health professionals often lack the skills and usually lack the time needed to recognize, accurately assess and adequately monitor pain in people with dementia.[135][138] Family members and friends can make a valuable contribution to the care of a person with dementia by learning to recognize and assess their pain. Educational resources (such as the Understand Pain and Dementia tutorial) and observational assessment tools are available.[135][139][140]
Eating difficulties
Persons with dementia may have difficulty eating. Whenever it is available as an option, the recommended response to eating problems is having a caretaker assist them.[117] A secondary option for people who cannot swallow effectively is to consider gastrostomy feeding tube placement as a way to give nutrition. However, in bringing comfort and maintaining functional status while lowering risk of aspiration pneumonia and death, assistance with oral feeding is at least as good as tube feeding.[117][141] Tube-feeding is associated with agitation, increased use of physical and chemical restraints and worsening pressure ulcers. Tube feedings may cause fluid overload, diarrhea, abdominal pain, local complications, less human interaction and may increase the risk of aspiration.[142][143]
Benefits in those with advanced dementia has not been shown.[144] The risks of using tube feeding include agitation, rejection by the person (pulling out the tube, or otherwise physical or chemical immobilization to prevent them from doing this), or developing pressure ulcers.[117] The procedure is directly related to a 1% fatality rate[145] with a 3% major complication rate.[146] The percentage of people at end of life with dementia using feeding tubes in the US has dropped from 12% in 2000 to 6% as of 2014.[147][148]
Diet
In those with celiac disease or non-celiac gluten sensitivity, a strict gluten-free diet may relieve the symptoms given a mild cognitive impairment.[45][46] Once dementia is advanced no evidence suggests that a gluten free diet is useful.[45]
Alternative medicine
Aromatherapy and massage have unclear evidence.[149][150] Studies support the efficacy and safety of cannabinoids in relieving behavioral and psychological symptoms of dementia.[151]
Omega-3 fatty acid supplements from plants or fish sources do not appear to benefit or harm people with mild to moderate Alzheimer's disease. It is unclear whether taking omega-3 fatty acid supplements can improve other types of dementia.[152]
Palliative care
Given the progressive and terminal nature of dementia, palliative care can be helpful to patients and their caregivers by helping people with the disorder and their caregivers understand what to expect, deal with loss of physical and mental abilities, support the person's wishes and goals including surrogate decision making, and discuss wishes for or against CPR and life support.[153][154] Because the decline can be rapid, and because most people prefer to allow the person with dementia to make their own decisions, palliative care involvement before the late stages of dementia is recommended.[155][156] Further research is required to determine the appropriate palliative care interventions and how well they help people with advanced dementia.[157]
Person-centered care helps maintain the dignity of people with dementia.[158]
Epidemiology
<100 100–120 120–140 140–160 160–180 180–200 |
200–220 220–240 240–260 260–280 280–300 >300 |
The number of cases of dementia worldwide in 2010 was estimated at 35.6 million.[159] In 2015, 46.8 million people live with dementia, with 58% living in low and middle income countries.[160] The prevalence of dementia differs in different world regions, ranging from 4.7% in Central Europe to 8.7% in North Africa/Middle East; the prevalence in other regions is estimated to be between 5.6 and 7.6%.[160] The number of people living with dementia is estimated to double every 20 years. In 2013 dementia resulted in about 1.7 million deaths, up from 0.8 million in 1990.[24] Around two-thirds of individuals with dementia live in low- and middle-income countries, where the sharpest increases in numbers were predicted in a 2009 study.[159]
The annual incidence of dementia diagnosis is over 9.9 million worldwide. Almost half of new dementia cases occur in Asia, followed by Europe (25%), the Americas (18%) and Africa (8%). The incidence of dementia increases exponentially with age, doubling with every 6.3 year increase in age.[160] Dementia affects 5% of the population older than 65 and 20–40% of those older than 85.[161] Rates are slightly higher in women than men at ages 65 and greater.[161]
Dementia impacts not only individuals with dementia, but also their carers and the wider society. Among people aged 60 years and over, dementia is ranked the 9th most burdensome condition according to the 2010 Global Burden of Disease (GBD) estimates. The global costs of dementia was around US$818 billion in 2015, a 35.4% increase from US$604 billion in 2010.[160]
History
Until the end of the 19th century, dementia was a much broader clinical concept. It included mental illness and any type of psychosocial incapacity, including reversible conditions.[162] Dementia at this time simply referred to anyone who had lost the ability to reason, and was applied equally to psychosis, "organic" diseases like syphilis that destroy the brain, and to the dementia associated with old age, which was attributed to "hardening of the arteries".
Dementia has been referred to in medical texts since antiquity. One of the earliest known allusions to dementia is attributed to the 7th-century BC Greek philosopher Pythagoras, who divided the human lifespan into six distinct phases: 0–6 (infancy), 7–21 (adolescence), 22–49 (young adulthood), 50–62 (middle age), 63–79 (old age), and 80–death (advanced age). The last two he described as the "senium", a period of mental and physical decay, and that the final phase was when "the scene of mortal existence closes after a great length of time that very fortunately, few of the human species arrive at, where the mind is reduced to the imbecility of the first epoch of infancy".[163] In 550 BC, the Athenian statesman and poet Solon argued that the terms of a man's will might be invalidated if he exhibited loss of judgement due to advanced age. Chinese medical texts made allusions to the condition as well, and the characters for "dementia" translate literally to "foolish old person".
Athenians Aristotle and Plato spoke of the mental decay of advanced age, apparently viewing it as an inevitable process that affected all old men, and which nothing could prevent. Plato stated that the elderly were unsuited for any position of responsibility because, "There is not much acumen of the mind that once carried them in their youth, those characteristics one would call judgement, imagination, power of reasoning, and memory. They see them gradually blunted by deterioration and can hardly fulfill their function."
For comparison, the Roman statesman Cicero held a view much more in line with modern-day medical wisdom that loss of mental function was not inevitable in the elderly and "affected only those old men who were weak-willed". He spoke of how those who remained mentally active and eager to learn new things could stave off dementia. However, Cicero's views on aging, although progressive, were largely ignored in a world that would be dominated for centuries by Aristotle's medical writings. Physicians during the Roman Empire, such as Galen and Celsus, simply repeated the beliefs of Aristotle while adding few new contributions to medical knowledge.
Byzantine physicians sometimes wrote of dementia. It is recorded that at least seven emperors whose lifespans exceeded 70 years displayed signs of cognitive decline. In Constantinople, special hospitals housed those diagnosed with dementia or insanity, but these did not apply to the emperors, who were above the law and whose health conditions could not be publicly acknowledged.
Otherwise, little is recorded about dementia in Western medical texts for nearly 1700 years. One of the few references was the 13th-century friar Roger Bacon, who viewed old age as divine punishment for original sin. Although he repeated existing Aristotelian beliefs that dementia was inevitable, he did make the progressive assertion that the brain was the center of memory and thought rather than the heart.
Poets, playwrights, and other writers made frequent allusions to the loss of mental function in old age. William Shakespeare notably mentions it in plays such as Hamlet and King Lear.
During the 19th century, doctors generally came to believe that elderly dementia was the result of cerebral atherosclerosis, although opinions fluctuated between the idea that it was due to blockage of the major arteries supplying the brain or small strokes within the vessels of the cerebral cortex.
In 1907 Alzheimer's disease was described. This was associated with particular microscopic changes in the brain, but was seen as a rare disease of middle age because the first person diagnosed with it was a 50-year-old woman. By 1913–20, schizophrenia had been well-defined in a way similar to later times.
This viewpoint remained conventional medical wisdom through the first half of the 20th century, but by the 1960s it was increasingly challenged as the link between neurodegenerative diseases and age-related cognitive decline was established. By the 1970s, the medical community maintained that vascular dementia was rarer than previously thought and Alzheimer's disease caused the vast majority of old age mental impairments. More recently however, it is believed that dementia is often a mixture of conditions.
In 1976, neurologist Robert Katzmann suggested a link between senile dementia and Alzheimer's disease.[164] Katzmann suggested that much of the senile dementia occurring (by definition) after the age of 65, was pathologically identical with Alzheimer's disease occurring in people under age 65 and therefore should not be treated differently.[165] Katzmann thus suggested that Alzheimer's disease, if taken to occur over age 65, is actually common, not rare, and was the fourth- or 5th-leading cause of death, even though rarely reported on death certificates in 1976.
A helpful finding was that although the incidence of Alzheimer's disease increased with age (from 5–10% of 75-year-olds to as many as 40–50% of 90-year-olds), no threshold was found by which age all persons developed it. This is shown by documented supercentenarians (people living to 110 or more) who experienced no substantial cognitive impairment. Some evidence suggests that dementia is most likely to develop between ages 80 and 84 and individuals who pass that point without being affected have a lower chance of developing it. Women account for a larger percentage of dementia cases than men, although this can be attributed to their longer overall lifespan and greater odds of attaining an age where the condition is likely to occur.
Much like other diseases associated with aging, dementia was comparatively rare before the 20th century, because few people lived past 80. Conversely, syphilitic dementia was widespread in the developed world until it was largely eradicated by the use of penicillin after World War II. With significant increases in life expectancy thereafter, the number of people over 65 started rapidly climbing. While elderly persons constituted an average of 3–5% of the population prior to 1945, by 2010 many countries reached 10–14% and in Germany and Japan, this figure exceeded 20%. Public awareness of Alzheimer's Disease greatly increased in 1994 when former US president Ronald Reagan announced that he had been diagnosed with the condition.
In the 21st century, other types of dementia were differentiated from Alzheimer's disease and vascular dementias (the most common types). This differentiation is on the basis of pathological examination of brain tissues, by symptomatology, and by different patterns of brain metabolic activity in nuclear medical imaging tests such as SPECT and PETscans of the brain. The various forms have differing prognoses and differing epidemiologic risk factors. The causal etiology of many of them, including Alzheimer's disease, remains unclear.
Terminology
Dementia in the elderly was once called senile dementia or senility, and viewed as a normal and somewhat inevitable aspect of growing old. This terminology is no longer standard.[166][167]
By 1913–20 the term dementia praecox was introduced to suggest the development of senile-type dementia at a younger age. Eventually the two terms fused, so that until 1952 physicians used the terms dementia praecox (precocious dementia) and schizophrenia interchangeably. The term precocious dementia for a mental illness suggested that a type of mental illness like schizophrenia (including paranoia and decreased cognitive capacity) could be expected to arrive normally in all persons with greater age (see paraphrenia). After about 1920, the beginning use of dementia for what is now understood as schizophrenia and senile dementia helped limit the word's meaning to "permanent, irreversible mental deterioration". This began the change to the later use of the term.
The view that dementia must always be the result of a particular disease process led for a time to the proposed diagnosis of "senile dementia of the Alzheimer's type" (SDAT) in persons over the age of 65, with "Alzheimer's disease" diagnosed in persons younger than 65 who had the same pathology. Eventually, however, it was agreed that the age limit was artificial, and that Alzheimer's disease was the appropriate term for persons with that particular brain pathology, regardless of age.
After 1952, mental illnesses including schizophrenia were removed from the category of organic brain syndromes, and thus (by definition) removed from possible causes of "dementing illnesses" (dementias). At the same, however, the traditional cause of senile dementia – "hardening of the arteries" – now returned as a set of dementias of vascular cause (small strokes). These were now termed multi-infarct dementias or vascular dementias.
Society and culture
.jpg)
The societal cost of dementia is high, especially for family caregivers.[168]
Many countries consider the care of people living with dementia a national priority and invest in resources and education to better inform health and social service workers, unpaid caregivers, relatives and members of the wider community. Several countries have authored national plans or strategies.[169][170] These plans recognize that people can live reasonably with dementia for years, as long as the right support and timely access to a diagnosis are available. British Prime Minister David Cameron described dementia as a "national crisis", affecting 800,000 people in the United Kingdom.[171]
There, as with all mental disorders, people with dementia could potentially be a danger to themselves or others, they can be detained under the Mental Health Act 1983 for assessment, care and treatment. This is a last resort, and is usually avoided by people with family or friends who can ensure care.
Some hospitals in Britain work to provide enriched and friendlier care. To make the hospital wards calmer and less overwhelming to residents, staff replaced the usual nurses' station with a collection of smaller desks, similar to a reception area. The incorporation of bright lighting helps increase positive mood and allow residents to see more easily.[172]
Driving with dementia can lead to injury or death. Doctors should advise appropriate testing on when to quit driving.[173] The United Kingdom DVLA (Driver & Vehicle Licensing Agency) states that people with dementia who specifically have poor short-term memory, disorientation, or lack of insight or judgment are not allowed to drive, and in these instances the DVLA must be informed so that the driving licence can be revoked. They acknowledge that in low-severity cases and those with an early diagnosis, drivers may be permitted to continue driving.
Many support networks are available to people with dementia and their families and caregivers. Charitable organisations aim to raise awareness and campaign for the rights of people living with dementia. Support and guidance are available on assessing testamentary capacity in people with dementia.[174]
In 2015, Atlantic Philanthropies announced a $177 million gift aimed at understanding and reducing dementia. The recipient was Global Brain Health Institute, a program co-led by the University of California, San Francisco and Trinity College Dublin. This donation is the largest non-capital grant Atlantic has ever made, and the biggest philanthropic donation in Irish history.[175]
Dental health
Limited evidence links poor oral health to cognitive decline. However, failure to perform tooth brushing and gingival inflammation can be used as dementia risk predictors.[176]
Oral bacteria
The link between Alzheimer's and gum disease is oral bacteria.[177] In the oral cavity, bacterial species include P. gingivalis, F. nucleatum, P. intermedia, and T. forsythia. Six oral trepomena spirochetes have been examined in the brains of Alzheimer's patients.[178] Spirochetes are neurotropic in nature, meaning they act to destroy nerve tissue and create inflammation. Inflammatory pathogens are an indicator of Alzheimer's disease and bacteria related to gum disease have been found in the brains of Alzheimer's disease sufferers.[178] The bacteria invade nerve tissue in the brain, increasing the permeability of the blood-brain barrier and promoting the onset of Alzheimer's. Individuals with a plethora of tooth plaque risk cognitive decline.[179] Poor oral hygiene can have an adverse effect on speech and nutrition, causing general and cognitive health decline.
Oral viruses
Herpes simplex virus (HSV) has been found in over 70% of the 50 and older population. HSV persists in the peripheral nervous system and can be triggered by stress, illness or fatigue.[178] High proportions of viral-associated proteins in amyloid-containing plaques or neurofibrillary tangles (NFTs) confirm the involvement of HSV-1 in Alzheimer's disease pathology. NFTs are known as the primary marker of Alzheimer's disease. HSV-1 produces the main components of NFTs.[180]
References
- "Dementia". MedlinePlus. U.S. National Library of Medicine. 14 May 2015. Archived from the original on 12 May 2015. Retrieved 6 August 2018.
Dementia Also called: Senility
- "Dementia Fact sheet N°362". who.int. April 2012. Archived from the original on 18 March 2015. Retrieved 28 November 2014.
- Burns A, Iliffe S (February 2009). "Dementia". BMJ. 338: b75. doi:10.1136/bmj.b75. PMID 19196746.
- "Dementia diagnosis and assessment" (PDF). pathways.nice.org.uk. Archived from the original (PDF) on 5 December 2014. Retrieved 30 November 2014.
- Hales, Robert E. (2008). The American Psychiatric Publishing Textbook of Psychiatry. American Psychiatric Pub. p. 311. ISBN 978-1-58562-257-3. Archived from the original on 2017-09-08.
- Livingston G, Sommerlad A, Orgeta V, Costafreda SG, Huntley J, Ames D, et al. (December 2017). "Dementia prevention, intervention, and care". Lancet (Submitted manuscript). 390 (10113): 2673–2734. doi:10.1016/S0140-6736(17)31363-6. PMID 28735855.
- Kavirajan H, Schneider LS (September 2007). "Efficacy and adverse effects of cholinesterase inhibitors and memantine in vascular dementia: a meta-analysis of randomised controlled trials". The Lancet. Neurology. 6 (9): 782–92. doi:10.1016/s1474-4422(07)70195-3. PMID 17689146.
- Commission de la transparence (June 2012). "Drugs for Alzheimer's disease: best avoided. No therapeutic advantage" [Drugs for Alzheimer's disease: best avoided. No therapeutic advantage]. Prescrire International. 21 (128): 150. PMID 22822592.
- GBD 2015 Disease and Injury Incidence and Prevalence Collaborators (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- GBD 2015 Mortality and Causes of Death Collaborators (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/s0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- "Dementia". www.who.int. Retrieved 14 April 2020.
- Budson A, Solomon P (2011). Memory loss : a practical guide for clinicians. [Edinburgh?]: Elsevier Saunders. ISBN 978-1-4160-3597-8.
- Gauthier S (2006). Clinical diagnosis and management of Alzheimer's disease (3rd ed.). Abingdon, Oxon: Informa Healthcare. pp. 53–54. ISBN 978-0-203-93171-4. Archived from the original on 2016-05-03.
- Loy CT, Schofield PR, Turner AM, Kwok JB (March 2014). "Genetics of dementia". Lancet. 383 (9919): 828–40. doi:10.1016/s0140-6736(13)60630-3. PMID 23927914.
- Association, American Psychiatric (2013). Diagnostic and statistical manual of mental disorders : DSM-5 (5th ed.). Washington, DC: American Psychiatric Association. pp. 591–603. ISBN 978-0-89042-554-1.
- "Dementia overview" (PDF). pathways.nice.org.uk. Archived (PDF) from the original on 5 December 2014. Retrieved 30 November 2014.
- Birks J (January 2006). "Cholinesterase inhibitors for Alzheimer's disease". The Cochrane Database of Systematic Reviews (1): CD005593. doi:10.1002/14651858.CD005593. PMID 16437532.
- Rolinski M, Fox C, Maidment I, McShane R (March 2012). "Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson's disease dementia and cognitive impairment in Parkinson's disease" (PDF). The Cochrane Database of Systematic Reviews. 3 (3): CD006504. doi:10.1002/14651858.CD006504.pub2. PMID 22419314.
- Forbes D, Forbes SC, Blake CM, Thiessen EJ, Forbes S (April 2015). "Exercise programs for people with dementia". The Cochrane Database of Systematic Reviews (Submitted manuscript). 132 (4): 195–96. doi:10.1002/14651858.CD006489.pub4. PMID 25874613.
- National Institute for Health and Clinical Excellence. "Low-dose antipsychotics in people with dementia". nice.org.uk. Archived from the original on 5 December 2014. Retrieved 29 November 2014.
- "Information for Healthcare Professionals: Conventional Antipsychotics". fda.gov. 2008-06-16. Archived from the original on 29 November 2014. Retrieved 29 November 2014.
- Larson EB, Yaffe K, Langa KM (December 2013). "New insights into the dementia epidemic". The New England Journal of Medicine. 369 (24): 2275–77. doi:10.1056/nejmp1311405. PMC 4130738. PMID 24283198.
- Umphred, Darcy (2012). Neurological rehabilitation (6th ed.). St. Louis, MO: Elsevier Mosby. p. 838. ISBN 978-0-323-07586-2. Archived from the original on 2016-04-22.
- GBD 2013 Mortality and Causes of Death Collaborators (January 2015). "Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". Lancet. 385 (9963): 117–71. doi:10.1016/S0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
- "Dementia – Signs and Symptoms". American Speech Language Hearing Association.
- Şahin Cankurtaran, E (December 2014). "Management of Behavioral and Psychological Symptoms of Dementia". Noro Psikiyatri Arsivi. 51 (4): 303–12. doi:10.5152/npa.2014.7405. PMC 5353163. PMID 28360647.
- "Sight, perception and hallucinations in dementia" (PDF). Alzheimer's Society. October 2015. Archived (PDF) from the original on 13 August 2017. Retrieved 4 November 2015.
- Cerejeira J, Lagarto L, Mukaetova-Ladinska EB (2012). "Behavioral and psychological symptoms of dementia". Frontiers in Neurology. 3: 73. doi:10.3389/fneur.2012.00073. PMC 3345875. PMID 22586419.
- Calleo J, Stanley M (2008). "Anxiety Disorders in Later Life Differentiated Diagnosis and Treatment Strategies". Psychiatric Times. 25 (8). Archived from the original on 2009-09-04.
- Geddes J, Gelder MG, Mayou R (2005). Psychiatry. Oxford [Oxfordshire]: Oxford University Press. p. 141. ISBN 978-0-19-852863-0. OCLC 56348037.
- Shub D, Kunik ME (April 16, 2009). "Psychiatric Comorbidity in Persons With Dementia: Assessment and Treatment Strategies". Psychiatric Times. 26 (4). Archived from the original on April 27, 2009.
- Hugo J, Ganguli M (August 2014). "Dementia and cognitive impairment: epidemiology, diagnosis, and treatment". Clinics in Geriatric Medicine. 30 (3): 421–42. doi:10.1016/j.cger.2014.04.001. PMC 4104432. PMID 25037289.
- Jenkins, Catharine (2016-01-26). Dementia care at a glance. Ginesi, Laura; Keenan, Bernie. Chichester, West Sussex. ISBN 978-1-118-85998-8. OCLC 905089525.
- Rohrer JD, Knight WD, Warren JE, Fox NC, Rossor MN, Warren JD (January 2008). "Word-finding difficulty: a clinical analysis of the progressive aphasias". Brain. 131 (Pt 1): 8–38. doi:10.1093/brain/awm251. PMC 2373641. PMID 17947337.
- Islam, Maheen; Mazumder, Mridul; Schwabe-Warf, Derek; Stephan, Yannick; Sutin, Angelina R.; Terracciano, Antonio (February 2019). "Personality Changes With Dementia From the Informant Perspective: New Data and Meta-Analysis". Journal of the American Medical Directors Association. 20 (2): 131–137. doi:10.1016/j.jamda.2018.11.004. PMC 6432780. PMID 30630729.
- Erickson K (2013-09-27). How We Die Now: Intimacy and the Work of Dying. Temple University Press. pp. 109–11. ISBN 978-1-4399-0823-5. Archived from the original on 2016-12-23.
- Dawes, P (March 2019). "Hearing interventions to prevent dementia". HNO. 67 (3): 165–171. doi:10.1007/s00106-019-0617-7. PMC 6399173. PMID 30767054.
- Thomson, RS; Auduong, P; Miller, AT; Gurgel, RK (April 2017). "Hearing loss as a risk factor for dementia: A systematic review". Laryngoscope Investigative Otolaryngology. 2 (2): 69–79. doi:10.1002/lio2.65. PMC 5527366. PMID 28894825.
- Dubois B, Hampel H, Feldman HH, et al. (March 2016). "Preclinical Alzheimer's disease: Definition, natural history, and diagnostic criteria". Alzheimers Dement. 12 (3): 292–323. doi:10.1016/j.jalz.2016.02.002. PMC 6417794. PMID 27012484.
- Hussain M, Berger M, Eckenhoff RG, Seitz DP (2014). "General anesthetic and the risk of dementia in elderly patients: current insights". Clinical Interventions in Aging. 9: 1619–28. doi:10.2147/CIA.S49680. PMC 4181446. PMID 25284995.
- Iadecola C (November 2013). "The pathobiology of vascular dementia". Neuron. 80 (4): 844–66. doi:10.1016/j.neuron.2013.10.008. PMC 3842016. PMID 24267647.
- Finger, Elizabeth C. (April 2016). "Frontotemporal Dementias". Continuum (Minneapolis, Minn.). 22 (2 Dementia): 464–489. doi:10.1212/CON.0000000000000300. ISSN 1538-6899. PMC 5390934. PMID 27042904.
- Schofield P (2005). "Dementia associated with toxic causes and autoimmune disease". International Psychogeriatrics (Review). 17 Suppl 1: S129–47. doi:10.1017/s1041610205001997. PMID 16240488.
- Rosenbloom MH, Smith S, Akdal G, Geschwind MD (September 2009). "Immunologically mediated dementias". Current Neurology and Neuroscience Reports (Review). 9 (5): 359–67. doi:10.1007/s11910-009-0053-2. PMC 2832614. PMID 19664365.
- Zis P, Hadjivassiliou M (26 February 2019). "Treatment of Neurological Manifestations of Gluten Sensitivity and Coeliac Disease". Curr Treat Options Neurol (Review). 21 (3): 10. doi:10.1007/s11940-019-0552-7. PMID 30806821.
- Makhlouf S, Messelmani M, Zaouali J, Mrissa R (2018). "Cognitive impairment in celiac disease and non-celiac gluten sensitivity: review of literature on the main cognitive impairments, the imaging and the effect of gluten free diet". Acta Neurol Belg (Review). 118 (1): 21–27. doi:10.1007/s13760-017-0870-z. PMID 29247390.
- Aarsland D, Kurz MW (February 2010). "The epidemiology of dementia associated with Parkinson disease". Journal of the Neurological Sciences (Review). 289 (1–2): 18–22. doi:10.1016/j.jns.2009.08.034. PMID 19733364.
- Galvin JE, Pollack J, Morris JC (November 2006). "Clinical phenotype of Parkinson disease dementia". Neurology. 67 (9): 1605–11. doi:10.1212/01.wnl.0000242630.52203.8f. PMID 17101891.
- Abbasi, Jennifer (21 August 2019). "Debate Sparks Over LATE, a Recently Recognized Dementia". JAMA. 322 (10): 914. doi:10.1001/jama.2019.12232.
- Lamont P (2004). "Cognitive Decline in a Young Adult with Pre-Existent Developmental Delay – What the Adult Neurologist Needs to Know". Practical Neurology. 4 (2): 70–87. doi:10.1111/j.1474-7766.2004.02-206.x. Archived from the original on 2008-10-07.
- Langa KM, Levine DA (December 2014). "The diagnosis and management of mild cognitive impairment: a clinical review". JAMA. 312 (23): 2551–61. doi:10.1001/jama.2014.13806. PMC 4269302. PMID 25514304.
- Neuropathology Group. Medical Research Council Cognitive Function and Aging Study (January 2001). "Pathological correlates of late-onset dementia in a multicentre, community-based population in England and Wales. Neuropathology Group of the Medical Research Council Cognitive Function and Ageing Study (MRC CFAS)". Lancet. 357 (9251): 169–75. doi:10.1016/S0140-6736(00)03589-3. PMID 11213093.
- Wakisaka Y, Furuta A, Tanizaki Y, Kiyohara Y, Iida M, Iwaki T (October 2003). "Age-associated prevalence and risk factors of Lewy body pathology in a general population: the Hisayama study". Acta Neuropathologica. 106 (4): 374–82. doi:10.1007/s00401-003-0750-x. PMID 12904992.
- White L, Petrovitch H, Hardman J, Nelson J, Davis DG, Ross GW, et al. (November 2002). "Cerebrovascular pathology and dementia in autopsied Honolulu-Asia Aging Study participants". Annals of the New York Academy of Sciences. 977 (9): 9–23. Bibcode:2002NYASA.977....9W. doi:10.1111/j.1749-6632.2002.tb04794.x. PMID 12480729.
- Ratnavalli E, Brayne C, Dawson K, Hodges JR (June 2002). "The prevalence of frontotemporal dementia". Neurology. 58 (11): 1615–21. doi:10.1212/WNL.58.11.1615. PMID 12058088.
- McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, Budson AE, Santini VE, Lee HS, Kubilus CA, Stern RA (July 2009). "Chronic traumatic encephalopathy in athletes: progressive tauopathy after repetitive head injury". Journal of Neuropathology and Experimental Neurology. 68 (7): 709–35. doi:10.1097/NEN.0b013e3181a9d503. PMC 2945234. PMID 19535999.
- Nelson PT, Dickson DW, Trojanowski JQ, Jack CR, Boyle PA, Arfanakis K, et al. (April 2019). "Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report". Brain. 142 (6): 1503–1527. doi:10.1093/brain/awz099. PMC 6536849. PMID 31039256.
- What is vascular dementia? Archived 2013-10-19 at the Wayback Machine Alzheimer's Society.
- Lee AY (August 2011). "Vascular dementia". Chonnam Medical Journal. 47 (2): 66–71. doi:10.4068/cmj.2011.47.2.66. PMC 3214877. PMID 22111063.
- Lin JS, O'Connor E, Rossom RC, Perdue LA, Eckstrom E (November 2013). "Screening for cognitive impairment in older adults: A systematic review for the U.S. Preventive Services Task Force". Annals of Internal Medicine. 159 (9): 601–12. doi:10.7326/0003-4819-159-9-201311050-00730. PMID 24145578.
- "Dementia definition". MDGuidelines. Reed Group. Archived from the original on 2009-06-29. Retrieved 2009-06-04.
- Caplan JP, Rabinowitz T (November 2010). "An approach to the patient with cognitive impairment: delirium and dementia". The Medical Clinics of North America. 94 (6): 1103–16, ix. doi:10.1016/j.mcna.2010.08.004. PMID 20951272.
- Gleason OC (March 2003). "Delirium". American Family Physician. 67 (5): 1027–34. PMID 12643363. Archived from the original on 2007-09-29.
- Worrall L, Hickson LM (2003). "Implications for theory, practice, and policy". In Worrall LE, Hickson LM (eds.). Communication disability in aging: from prevention to intervention. Clifton Park, NY: Delmar Learning. pp. 297–98. ISBN 978-0-7693-0015-3.
- Boustani M, Peterson B, Hanson L, Harris R, Lohr KN (June 2003). "Screening for dementia in primary care: a summary of the evidence for the U.S. Preventive Services Task Force". Annals of Internal Medicine. 138 (11): 927–37. doi:10.7326/0003-4819-138-11-200306030-00015. PMID 12779304.
- Cullen B, O'Neill B, Evans JJ, Coen RF, Lawlor BA (August 2007). "A review of screening tests for cognitive impairment". Journal of Neurology, Neurosurgery, and Psychiatry. 78 (8): 790–99. doi:10.1136/jnnp.2006.095414. PMC 2117747. PMID 17178826.
- Sager MA, Hermann BP, La Rue A, Woodard JL (October 2006). "Screening for dementia in community-based memory clinics" (PDF). WMJ. 105 (7): 25–29. PMID 17163083. Archived from the original (PDF) on 2010-06-26.
- Fleisher AS, Sowell BB, Taylor C, Gamst AC, Petersen RC, Thal LJ (May 2007). "Clinical predictors of progression to Alzheimer disease in amnestic mild cognitive impairment". Neurology. 68 (19): 1588–95. doi:10.1212/01.wnl.0000258542.58725.4c. PMID 17287448.
- Karlawish JH, Clark CM (March 2003). "Diagnostic evaluation of elderly patients with mild memory problems". Annals of Internal Medicine. 138 (5): 411–19. doi:10.7326/0003-4819-138-5-200303040-00011. PMID 12614094.
- Creavin ST, Wisniewski S, Noel-Storr AH, Trevelyan CM, Hampton T, Rayment D, et al. (January 2016). "Mini-Mental State Examination (MMSE) for the detection of dementia in clinically unevaluated people aged 65 and over in community and primary care populations". The Cochrane Database of Systematic Reviews (1): CD011145. doi:10.1002/14651858.CD011145.pub2. hdl:1983/00876aeb-2061-43f5-b7e1-938c666030ab. PMID 26760674.
- Teng EL, Chui HC (August 1987). "The Modified Mini-Mental State (3MS) examination". The Journal of Clinical Psychiatry. 48 (8): 314–8. PMID 3611032.
- Teng EL, Hasegawa K, Homma A, Imai Y, Larson E, Graves A, et al. (1994). "The Cognitive Abilities Screening Instrument (CASI): a practical test for cross-cultural epidemiological studies of dementia". International Psychogeriatrics. 6 (1): 45–58, discussion 62. doi:10.1017/S1041610294001602. PMID 8054493.
- Tombaugh TN (March 2004). "Trail Making Test A and B: normative data stratified by age and education". Archives of Clinical Neuropsychology. 19 (2): 203–14. doi:10.1016/S0887-6177(03)00039-8. PMID 15010086.
- Royall DR, Cordes JA, Polk M (May 1998). "CLOX: an executive clock drawing task". Journal of Neurology, Neurosurgery, and Psychiatry. 64 (5): 588–94. doi:10.1136/jnnp.64.5.588. PMC 2170069. PMID 9598672.
- Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (April 2005). "The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment". Journal of the American Geriatrics Society. 53 (4): 695–99. doi:10.1111/j.1532-5415.2005.53221.x. PMID 15817019.
- Breton, Alexandre; Casey, Daniel; Arnaoutoglou, Nikitas A. (2019). "Cognitive tests for the detection of mild cognitive impairment (MCI), the prodromal stage of dementia: Meta-analysis of diagnostic accuracy studies". International Journal of Geriatric Psychiatry. 34 (2): 233–242. doi:10.1002/gps.5016. ISSN 1099-1166. PMID 30370616.
- K, Hendry; C, Green; R, McShane; Ah, Noel-Storr; Dj, Stott; S, Anwer; Aj, Sutton; Jk, Burton; Tj, Quinn (2019-03-04). "AD-8 for detection of dementia across a variety of healthcare settings". The Cochrane Database of Systematic Reviews. doi:10.1002/14651858.CD011121.pub2. PMC 6398085. PMID 30828783.
- Ranson JM, Kuźma E, Hamilton W, Muniz-Terrera G, Langa KM, Llewellyn D (2018-11-28). "Predictors of dementia misclassification when using brief cognitive assessments". Neurology: Clinical Practice. 9 (2): 109–117. doi:10.1212/CPJ.0000000000000566. PMC 6461420. PMID 31041124.
- Jorm AF (September 2004). "The Informant Questionnaire on cognitive decline in the elderly (IQCODE): a review". International Psychogeriatrics. 16 (3): 275–93. doi:10.1017/S1041610204000390. PMID 15559753.
- Harrison JK, Stott DJ, McShane R, Noel-Storr AH, Swann-Price RS, Quinn TJ (November 2016). "Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) for the early diagnosis of dementia across a variety of healthcare settings". The Cochrane Database of Systematic Reviews. 11: CD011333. doi:10.1002/14651858.cd011333.pub2. PMC 6477966. PMID 27869298.
- Reisberg B, Ferris SH, de Leon MJ, Crook T (Sep 1982). "The Global Deterioration Scale for assessment of primary degenerative dementia". Am J Psychiatry. 139 (9): 1136–9. doi:10.1176/ajp.139.9.1136. PMID 7114305.CS1 maint: multiple names: authors list (link)
- Sclan SG, Reisberg B (1992). "Functional assessment staging (FAST) in Alzheimer's disease: reliability, validity, and ordinality". Int Psychogeriatr. 4 (Suppl 1): 55–69. doi:10.1017/s1041610292001157. PMID 1504288.
- Bonte FJ, Harris TS, Hynan LS, Bigio EH, White CL (July 2006). "Tc-99m HMPAO SPECT in the differential diagnosis of the dementias with histopathologic confirmation". Clinical Nuclear Medicine. 31 (7): 376–78. doi:10.1097/01.rlu.0000222736.81365.63. PMID 16785801.
- Dougall NJ, Bruggink S, Ebmeier KP (2004). "Systematic review of the diagnostic accuracy of 99mTc-HMPAO-SPECT in dementia". The American Journal of Geriatric Psychiatry. 12 (6): 554–70. doi:10.1176/appi.ajgp.12.6.554. PMID 15545324.
- Abella HA (June 16, 2009). "Report from SNM: PET imaging of brain chemistry bolsters characterization of dementias". Diagnostic Imaging.
- Ding, Jie; Davis-Plourde, Kendra L; Sedaghat, Sanaz; Tully, Phillip J; Wang, Wanmei; Phillips, Caroline; Pase, Matthew P; Himali, Jayandra J; Gwen Windham, B; Griswold, Michael; Gottesman, Rebecca; Mosley, Thomas H; White, Lon; Guðnason, Vilmundur; Debette, Stéphanie; Beiser, Alexa S; Seshadri, Sudha; Arfan Ikram, M; Meirelles, Osorio; Tzourio, Christophe; Launer, Lenore J (November 2019). "Antihypertensive medications and risk for incident dementia and Alzheimer's disease: a meta-analysis of individual participant data from prospective cohort studies". The Lancet Neurology. 19: 61–70. doi:10.1016/S1474-4422(19)30393-X. PMID 31706889.
- Llewellyn, David J.; Kuźma, Elżbieta; Hyppönen, Elina; Langa, Kenneth M.; Littlejohns, Thomas J.; Hannon, Eilis; Lourida, Ilianna (2019-07-14). "Association of Lifestyle and Genetic Risk With Incidence of Dementia". JAMA. 322 (5): 430. doi:10.1001/jama.2019.9879. PMC 6628594. PMID 31302669.
- Fink HA, Jutkowitz E, McCarten JR, Hemmy LS, Butler M, Davila H, et al. (January 2018). "Pharmacologic Interventions to Prevent Cognitive Decline, Mild Cognitive Impairment, and Clinical Alzheimer-Type Dementia: A Systematic Review". Annals of Internal Medicine. 168 (1): 39–51. doi:10.7326/M17-1529. PMID 29255847.
- Hughes, Diarmaid; Judge, Conor; Murphy, Robert; Loughlin, Elaine; Costello, Maria; Whiteley, William; Bosch, Jackie; O’Donnell, Martin J.; Canavan, Michelle (19 May 2020). "Association of Blood Pressure Lowering With Incident Dementia or Cognitive Impairment". JAMA. 323 (19): 1934. doi:10.1001/jama.2020.4249.
- Gates, Nicola J.; Rutjes, Anne Ws; Di Nisio, Marcello; Karim, Salman; Chong, Lee-Yee; March, Evrim; Martínez, Gabriel; Vernooij, Robin Wm (27 February 2020). "Computerised cognitive training for 12 or more weeks for maintaining cognitive function in cognitively healthy people in late life". The Cochrane Database of Systematic Reviews. 2: CD012277. doi:10.1002/14651858.CD012277.pub3. ISSN 1469-493X. PMC 7045394. PMID 32104914.
- Butler M, McCreedy E, Nelson VA, Desai P, Ratner E, Fink HA, Hemmy LS, McCarten JR, Barclay TR, Brasure M, Davila H, Kane RL (January 2018). "Does Cognitive Training Prevent Cognitive Decline?: A Systematic Review". Annals of Internal Medicine. 168 (1): 63–68. doi:10.7326/M17-1531. PMID 29255842.
- Lampit A, Hallock H, Valenzuela M (November 2014). "Computerized cognitive training in cognitively healthy older adults: a systematic review and meta-analysis of effect modifiers". PLoS Medicine. 11 (11): e1001756. doi:10.1371/journal.pmed.1001756. PMC 4236015. PMID 25405755.
- Brasure M, Desai P, Davila H, Nelson VA, Calvert C, Jutkowitz E, Butler M, Fink HA, Ratner E, Hemmy LS, McCarten JR, Barclay TR, Kane RL (January 2018). "Physical Activity Interventions in Preventing Cognitive Decline and Alzheimer-Type Dementia: A Systematic Review". Annals of Internal Medicine. 168 (1): 30–38. doi:10.7326/M17-1528. PMID 29255839.
- Kivimäki M, Singh-Manoux A, Pentti J, Sabia S, Nyberg ST, Alfredsson L, et al. (April 2019). "Physical inactivity, cardiometabolic disease, and risk of dementia: an individual-participant meta-analysis". BMJ. 365: l1495. doi:10.1136/bmj.l1495. PMC 6468884. PMID 30995986.
- Fink HA, Jutkowitz E, McCarten JR, Hemmy LS, Butler M, Davila H, Ratner E, Calvert C, Barclay TR, Brasure M, Nelson VA, Kane RL (January 2018). "Pharmacologic Interventions to Prevent Cognitive Decline, Mild Cognitive Impairment, and Clinical Alzheimer-Type Dementia: A Systematic Review". Annals of Internal Medicine. 168 (1): 39–51. doi:10.7326/M17-1529. PMID 29255847.
- Butler M, Nelson VA, Davila H, Ratner E, Fink HA, Hemmy LS, McCarten JR, Barclay TR, Brasure M, Kane RL (January 2018). "Over-the-Counter Supplement Interventions to Prevent Cognitive Decline, Mild Cognitive Impairment, and Clinical Alzheimer-Type Dementia: A Systematic Review". Annals of Internal Medicine. 168 (1): 52–62. doi:10.7326/M17-1530. PMID 29255909.
- Schneider LS, Mangialasche F, Andreasen N, Feldman H, Giacobini E, Jones R, Mantua V, Mecocci P, Pani L, Winblad B, Kivipelto M (March 2014). "Clinical trials and late-stage drug development for Alzheimer's disease: an appraisal from 1984 to 2014". Journal of Internal Medicine. 275 (3): 251–83. doi:10.1111/joim.12191. PMC 3956752. PMID 24605808.
- Watt, Jennifer A.; Goodarzi, Zahra; Veroniki, Areti Angeliki; Nincic, Vera; Khan, Paul A.; Ghassemi, Marco; Thompson, Yuan; Tricco, Andrea C.; Straus, Sharon E. (15 October 2019). "Comparative Efficacy of Interventions for Aggressive and Agitated Behaviors in Dementia". Annals of Internal Medicine. 171 (9): 633. doi:10.7326/M19-0993.
- Vandepitte S, Van Den Noortgate N, Putman K, Verhaeghe S, Verdonck C, Annemans L (December 2016). "Effectiveness of respite care in supporting informal caregivers of persons with dementia: a systematic review". International Journal of Geriatric Psychiatry. 31 (12): 1277–88. doi:10.1002/gps.4504. PMID 27245986.
- Woods B, O'Philbin L, Farrell EM, Spector AE, Orrell M (March 2018). "Reminiscence therapy for dementia". The Cochrane Database of Systematic Reviews. 3: CD001120. doi:10.1002/14651858.CD001120.pub3. PMC 6494367. PMID 29493789.
- Vernooij-Dassen M, Draskovic I, McCleery J, Downs M (November 2011). "Cognitive reframing for carers of people with dementia". The Cochrane Database of Systematic Reviews (11): CD005318. arXiv:0706.4406. doi:10.1002/14651858.CD005318.pub2. hdl:2066/97731. PMID 22071821.
- Neal M, Barton Wright P (2003). "Validation therapy for dementia". The Cochrane Database of Systematic Reviews (3): CD001394. doi:10.1002/14651858.CD001394. PMID 12917907.
- Woods B, Aguirre E, Spector AE, Orrell M (February 2012). "Cognitive stimulation to improve cognitive functioning in people with dementia". The Cochrane Database of Systematic Reviews. 2 (2): CD005562. doi:10.1002/14651858.CD005562.pub2. PMID 22336813.
- Barker P (2003). Psychiatric and mental health nursing: the craft of caring. London: Arnold. ISBN 978-0-340-81026-2. OCLC 53373798.
- Weitzel T, Robinson S, Barnes MR, Berry TA, Holmes JM, Mercer S, et al. (2011). "The special needs of the hospitalized patient with dementia". Medsurg Nursing. 20 (1): 13–18, quiz 19. PMID 21446290.
- Cunningham C (2006). "Understanding challenging behaviour in patients with dementia". Nursing Standard. 20 (47): 42–45. doi:10.7748/ns2006.08.20.47.42.c4477. PMID 16913375.
- Dyer SM, Harrison SL, Laver K, Whitehead C, Crotty M (March 2018). "An overview of systematic reviews of pharmacological and non-pharmacological interventions for the treatment of behavioral and psychological symptoms of dementia". International Psychogeriatrics. 30 (3): 295–309. doi:10.1017/S1041610217002344. PMID 29143695.
- van der Steen JT, Smaling HJ, van der Wouden JC, Bruinsma MS, Scholten RJ, Vink AC (July 2018). "Music-based therapeutic interventions for people with dementia". The Cochrane Database of Systematic Reviews. 7: CD003477. doi:10.1002/14651858.CD003477.pub4. hdl:1874/350441. PMC 6513122. PMID 30033623.
- Eldirdiry Osman, Sara; Tischler, Victoria; Schneider, Justine (2014-11-24). "'Singing for the Brain': A qualitative study exploring the health and well-being benefits of singing for people with dementia and their carers". Dementia. 15 (6): 1328. doi:10.1177/1471301214556291. PMC 5089222. PMID 25425445. Retrieved 4 February 2020.
- "British hospitals are having a dementia-friendly makeover". The Economist. Retrieved 2018-09-19.
- Rafii MS, Aisen PS (February 2009). "Recent developments in Alzheimer's disease therapeutics". BMC Medicine. 7: 7. doi:10.1186/1741-7015-7-7. PMC 2649159. PMID 19228370.
- Lleó A, Greenberg SM, Growdon JH (2006). "Current pharmacotherapy for Alzheimer's disease". Annual Review of Medicine. 57 (1): 513–33. doi:10.1146/annurev.med.57.121304.131442. PMID 16409164.
- Bond M, Rogers G, Peters J, Anderson R, Hoyle M, Miners A, Moxham T, Davis S, Thokala P, Wailoo A, Jeffreys M, Hyde C (2012). "The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer's disease (review of Technology Appraisal No. 111): a systematic review and economic model". Health Technology Assessment. 16 (21): 1–470. doi:10.3310/hta16210. PMC 4780923. PMID 22541366.
- Rodda J, Morgan S, Walker Z (October 2009). "Are cholinesterase inhibitors effective in the management of the behavioral and psychological symptoms of dementia in Alzheimer's disease? A systematic review of randomized, placebo-controlled trials of donepezil, rivastigmine and galantamine". International Psychogeriatrics. 21 (5): 813–24. doi:10.1017/S1041610209990354. PMID 19538824.
- Gill SS, Anderson GM, Fischer HD, Bell CM, Li P, Normand SL, Rochon PA (May 2009). "Syncope and its consequences in patients with dementia receiving cholinesterase inhibitors: a population-based cohort study". Archives of Internal Medicine. 169 (9): 867–73. doi:10.1001/archinternmed.2009.43. PMID 19433698.
- AMDA – The Society for Post-Acute and Long-Term Care Medicine (February 2014), "Ten Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, AMDA – The Society for Post-Acute and Long-Term Care Medicine, archived from the original on 12 April 2015, retrieved 20 April 2015
- American Geriatrics Society. "Five Things Physicians and Patients Should Question". Choosing Wisely: An Initiative of the ABIM Foundation. Archived from the original on September 1, 2013. Retrieved August 1, 2013.
- American Psychiatric Association (September 2013), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American Psychiatric Association, archived from the original on 3 December 2013, retrieved 30 December 2013
- "Dementia: assessment, management and support for people living with dementia and their carers | Guidance and guidelines | NICE". NICE. Retrieved 18 December 2018.
- Dyer SM, Laver K, Pond CD, Cumming RG, Whitehead C, Crotty M (December 2016). "Clinical practice guidelines and principles of care for people with dementia in Australia". Australian Family Physician. 45 (12): 884–889. PMID 27903038.
- Declercq T, Petrovic M, Azermai M, Vander Stichele R, De Sutter AI, van Driel ML, Christiaens T (March 2013). "Withdrawal versus continuation of chronic antipsychotic drugs for behavioural and psychological symptoms in older people with dementia" (PDF). The Cochrane Database of Systematic Reviews. 3 (3): CD007726. doi:10.1002/14651858.CD007726.pub2. hdl:1854/LU-3109108. PMID 23543555.
- Bond M, Rogers G, Peters J, Anderson R, Hoyle M, Miners A, Moxham T, Davis S, Thokala P, Wailoo A, Jeffreys M, Hyde C (2012). "The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer's disease (review of Technology Appraisal No. 111): a systematic review and economic model". Health Technology Assessment. 16 (21): 1–470. doi:10.3310/hta16210. PMC 4780923. PMID 22541366.
- Raina P, Santaguida P, Ismaila A, Patterson C, Cowan D, Levine M, et al. (March 2008). "Effectiveness of cholinesterase inhibitors and memantine for treating dementia: evidence review for a clinical practice guideline". Annals of Internal Medicine. 148 (5): 379–97. doi:10.7326/0003-4819-148-5-200803040-00009. PMID 18316756.
- Atri A, Shaughnessy LW, Locascio JJ, Growdon JH (2008). "Long-term course and effectiveness of combination therapy in Alzheimer disease". Alzheimer Disease and Associated Disorders. 22 (3): 209–21. doi:10.1097/WAD.0b013e31816653bc. PMC 2718545. PMID 18580597.
- Jones HE, Joshi A, Shenkin S, Mead GE (July 2016). "The effect of treatment with selective serotonin reuptake inhibitors in comparison to placebo in the progression of dementia: a systematic review and meta-analysis". Age and Ageing. 45 (4): 448–56. doi:10.1093/ageing/afw053. PMID 27055878.
- Dudas, Robert; Malouf, Reem; McCleery, Jenny; Dening, Tom (2018-08-31). Cochrane Dementia and Cognitive Improvement Group (ed.). "Antidepressants for treating depression in dementia". Cochrane Database of Systematic Reviews. 8: CD003944. doi:10.1002/14651858.CD003944.pub2. PMC 6513376. PMID 30168578.
- Seitz DP, Adunuri N, Gill SS, Gruneir A, Herrmann N, Rochon P (February 2011). "Antidepressants for agitation and psychosis in dementia". The Cochrane Database of Systematic Reviews (2): CD008191. doi:10.1002/14651858.CD008191.pub2. PMID 21328305.
- McCleery J, Cohen DA, Sharpley AL (November 2016). "Pharmacotherapies for sleep disturbances in dementia". The Cochrane Database of Systematic Reviews. 11 (11): CD009178. doi:10.1002/14651858.CD009178.pub3. PMC 6464889. PMID 27851868.
- American Geriatrics Society 2012 Beers Criteria Update Expert Panel (April 2012). "American Geriatrics Society updated Beers Criteria for potentially inappropriate medication use in older adults". Journal of the American Geriatrics Society. 60 (4): 616–31. doi:10.1111/j.1532-5415.2012.03923.x. PMC 3571677. PMID 22376048.
- Lolk A, Gulmann NC (October 2006). "[Psychopharmacological treatment of behavioral and psychological symptoms in dementia]". Ugeskrift for Laeger (in Danish). 168 (40): 3429–32. PMID 17032610.
- Malouf R, Grimley Evans J (October 2008). "Folic acid with or without vitamin B12 for the prevention and treatment of healthy elderly and demented people". The Cochrane Database of Systematic Reviews (4): CD004514. doi:10.1002/14651858.CD004514.pub2. PMID 18843658.
- McGuinness B, Craig D, Bullock R, Malouf R, Passmore P (July 2014). "Statins for the treatment of dementia" (PDF). The Cochrane Database of Systematic Reviews. 7 (7): CD007514. doi:10.1002/14651858.CD007514.pub3. PMID 25004278. Archived from the original (PDF) on 2019-09-03. Retrieved 2019-09-03.
- Jongstra S, Harrison JK, Quinn TJ, Richard E (November 2016). "Antihypertensive withdrawal for the prevention of cognitive decline". The Cochrane Database of Systematic Reviews. 11: CD011971. doi:10.1002/14651858.CD011971.pub2. PMC 6465000. PMID 27802359.
- Page AT, Potter K, Clifford R, McLachlan AJ, Etherton-Beer C (October 2016). "Medication appropriateness tool for co-morbid health conditions in dementia: consensus recommendations from a multidisciplinary expert panel". Internal Medicine Journal. 46 (10): 1189–1197. doi:10.1111/imj.13215. PMC 5129475. PMID 27527376.
- Hadjistavropoulos T, Herr K, Turk DC, Fine PG, Dworkin RH, Helme R, Jackson K, Parmelee PA, Rudy TE, Lynn Beattie B, Chibnall JT, Craig KD, Ferrell B, Ferrell B, Fillingim RB, Gagliese L, Gallagher R, Gibson SJ, Harrison EL, Katz B, Keefe FJ, Lieber SJ, Lussier D, Schmader KE, Tait RC, Weiner DK, Williams J (January 2007). "An interdisciplinary expert consensus statement on assessment of pain in older persons". The Clinical Journal of Pain. 23 (1 Suppl): S1–43. doi:10.1097/AJP.0b013e31802be869. PMID 17179836.
- Shega J, Emanuel L, Vargish L, Levine SK, Bursch H, Herr K, Karp JF, Weiner DK (May 2007). "Pain in persons with dementia: complex, common, and challenging". The Journal of Pain. 8 (5): 373–78. doi:10.1016/j.jpain.2007.03.003. PMID 17485039.
- Blyth FM, Cumming R, Mitchell P, Wang JJ (July 2007). "Pain and falls in older people". European Journal of Pain. 11 (5): 564–71. doi:10.1016/j.ejpain.2006.08.001. PMID 17015026.
- Brown, C. (2009). "Pain, aging and dementia: The crisis is looming, but are we ready?". British Journal of Occupational Therapy. 72 (8): 371–75. doi:10.1177/030802260907200808.
- Herr K, Bjoro K, Decker S (February 2006). "Tools for assessment of pain in nonverbal older adults with dementia: a state-of-the-science review". Journal of Pain and Symptom Management. 31 (2): 170–92. doi:10.1016/j.jpainsymman.2005.07.001. PMID 16488350.
- Stolee P, Hillier LM, Esbaugh J, Bol N, McKellar L, Gauthier N (February 2005). "Instruments for the assessment of pain in older persons with cognitive impairment". Journal of the American Geriatrics Society. 53 (2): 319–26. doi:10.1111/j.1532-5415.2005.53121.x. PMID 15673359.
- AMDA – The Society for Post-Acute and Long-Term Care Medicine (February 2014), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, AMDA – The Society for Post-Acute and Long-Term Care Medicine, archived from the original on 13 September 2014, retrieved 10 February 2013
- AMDA – The Society for Post-Acute and Long-Term Care Medicine (February 2014), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, AMDA – The Society for Post-Acute and Long-Term Care Medicine, archived from the original on 13 September 2014, retrieved 10 February 2013, which cites:
- Teno JM, Gozalo PL, Mitchell SL, Kuo S, Rhodes RL, Bynum JP, Mor V (October 2012). "Does feeding tube insertion and its timing improve survival?". Journal of the American Geriatrics Society. 60 (10): 1918–21. doi:10.1111/j.1532-5415.2012.04148.x. PMC 3470758. PMID 23002947.
- Palecek EJ, Teno JM, Casarett DJ, Hanson LC, Rhodes RL, Mitchell SL (March 2010). "Comfort feeding only: a proposal to bring clarity to decision-making regarding difficulty with eating for persons with advanced dementia". Journal of the American Geriatrics Society. 58 (3): 580–84. doi:10.1111/j.1532-5415.2010.02740.x. PMC 2872797. PMID 20398123.
- Gillick MR, Volandes AE (June 2008). "The standard of caring: why do we still use feeding tubes in patients with advanced dementia?". Journal of the American Medical Directors Association. 9 (5): 364–67. doi:10.1016/j.jamda.2008.03.011. PMID 18519120.
- Mitchell SL, Kiely DK, Lipsitz LA (February 1997). "The risk factors and impact on survival of feeding tube placement in nursing home residents with severe cognitive impairment". Archives of Internal Medicine. 157 (3): 327–32. doi:10.1001/archinte.1997.00440240091014. PMID 9040301.
- Sampson EL, Candy B, Jones L (April 2009). "Enteral tube feeding for older people with advanced dementia". The Cochrane Database of Systematic Reviews (2): CD007209. doi:10.1002/14651858.CD007209.pub2. PMC 7182132. PMID 19370678.
- Lockett MA, Templeton ML, Byrne TK, Norcross ED (February 2002). "Percutaneous endoscopic gastrostomy complications in a tertiary-care center". The American Surgeon. 68 (2): 117–20. PMID 11842953.
- Finocchiaro C, Galletti R, Rovera G, Ferrari A, Todros L, Vuolo A, Balzola F (June 1997). "Percutaneous endoscopic gastrostomy: a long-term follow-up". Nutrition. 13 (6): 520–3. doi:10.1016/S0899-9007(97)00030-0. PMID 9263232.
- Mitchell SL, Mor V, Gozalo PL, Servadio JL, Teno JM (August 2016). "Tube Feeding in US Nursing Home Residents With Advanced Dementia, 2000–2014" (PDF). JAMA. 316 (7): 769–70. doi:10.1001/jama.2016.9374. PMC 4991625. PMID 27533163. Archived (PDF) from the original on 2017-09-21.
- Span P (29 August 2016). "The Decline of Tube Feeding for Dementia Patients". New York Times. Archived from the original on 3 September 2016. Retrieved 31 August 2016.
- Viggo Hansen N, Jørgensen T, Ørtenblad L (October 2006). "Massage and touch for dementia". The Cochrane Database of Systematic Reviews (4): CD004989. doi:10.1002/14651858.CD004989.pub2. PMC 6823223. PMID 17054228.
- Forrester LT, Maayan N, Orrell M, Spector AE, Buchan LD, Soares-Weiser K (February 2014). "Aromatherapy for dementia". The Cochrane Database of Systematic Reviews. 2 (2): CD003150. doi:10.1002/14651858.CD003150.pub2. PMID 24569873.
- van den Elsen GA, Ahmed AI, Lammers M, Kramers C, Verkes RJ, van der Marck MA, Rikkert MG (March 2014). "Efficacy and safety of medical cannabinoids in older subjects: a systematic review". Ageing Research Reviews. 14: 56–64. doi:10.1016/j.arr.2014.01.007. PMID 24509411.
- Burckhardt M, Herke M, Wustmann T, Watzke S, Langer G, Fink A (April 2016). "Omega-3 fatty acids for the treatment of dementia". The Cochrane Database of Systematic Reviews. 4: CD009002. doi:10.1002/14651858.CD009002.pub3. PMC 7117565. PMID 27063583.
- Sampson EL, Ritchie CW, Lai R, Raven PW, Blanchard MR (March 2005). "A systematic review of the scientific evidence for the efficacy of a palliative care approach in advanced dementia". International Psychogeriatrics. 17 (1): 31–40. doi:10.1017/S1041610205001018. PMID 15945590.
- Van den Block L (October 2014). "The need for integrating palliative care in ageing and dementia policies". European Journal of Public Health. 24 (5): 705–06. doi:10.1093/eurpub/cku084. PMID 24997202.
- Van Der Steen, Jenny T.; Radbruch, Lukas; Hertogh, Cees MPM; De Boer, Marike E.; Hughes, Julian C.; Larkin, Philip; Francke, Anneke L.; Jünger, Saskia; Gove, Dianne; Firth, Pam; Koopmans, Raymond TCM; Volicer, Ladislav; European Association for Palliative Care (EAPC) (March 11, 2014). "White paper defining optimal palliative care in older people with dementia: A Delphi study and recommendations from the European Association for Palliative Care". Palliative Medicine. 28 (3): 197–209. doi:10.1177/0269216313493685. PMID 23828874.
- Birch D, Draper J (May 2008). "A critical literature review exploring the challenges of delivering effective palliative care to older people with dementia" (PDF). Journal of Clinical Nursing. 17 (9): 1144–63. doi:10.1111/j.1365-2702.2007.02220.x. PMID 18416791.
- Murphy E, Froggatt K, Connolly S, O'Shea E, Sampson EL, Casey D, Devane D (December 2016). "Palliative care interventions in advanced dementia". The Cochrane Database of Systematic Reviews. 12: CD011513. doi:10.1002/14651858.CD011513.pub2. PMC 6463843. PMID 27911489.
- Mitchell G, Agnelli J (October 2015). "Person-centred care for people with dementia: Kitwood reconsidered". Nursing Standard. 30 (7): 46–50. doi:10.7748/ns.30.7.46.s47. PMID 26463810.
- Prince M, Jackson J (2009). "World Alzheimer Report 2009". Alzheimer's Disease International: 38. Archived from the original on 11 March 2012. Retrieved 11 March 2012.
- Alzheimer's Disease International (Sep 2015). "World Alzheimer Report 2015" (PDF). Retrieved 30 October 2018.
- Sadock BJ, Sadock VA (2008). "Delirium, Dementia, and Amnestic and Other Cobnitive Disorders and Mental Disorders Due to a General Medical Condition". Kaplan & Sadock's concise textbook of clinical psychiatry (3rd ed.). Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins. p. 52. ISBN 978-0-7817-8746-8.
- Berrios GE (November 1987). "Dementia during the seventeenth and eighteenth centuries: a conceptual history". Psychological Medicine. 17 (4): 829–37. doi:10.1017/S0033291700000623. PMID 3324141.
- Berchtold NC, Cotman CW (1998). "Evolution in the conceptu-alization of dementia and Alzheimer's disease: Greco-Roman period to the 1960s". Neurobiol Aging. 19: 173–89. doi:10.1016/s0197-4580(98)00052-9. PMID 9661992.
- Kolata G (June 17, 2010). "Drug Trials Test Bold Plan to Slow Alzheimer's". The New York Times. Archived from the original on April 9, 2012. Retrieved June 17, 2010.
- Katzman R (April 1976). "Editorial: The prevalence and malignancy of Alzheimer disease. A major killer". Archives of Neurology. 33 (4): 217–18. doi:10.1001/archneur.1976.00500040001001. PMID 1259639.
- "What is dementia?". Alzheimer's Association. Retrieved 6 August 2018.
Dementia is often incorrectly referred to as "senility" or "senile dementia," which reflects the formerly widespread but incorrect belief that serious mental decline is a normal part of aging.
- Taylor, Danette C. "Dementia". MedicineNet. Retrieved 6 August 2018.
Senile dementia ("senility") is a term that was once used to describe all dementias; this term is no longer used as a diagnosis.
- Brodaty H, Donkin M (29 April 2017). "Family caregivers of people with dementia". Dialogues in Clinical Neuroscience. 11 (2): 217–28. PMC 3181916. PMID 19585957.
- "National Alzheimer and Dementia Plans Planned Policies and Activities (PDF)" (PDF). London: Alzheimer's Disease International. April 2012. Archived (PDF) from the original on 2012-05-18.
- "Addressing Alzheimer's and Other Types of Dementia:Israeli National Strategy Summary Document of the Interdisciplinary, Inter-Organizational Group of Experts » Brookdale". Brookdale. Retrieved 2018-06-04.
- Boseley S (26 March 2012). "Dementia research funding to more than double to £66m by 2015". The Guardian. London. ISSN 0261-3077. OCLC 60623878. Archived from the original on 20 October 2013. Retrieved 27 April 2012.
- "British hospitals are having a dementia-friendly makeover". The Economist. Retrieved 2018-09-17.
- "Drivers with dementia a growing problem, MDs warn". CBC News, Canada. September 19, 2007. Archived from the original on October 2, 2007.
- Thompson SB (2009). "Testamentary capacity and cognitive rehabilitation: implications for head-injured and neurologically impaired individuals". Journal of Cognitive Rehabilitation. 27: 11–13.
- "Tackling dementia". Philanthropy magazine. Winter 2016. Archived from the original on 2016-02-11.
- Daly B, Thompsell A, Sharpling J, Rooney YM, Hillman L, Wanyonyi KL, White S, Gallagher JE (January 2018). "Evidence summary: the relationship between oral health and dementia". British Dental Journal. 223 (11): 846–53. doi:10.1038/sj.bdj.2017.992. PMID 29192686.
- Miklossy, J (2015). "Historic evidence to support a causal relationship between spirochetal infections and Alzheimer's disease". Frontiers in Aging Neuroscience. 7: 46. doi:10.3389/fnagi.2015.00046. PMC 4399390. PMID 25932012.
- Olsen I, Singhrao SK (2015-09-17). "Can oral infection be a risk factor for Alzheimer's disease?". Journal of Oral Microbiology. 7: 29143. doi:10.3402/jom.v7.29143. PMC 4575419. PMID 26385886.
- "Can poor oral health lead to dementia?". British Dental Journal. 223 (11): 840. December 2017. doi:10.1038/sj.bdj.2017.1064. PMID 29243693.
- Carter CJ (February 2011). "Alzheimer's disease plaques and tangles: cemeteries of a pyrrhic victory of the immune defence network against herpes simplex infection at the expense of complement and inflammation-mediated neuronal destruction". Neurochemistry International. 58 (3): 301–20. doi:10.1016/j.neuint.2010.12.003. PMID 21167244.