Polycarbonate
Polycarbonates (PC) are a group of thermoplastic polymers containing carbonate groups in their chemical structures. Polycarbonates used in engineering are strong, tough materials, and some grades are optically transparent. They are easily worked, molded, and thermoformed. Because of these properties, polycarbonates find many applications. Polycarbonates do not have a unique resin identification code (RIC) and are identified as "Other", 7 on the RIC list. Products made from polycarbonate can contain the precursor monomer bisphenol A (BPA).
| Polycarbonate | |
|---|---|
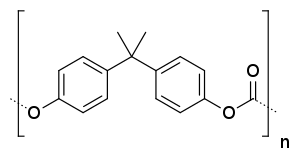 Repeating chemical structure unit of Polycarbonate made from bisphenol A  | |
| Physical properties | |
| Density (ρ) | 1.20–1.22 g/cm3 |
| Abbe number (V) | 34.0 |
| Refractive index (n) | 1.584–1.586 |
| Flammability | V0-V2 |
| Limiting oxygen index | 25–27% |
| Water absorption—Equilibrium (ASTM) | 0.16–0.35% |
| Water absorption—over 24 hours | 0.1% |
| Radiation resistance | Fair |
| Ultraviolet (1–380 nm) resistance | Fair |
| Mechanical properties | |
| Young's modulus (E) | 2.0–2.4 GPa |
| Tensile strength (σt) | 55–75 MPa |
| Elongation (ε) at break | 80–150% |
| Compressive strength (σc) | >80 MPa |
| Poisson's ratio (ν) | 0.37 |
| Hardness—Rockwell | M70 |
| Izod impact strength | 600–850 J/m |
| Notch test | 20–35 kJ/m2 |
| Abrasive resistance ASTM D1044 | 10–15 mg/1000 cycles |
| Coefficient of friction (μ) | 0.31 |
| Speed of sound | 2270 m/s |
| Thermal properties | |
| Glass transition temperature (Tg) | 147 °C (297 °F) |
| Heat deflection temperature |
|
| Vicat softening point at 50 N | 145–150 °C (293–302 °F)[1] |
| Upper working temperature | 115–130 °C (239–266 °F) |
| Lower working temperature | −40 °C (−40 °F)[2] |
| Thermal conductivity (k) at 23 °C | 0.19–0.22 W/(m·K) |
| Thermal diffusivity (a) at 25 °C | 0.144 mm²/s[3] |
| Linear thermal expansion coefficient (α) | 65–70 × 10−6/K |
| Specific heat capacity (c) | 1.2–1.3 kJ/(kg·K) |
| Electrical properties | |
| Dielectric constant (εr) at 1 MHz | 2.9 |
| Permittivity (ε) | 2.568 × 10−11 F/m |
| Relative permeability (μr) at 1 MHz | 0.866(2) |
| Permeability (μ) at 1 MHz | 1.089(2) μN/A2 |
| Dissipation factor at 1 MHz | 0.01 |
| Surface resistivity | 1015 Ω/sq |
| Volume resistivity (ρ) | 1012–1014 Ω·m |
| Chemical resistance | |
| Acids—concentrated | Poor |
| Acids—dilute | Good |
| Alcohols | Good |
| Alkalis | Good-Poor |
| Aromatic hydrocarbons | Poor |
| Greases and oils | Good-fair |
| Halogenated hydrocarbons | Good-poor |
| Halogens | Poor |
| Ketones | Poor |
| Gas permeation at 20 °C | |
| Nitrogen | 10–25 cm3·mm/(m2·day·Bar) |
| Oxygen | 70–130 cm3·mm/(m2·day·Bar) |
| Carbon dioxide | 400–800 cm3·mm/(m2·day·Bar) |
| Water vapour | 1–2 g·mm/(m2·day) @ 85%–0% RH gradient |
| Economics | |
| Price | 2.6–2.8 €/kg[4] |
Structure
Carbonate esters have planar OC(OC)2 cores, which confers rigidity. The unique O=C bond is short (1.173 Å in the depicted example), while the C-O bonds are more ether-like (the bond distances of 1.326 Å for the example depicted). Polycarbonates received their name because they are polymers containing carbonate groups (−O−(C=O)−O−). A balance of useful features, including temperature resistance, impact resistance and optical properties, positions polycarbonates between commodity plastics and engineering plastics.
Production
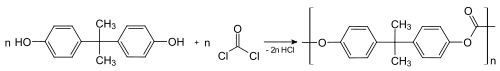
The main polycarbonate material is produced by the reaction of bisphenol A (BPA) and phosgene COCl
2. The overall reaction can be written as follows:
The first step of the synthesis involves treatment of bisphenol A with sodium hydroxide, which deprotonates the hydroxyl groups of the bisphenol A.[6]
- (HOC6H4)2CMe2 + 2 NaOH → Na2(OC6H4)2CMe2 + 2 H2O
The diphenoxide (Na2(OC6H4)2CMe2) reacts with phosgene to give a chloroformate, which subsequently is attacked by another phenoxide. The net reaction from the diphenoxide is:
- Na2(OC6H4)2CMe2 + COCl2 → 1/n [OC(OC6H4)2CMe2]n + 2 NaCl
In this way, approximately one billion kilograms of polycarbonate is produced annually. Many other diols have been tested in place of bisphenol A, e.g. 1,1-bis(4-hydroxyphenyl)cyclohexane and dihydroxybenzophenone. The cyclohexane is used as a comonomer to suppress crystallisation tendency of the BPA-derived product. Tetrabromobisphenol A is used to enhance fire resistance. Tetramethylcyclobutanediol has been developed as a replacement for BPA.[6]
An alternative route to polycarbonates entails transesterification from BPA and diphenyl carbonate:
- (HOC6H4)2CMe2 + (C6H5O)2CO → 1/n [OC(OC6H4)2CMe2]n + 2 C6H5OH
The diphenyl carbonate was derived in part from carbon monoxide, this route being greener than the phosgene method.[6]
Properties and processing
Polycarbonate is a durable material. Although it has high impact-resistance, it has low scratch-resistance. Therefore, a hard coating is applied to polycarbonate eyewear lenses and polycarbonate exterior automotive components. The characteristics of polycarbonate compare to those of polymethyl methacrylate (PMMA, acrylic), but polycarbonate is stronger and will hold up longer to extreme temperature. Polycarbonate is highly transparent to visible light, with better light transmission than many kinds of glass.
Polycarbonate has a glass transition temperature of about 147 °C (297 °F),[7] so it softens gradually above this point and flows above about 155 °C (311 °F).[8] Tools must be held at high temperatures, generally above 80 °C (176 °F) to make strain-free and stress-free products. Low molecular mass grades are easier to mold than higher grades, but their strength is lower as a result. The toughest grades have the highest molecular mass, but are much more difficult to process.
Unlike most thermoplastics, polycarbonate can undergo large plastic deformations without cracking or breaking. As a result, it can be processed and formed at room temperature using sheet metal techniques, such as bending on a brake. Even for sharp angle bends with a tight radius, heating may not be necessary. This makes it valuable in prototyping applications where transparent or electrically non-conductive parts are needed, which cannot be made from sheet metal. PMMA/Acrylic, which is similar in appearance to polycarbonate, is brittle and cannot be bent at room temperature.
Main transformation techniques for polycarbonate resins:
- extrusion into tubes, rods and other profiles including multiwall
- extrusion with cylinders (calenders) into sheets (0.5–20 mm (0.020–0.787 in)) and films (below 1 mm (0.039 in)), which can be used directly or manufactured into other shapes using thermoforming or secondary fabrication techniques, such as bending, drilling, or routing. Due to its chemical properties it is not conducive to laser-cutting.
- injection molding into ready articles
Polycarbonate may become brittle when exposed to ionizing radiation above 25 kGy (J/kg).[9]
Applications
Electronic components
Polycarbonate is mainly used for electronic applications that capitalize on its collective safety features. Being a good electrical insulator and having heat-resistant and flame-retardant properties, it is used in various products associated with electrical and telecommunications hardware. It can also serve as a dielectric in high-stability capacitors.[6] However, commercial manufacture of polycarbonate capacitors mostly stopped after sole manufacturer Bayer AG stopped making capacitor-grade polycarbonate film at the end of year 2000.[10][11]
Construction materials

The second largest consumer of polycarbonates is the construction industry, e.g. for domelights, flat or curved glazing, and sound walls.
Data storage

A major application of polycarbonate is the production of Compact Discs, DVDs, and Blu-ray Discs. These discs are produced by injection molding polycarbonate into a mold cavity that has on one side a metal stamper containing a negative image of the disc data, while the other mold side is a mirrored surface. Typical products of sheet/film production include applications in advertisement (signs, displays, poster protection).[6]
Automotive, aircraft, and security components
In the automotive industry, injection-molded polycarbonate can produce very smooth surfaces that make it well-suited for sputter deposition or evaporation deposition of aluminium without the need for a base-coat. Decorative bezels and optical reflectors are commonly made of polycarbonate. Due to its low weight and high impact resistance, polycarbonate is the dominant material for making automotive headlamp lenses. However, automotive headlamps require outer surface coatings because of its low scratch resistance and susceptibility to ultraviolet degradation (yellowing). The use of polycarbonate in automotive applications is limited to low stress applications. Stress from fasteners, plastic welding and molding render polycarbonate susceptible to stress corrosion cracking when it comes in contact with certain accelerants such as salt water and plastisol. It can be laminated to make bullet-proof "glass", although "bullet-resistant" is more accurate for the thinner windows, such as are used in bullet-resistant windows in automobiles. The thicker barriers of transparent plastic used in teller's windows and barriers in banks are also polycarbonate.
So-called "theft-proof" large plastic packaging for smaller items, which cannot be opened by hand, is uniformly made from polycarbonate.

The cockpit canopy of the Lockheed Martin F-22 Raptor jet fighter is made from a piece of high optical quality polycarbonate, and is the largest piece of its type formed in the world.[12][13]
Niche applications
Polycarbonate, being a versatile material with attractive processing and physical properties, has attracted myriad smaller applications. The use of injection molded drinking bottles, glasses and food containers is common, but the use of BPA in the manufacture of polycarbonate has stirred concerns (see Potential hazards in food contact applications), leading to development and use of "BPA-free" plastics in various formulations.

Polycarbonate is commonly used in eye protection, as well as in other projectile-resistant viewing and lighting applications that would normally indicate the use of glass, but require much higher impact-resistance. Polycarbonate lenses also protect the eye from UV light. Many kinds of lenses are manufactured from polycarbonate, including automotive headlamp lenses, lighting lenses, sunglass/eyeglass lenses, swimming goggles and SCUBA masks, and safety glasses/goggles/visors including visors in sporting helmets/masks and police riot gear (helmet visors, riot shields, etc.). Windscreens in small motorized vehicles are commonly made of polycarbonate, such as for motorcycles, ATVs, golf carts, and small airplanes and helicopters.
The light weight of polycarbonate as opposed to glass has led to development of electronic display screens that replace glass with polycarbonate, for use in mobile and portable devices. Such displays include newer e-ink and some LCD screens, though CRT, plasma screen and other LCD technologies generally still require glass for its higher melting temperature and its ability to be etched in finer detail.
As more and more governments are restricting the use of glass in pubs and clubs due to the increased incidence of glassings, polycarbonate glasses are becoming popular for serving alcohol because of their strength, durability, and glass-like feel.[14][15]
Other miscellaneous items include durable, lightweight luggage, MP3/digital audio player cases, ocarinas, computer cases, riot shields, instrument panels, tealight candle containers and food blender jars. Many toys and hobby items are made from polycarbonate parts, like fins, gyro mounts, and flybar locks in radio-controlled helicopters,[16] and transparent LEGO (ABS is used for opaque pieces).[17]
Standard Polycarbonate resins are not suitable for long term exposure to UV radiation. To overcome this the primary resin can have UV stabilisers added. These grades are sold as UV stabilized polycarbonate to injection moulding and extrusion companies. Other applications, including polycarbonate sheet, may have the anti-UV layer added as a special coating or a coextrusion for enhanced weathering resistance.
Polycarbonate is also used as a printing substrate for nameplate and other forms of industrial grade under printed products. The polycarbonate provides a barrier to wear, the elements, and fading.
Medical applications
Many polycarbonate grades are used in medical applications and comply with both ISO 10993-1 and USP Class VI standards (occasionally referred to as PC-ISO). Class VI is the most stringent of the six USP ratings. These grades can be sterilized using steam at 120 °C, gamma radiation, or by the ethylene oxide (EtO) method.[18] Dow Chemical strictly limits all its plastics with regard to medical applications.[19][20] Aliphatic polycarbonates have been developed with improved biocompatibility and degradability for nanomedicine applications.[21]
Phones
Some major smartphone manufacturers use polycarbonate. Nokia used polycarbonate in their phones starting with the N9's unibody case in 2011. This practice continued with various phones in the Lumia series. Samsung has started using polycarbonate with Galaxy S III's battery cover in 2012. This practice continues with various phones in the Galaxy series. Apple started using polycarbonate with the iPhone 5C's unibody case in 2013.
History
Polycarbonates were first discovered in 1898 by Alfred Einhorn, a German scientist working at the University of Munich.[22] However, after 30 years of laboratory research, this class of materials was abandoned without commercialization. Research resumed in 1953, when Hermann Schnell at Bayer in Uerdingen, Germany patented the first linear polycarbonate. The brand name "Makrolon" was registered in 1955.[23]
Also in 1953, and one week after the invention at Bayer, Daniel Fox at General Electric in Schenectady, New York, independently synthesized a branched polycarbonate. Both companies filed for U.S. patents in 1955, and agreed that the company lacking priority would be granted a license to the technology.[24][25]
Once patent priority was resolved, in Bayer's favor, Bayer began commercial production under the trade name Makrolon in 1958 and GE began production under the name Lexan in 1960, creating the GE Plastics division in 1973.[26]
After 1970, the brownish original polycarbonate tint was improved to "glass-clear."
Potential hazards in food contact applications
The use of polycarbonate containers for the purpose of food storage is controversial. The basis of this controversy is their hydrolysis (degradation by water, often referred to as leaching) occurring at high temperature, releases bisphenol A:
- 1/n [OC(OC6H4)2CMe2]n + H2O → (HOC6H4)2CMe2 + CO2
More than 100 studies have explored the bioactivity of bisphenol A derived from polycarbonates. Bisphenol A appeared to be released from polycarbonate animal cages into water at room temperature and it may have been responsible for enlargement of the reproductive organs of female mice.[27] However, the animal cages used in the research were fabricated from industrial grade polycarbonate, rather than FDA food grade polycarbonate.
An analysis of the literature on bisphenol A leachate low-dose effects by vom Saal and Hughes published in August 2005 seems to have found a suggestive correlation between the source of funding and the conclusion drawn. Industry-funded studies tend to find no significant effects whereas government-funded studies tend to find significant effects.[28]
Sodium hypochlorite bleach and other alkali cleaners catalyze the release of the bisphenol A from polycarbonate containers.[29][30] A chemical compatibility chart shows that polycarbonate is incompatible with ammonia and acetone because it dissolves in their presence.[31] Alcohol is one recommended organic solvent for cleaning grease and oils from polycarbonate.
Environmental impact
Disposal
Studies have shown that at temperatures above 70 °C, and high humidity, polycarbonate will hydrolyze to Bis-phenol A (BPA). This condition is similar to that observed in most incinerators. After about 30 days at 85°C / 96% RH, surface crystals are formed which for 70% consisted of BPA.[32] BPA is a compound that is currently on the list of potential environmental hazardous chemicals. It is on the watch list of many countries, such as United States and Germany.[33]
-(-OC6H4)2C(CH3)2CO-)-n + H2O (CH3)2C(C6H4OH)2 + CO2
The leaching of BPA from polycarbonate can also occur at environmental temperature and normal pH (in landfills).The amount of leaching increases as the discs get older. A study found that the decomposition of BPA in landfills (under anaerobic conditions) will not occur.[33] It will therefore be persistent in landfills. Eventually, it will find its way into water bodies and contribute to aquatic pollution.[33][34]
Photo-oxidation of polycarbonate
In the presence of UV light, oxidation of this polymer yields compounds such as ketones, phenols, o-phenoxybenzoic acid, benzyl alcohol and other unsaturated compounds. This has been suggested through kinetic and spectral studies. The yellow color formed after long exposure to sun can also be related to further oxidation of phenolic end group[35]
(OC6H4)2C(CH3)2CO )n + O2 , R* → (OC6H4)2C(CH3CH2)CO)n
This product can be further oxidized to form smaller unsaturated compounds. This can proceed via two different pathways, the products formed depends on which mechanism takes place.
Pathway A
(OC6H4)2C(CH3CH2)CO + O2, H* HO(OC6H4)OCO + CH3COCH2(OC6H4)OCO
Pathway B
(OC6H4)2C(CH3CH2)CO)n + O2, H* OCO(OC6H4)CH2OH + OCO(OC6H4)COCH3
Photo-oxidation reaction.[36]
Photo-aging reaction
Photo-aging is another degradation route for polycarbonates. Polycarbonate molecules (such as the aromatic ring) absorb UV radiation. This absorbed energy causes cleavage of covalent bonds which initiates the photo-aging process. The reaction can be propagated via side chain oxidation, ring oxidation or photo-fries rearrangement. Products formed include phenyl salicylate, dihydroxybenzophenone groups, and hydroxydiphenyl ether groups.[35][37][38]
n(C16H14O3) C16H17O3 + C13H10O3
Polycarbonate Phenyl salicylate 2,2-dihydroxybenzophenone
Thermal degradation
Waste polycarbonate will degrade at high temperatures to form solid, liquid and gaseous pollutants. A study showed that the products were about 40–50 wt.% liquid, 14–16 wt.% gases, while 34–43 wt.% remained as solid residue. Liquid products contained mainly phenol derivatives (∼75wt.%) and bisphenol (∼10wt.%) also present.[37] Therefore, burning of these discs is also not a viable method of disposal. Polycarbonate, however, can be safely recycled as a carbon source in steel-making industry.[39]
Phenol derivatives are environmental pollutants, classified as volatile organic compounds (VOC). Studies show they are likely to facilitate ground level ozone formation and increase photo-chemical smog.[40] In aquatic bodies, they can potentially accumulate in organisms. They are persistent in landfills, do not readily evaporate and would remain in the atmosphere.[41]
Effect of fungi
In 2001 a species of fungus in Belize, Geotrichum candidum, was found to consume the polycarbonate found in compact discs (CD).[42] This has prospects for bioremediation.
See also
- CR-39, allyl diglycol carbonate (ADC) used for eyeglasses
- Organic electronics
- Mobile phone accessories
- Thermoplastic polyurethane
- Vapor polishing
References
- "Lexan sheet technical manual" (PDF). SABIC. 2009. Archived from the original (PDF) on 2015-03-16. Retrieved 2015-07-18.
- Parvin, M. & Williams, J. G. (1975). "The effect of temperature on the fracture of polycarbonate". Journal of Materials Science. 10 (11): 1883. Bibcode:1975JMatS..10.1883P. doi:10.1007/BF00754478.
- Blumm, J.; Lindemann, A. (2003). "Characterization of the thermophysical properties of molten polymers and liquids using the flash technique" (PDF). High Temperatures-High Pressures. 35/36 (6): 627. doi:10.1068/htjr144.
- CES Edupack 2010, Polycarbonate (PC) specs sheet
- Perez, Serge; Scaringe, Raymond P. (1987). "Crystalline features of 4,4'-isopropylidenediphenylbis(phenyl carbonate) and conformational analysis of the polycarbonate of 2,2-bis(4-hydroxyphenyl)propane". Macromolecules. 20 (1): 68–77. Bibcode:1987MaMol..20...68P. doi:10.1021/ma00167a014.
- Volker Serini "Polycarbonates" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2000. doi:10.1002/14356007.a21_207
- Answers to Common Questions about Bayer Polycarbonate Resins. bayermaterialsciencenafta.com
- "Polycarbonate". city plastics. Archived from the original on 2018-10-16. Retrieved 2013-12-18.
- David W. Plester B.SC. A.R.I.C. (1973). "The Effects of Radiation Sterilization on Plastics" (PDF). p. 9. Archived (PDF) from the original on July 12, 2018. Retrieved 2016-04-22.
Polycarbonate can satisfactorily be given a single-dose sterilization exposure (22) but tends to become brittle much above 2.5 Mrad.
- "Film". execpc.com.
- "WIMA". wima.com. Archived from the original on June 12, 2017.
- Egress technicians keep raptor pilots covered. Pacaf.af.mil. Retrieved on 2011-02-26.
- F-22 Cockpit. Globalsecurity.org (2008-01-21). Retrieved on 2011-02-26.
- Alcohol restrictions for violent venues. The State of Queensland (Department of Justice and Attorney-General)
- Ban on regular glass in licensed premises. The State of Queensland (Department of Justice and Attorney-General)
- "RDLohr's Clearly Superior Products" (PDF). wavelandps.com. Archived from the original (PDF) on 1 April 2010.
- Linda Jablanski (2015-03-31). "Which Plastic Material is Used in Lego Sets?". Archived from the original on 2017-03-05.CS1 maint: unfit url (link)
- Powell, Douglas G. (September 1998). "Medical Applications of Polycarbonate". Medical Plastics and Biomaterials Magazine. Archived from the original on 23 February 1999.
- "Dow Plastics Medical Application Policy". Plastics.dow.com. Archived from the original on February 9, 2010.
- "Makrolon Polycarbonate Biocompatibility Grades". Archived from the original on 2013-04-10. Retrieved 2007-04-14.
- Chan, Julian M. W.; Ke, Xiyu; Sardon, Haritz; Engler, Amanda C.; Yang, Yi Yan; Hedrick, James L. (2014). "Chemically Modifiable N-Heterocycle-Functionalized Polycarbonates as a Platform for Diverse Smart Biomimetic Nanomaterials". Chemical Science. 5 (8): 3294–3300. doi:10.1039/C4SC00789A.
- "Polycarbonate (PC)". UL Prospector. Retrieved 5 May 2014.
- Philip Kotler; Waldemar Pfoertsch (17 May 2010). Ingredient Branding: Making the Invisible Visible. Springer Science & Business Media. pp. 205–. ISBN 978-3-642-04214-0.
- "Polycarbonate is Polyfunctional". Chemical Institute of Canada. Archived from the original on 5 May 2014. Retrieved 5 May 2014.
- Jerome T. Coe (27 August 2010). "Lexan Polycarbonate: 1953–1968". Unlikely Victory: How General Electric Succeeded in the Chemical Industry. John Wiley & Sons. pp. 71–77. ISBN 978-0-470-93547-7.
- "General Electric to Sell Plastics Division". NY Times. 2007-05-22. Retrieved 2020-07-21.
- Howdeshell, KL; Peterman PH; Judy BM; Taylor JA; Orazio CE; Ruhlen RL; Vom Saal FS; Welshons WV (2003). "Bisphenol A is released from used polycarbonate animal cages into water at room temperature". Environmental Health Perspectives. 111 (9): 1180–7. doi:10.1289/ehp.5993. PMC 1241572. PMID 12842771.
- vom Saal FS, Hughes C (2005). "An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment". Environ. Health Perspect. 113 (8): 926–33. doi:10.1289/ehp.7713. PMC 1280330. PMID 16079060.
- Hunt, PA; Kara E. Koehler; Martha Susiarjo; Craig A. Hodges; Arlene Ilagan; Robert C. Voigt; Sally Thomas; Brian F. Thomas; Terry J. Hassold (2003). "Bisphenol A Exposure Causes Meiotic Aneuploidy in the Female Mouse". Current Biology. 13 (7): 546–553. doi:10.1016/S0960-9822(03)00189-1. PMID 12676084.
- Koehler, KE; Robert C. Voigt; Sally Thomas; Bruce Lamb; Cheryl Urban; Terry Hassold; Patricia A. Hunt (2003). "When disaster strikes: rethinking caging materials". Lab Animal. 32 (4): 24–27. doi:10.1038/laban0403-24. PMID 19753748. Archived from the original on 2009-07-06. Retrieved 2008-05-06.
- "Cloudtops – Greenhouses, Sunrooms, Misting, Fogging, Shade – Macrolux Polycarbonate". cloudtops.com. Archived from the original on 2010-07-27. Retrieved 2010-05-16.
- Bair, H. E.; Falcone, D. R.; Hellman, M. Y.; Johnson, G. E.; Kelleher, P. G. (1981-06-01). "Hydrolysis of polycarbonate to yield BPA". Journal of Applied Polymer Science. 26 (6): 1777. doi:10.1002/app.1981.070260603.
- Morin, Nicolas; Arp, Hans Peter H.; Hale, Sarah E. (2015). "Bisphenol A in Solid Waste Materials, Leachate Water, and Air Particles from Norwegian Waste-Handling Facilities: Presence and Partitioning Behavior". Environmental Science & Technology. 49 (13): 7675–7683. doi:10.1021/acs.est.5b01307. PMID 26055751.
- Chin, Yu-Ping; Miller, Penney L.; Zeng, Lingke; Cawley, Kaelin; Weavers, Linda K. (2004). "Photosensitized Degradation of Bisphenol A by Dissolved Organic Matter †". Environmental Science & Technology. pp. 5888–5894. doi:10.1021/es0496569.
- T., Chow, Jimmy (2007-08-06). "Environmental assessment for bisphenol-a and polycarbonate".
- Carroccio, Sabrina; Puglisi, Concetto; Montaudo, Giorgio (2002). "Mechanisms of Thermal Oxidation of Poly(bisphenol A carbonate)". Macromolecules. 35 (11): 4297–4305. doi:10.1021/ma012077t.
- Collin, S.; Bussière, P. -O.; Thérias, S.; Lambert, J. -M.; Perdereau, J.; Gardette, J. -L. (2012-11-01). "Physicochemical and mechanical impacts of photo-ageing on bisphenol a polycarbonate". Polymer Degradation and Stability. 97 (11): 2284–2293. doi:10.1016/j.polymdegradstab.2012.07.036.
- Tjandraatmadja, G. F.; Burn, L. S.; Jollands, M. J. (1999). "The effects of ultraviolet radiation on polycarbonate glazing" (PDF).
- Assadi, M. Hussein N.; Sahajwalla, V. (2014). "Recycling End-of-Life Polycarbonate in Steelmaking: Ab Initio Study of Carbon Dissolution in Molten Iron". Ind. Eng. Chem. Res. 53 (10): 3861–3864. doi:10.1021/ie4031105.
- "Pollution Database". pollution.unibuc.ro. Archived from the original on 2017-12-29. Retrieved 2016-11-14.
- "Pollutant Fact Sheet". apps.sepa.org.uk. Retrieved 2016-11-14.
- Bosch, Xavier (2001-06-27). "Fungus eats CD". Nature News. doi:10.1038/news010628-11.
External links
| Wikimedia Commons has media related to Polycarbonate. |