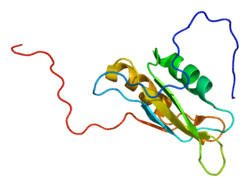
CUGBP1
CUG triplet repeat, RNA binding protein 1, also known as CUGBP1, is a protein which in humans is encoded by the CUGBP1 gene.[3][4][5]
Function
Members of the CELF/BRUNOL protein family contain two N-terminal RNA recognition motif (RRM) domains, one C-terminal RRM domain, and a divergent segment of 160-230 aa between the second and third RRM domains. Members of this protein family regulate pre-mRNA alternative splicing and may also be involved in mRNA editing, and translation. This gene may play a role in myotonic dystrophy type 1 (DM1) via interactions with the dystrophia myotonica-protein kinase (DMPK) gene. Alternative splicing results in multiple transcript variants encoding different isoforms.[3]
mRNA degradation factor
It is estimated that 5 to 8% of human mRNAs are unstable because of mRNA instability elements in their 3' untranslated regions (3'UTR).[6] A number of such elements have been called AU-rich elements (AREs). It is now known that AREs are binding sites for RNA-binding proteins that target mRNAs to rapid degradation. However, only few of the proteins reported to bind AREs were demonstrated to play a role in mRNA degradation. A shared feature of these proteins is to bind only to a subclass of the known AREs that contain the pentamer AUUUA. A convergent effort of several research teams now adds CUGBP1 (CUG binding protein 1) to the short list of ARE-Binding proteins that control mRNA stability, with the peculiarity that it binds to non-AUUUA AREs. CUGBP1 has been involved both as a key regulator of human myotonic dystrophy 1 (DM1) and more recently as a regulator of human papilloma virus mRNA expression.[7]
Evidence for CUGBP1 acting as a RNA degradation factor came first from the Xenopus model. Xenopus CUGBP1 (xCUGBP1, formerly known as EDEN-BP) was identified in 1998[8] for its ability to bind specifically to a GU-rich element (Embryonic deadenylation element EDEN) located in the 3'UTRs of some mRNAs that are rapidly deadenylated and translationally repressed after fertilization in early development. Because deadenylation is often the rate limiting step of mRNA degradation the enhancement of deadenylation increases mRNA turnover.[9]
Human CUGBP1 (hCUGBP1) had been previously identified by Timchenko and colleagues[4] for its ability to bind to CUG repeats located in the DMPK 3'UTR. A large amount of work has since described the role of hCUGBP1 on control of alternative splicing and will not be discussed here.[10] The demonstration that hCUGBP1 is involved in the control of mRNA deadenylation and instability like xCUGBP1 came next. In mammalian cell extract as well as in xenopus egg extracts, depletion and rescue experiments showed that specific binding of CUGBP1 to the 3'UTR of mRNA is required for the targeted specific deadenylation to occur.[11] In rescue experiments in xenopus egg extracts, the recombinant human protein can replace the xenopus one making them functional homolog.[12] Furthermore, the Poly(A) ribonuclease PARN was shown to interact with CUGBP1.[13] In human cells, tethering of hCUGBP1 to a mRNA decreases its steadystate suggesting the destabilization of the mRNA.[14] The first human mRNA reported to be targeted to rapid deadenylation and degradation by CUGBP1 is the oncogene c-jun. Years ago, it was shown that the class III ARE (devoid of any AUUUA motif) of the human c-jun oncogene directed rapid deadenylation and degradation to a reporter mRNA.[15] Both xCUGBP1 and hCUGBP1 were shown to specifically bind to c-jun ARE.[11] The binding of CUGBP1 to the 3'UTR of mRNAs bearing GU-rich element would target these mRNAs for rapid deadenylation by PARN and subsequent degradation. This was recently demonstrated by siRNA-mediated knockdown of hCUGBP1 that led to stabilization of a reporter RNA bearing the c-jun UG -rich ARE.[16]
UGU(G/A) tetranucleotides are key determinants of the binding site for xCUGBP1. A SELEX approach for the identification of artificial substrate of hCUGBP1 led to the proposition that UGU containing sequences were highly favoured for binding.[17] More recently, the reappraisal of CUGBP1 binding sites on the base of a combination of the SELEX approach and
Immunoprecipitation of the CUGBP1 containing complexes has led Graindorge et al. to propose a 15 nt motif as a key determinant of CUGBP1 binding.[18] Such a motif is found in a number of unstable mRNAs in human cells[16] suggesting that they are degraded by a CUGBP1 deadenylation dependent pathway.
References
- GRCh38: Ensembl release 89: ENSG00000149187 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Entrez Gene: CUGBP1 CUG triplet repeat, RNA binding protein 1".
- Timchenko LT, Miller JW, Timchenko NA, DeVore DR, Datar KV, Lin L, Roberts R, Caskey CT, Swanson MS (November 1996). "Identification of a (CUG)n triplet repeat RNA-binding protein and its expression in myotonic dystrophy". Nucleic Acids Res. 24 (22): 4407–14. doi:10.1093/nar/24.22.4407. PMC 146274. PMID 8948631.
- Roberts R, Timchenko NA, Miller JW, Reddy S, Caskey CT, Swanson MS, Timchenko LT (November 1997). "Altered phosphorylation and intracellular distribution of a (CUG)n triplet repeat RNA-binding protein in patients with myotonic dystrophy and in myotonin protein kinase knockout mice". Proc. Natl. Acad. Sci. U.S.A. 94 (24): 13221–6. doi:10.1073/pnas.94.24.13221. PMC 24290. PMID 9371827.
- Bakheet T, Frevel M, Williams BR, Greer W, Khabar KS (January 2001). "ARED: human AU-rich element-containing mRNA database reveals an unexpectedly diverse functional repertoire of encoded proteins". Nucleic Acids Res. 29 (1): 246–54. doi:10.1093/nar/29.1.246. PMC 29778. PMID 11125104.
- Goraczniak R, Gunderson SI (January 2008). "The regulatory element in the 3'-untranslated region of human papillomavirus 16 inhibits expression by binding CUG-binding protein 1". J. Biol. Chem. 283 (4): 2286–96. doi:10.1074/jbc.M708789200. PMID 18042543.
- Paillard L, Omilli F, Legagneux V, Bassez T, Maniey D, Osborne HB (January 1998). "EDEN and EDEN-BP, a cis element and an associated factor that mediate sequence-specific mRNA deadenylation in Xenopus embryos". EMBO J. 17 (1): 278–87. doi:10.1093/emboj/17.1.278. PMC 1170378. PMID 9427761.
- Meyer S, Temme C, Wahle E (2004). "Messenger RNA turnover in eukaryotes: pathways and enzymes". Crit. Rev. Biochem. Mol. Biol. 39 (4): 197–216. doi:10.1080/10409230490513991. PMID 15596551.
- Wang GS, Kearney DL, De Biasi M, Taffet G, Cooper TA (October 2007). "Elevation of RNA-binding protein CUGBP1 is an early event in an inducible heart-specific mouse model of myotonic dystrophy". J. Clin. Invest. 117 (10): 2802–11. doi:10.1172/JCI32308. PMC 1964514. PMID 17823658.
- Paillard L, Legagneux V, Maniey D, Osborne HB (February 2002). "c-Jun ARE targets mRNA deadenylation by an EDEN-BP (embryo deadenylation element-binding protein)-dependent pathway". J. Biol. Chem. 277 (5): 3232–5. doi:10.1074/jbc.M109362200. PMID 11707455.
- Paillard L, Legagneux V, Beverley Osborne H (2003). "A functional deadenylation assay identifies human CUG-BP as a deadenylation factor". Biol. Cell. 95 (2): 107–13. doi:10.1016/S0248-4900(03)00010-8. PMID 12799066.
- Moraes KC, Wilusz CJ, Wilusz J (June 2006). "CUG-BP binds to RNA substrates and recruits PARN deadenylase". RNA. 12 (6): 1084–91. doi:10.1261/rna.59606. PMC 1464848. PMID 16601207.
- Barreau C, Watrin T, Beverley Osborne H, Paillard L (September 2006). "Protein expression is increased by a class III AU-rich element and tethered CUG-BP1". Biochem. Biophys. Res. Commun. 347 (3): 723–30. doi:10.1016/j.bbrc.2006.06.177. PMID 16843434.
- Peng SS, Chen CY, Shyu AB (April 1996). "Functional characterization of a non-AUUUA AU-rich element from the c-jun proto-oncogene mRNA: evidence for a novel class of AU-rich elements". Mol. Cell. Biol. 16 (4): 1490–9. doi:10.1128/MCB.16.4.1490. PMC 231133. PMID 8657122.
- Vlasova IA, Tahoe NM, Fan D, Larsson O, Rattenbacher B, Sternjohn JR, Vasdewani J, Karypis G, Reilly CS, Bitterman PB, Bohjanen PR (February 2008). "Conserved GU-Rich Elements Mediate mRNA Decay by Binding to CUG-Binding Protein 1". Mol. Cell. 29 (2): 263–70. doi:10.1016/j.molcel.2007.11.024. PMC 2367162. PMID 18243120.
- Marquis J, Paillard L, Audic Y, Cosson B, Danos O, Le Bec C, Osborne HB (December 2006). "CUG-BP1/CELF1 requires UGU-rich sequences for high-affinity binding". Biochem. J. 400 (2): 291–301. doi:10.1042/BJ20060490. PMC 1652823. PMID 16938098.
- Graindorge A, Le Tonquèze O, Thuret R, Pollet N, Osborne HB, Audic Y (April 2008). "Identification of CUG-BP1/EDEN-BP target mRNAs in Xenopus tropicalis". Nucleic Acids Res. 36 (6): 1861–70. doi:10.1093/nar/gkn031. PMC 2330240. PMID 18267972.
External links
- Human CELF1 genome location and CELF1 gene details page in the UCSC Genome Browser.
Further reading
- Timchenko LT, Timchenko NA, Caskey CT, Roberts R (1996). "Novel proteins with binding specificity for DNA CTG repeats and RNA CUG repeats: implications for myotonic dystrophy". Hum. Mol. Genet. 5 (1): 115–21. doi:10.1093/hmg/5.1.115. PMID 8789448.
- Bhagwati S, Ghatpande A, Leung B (1996). "Identification of two nuclear proteins which bind to RNA CUG repeats: significance for myotonic dystrophy". Biochem. Biophys. Res. Commun. 228 (1): 55–62. doi:10.1006/bbrc.1996.1615. PMID 8912635.
- Timchenko LT, Miller JW, Timchenko NA, et al. (1997). "Identification of a (CUG)n triplet repeat RNA-binding protein and its expression in myotonic dystrophy". Nucleic Acids Res. 24 (22): 4407–14. doi:10.1093/nar/24.22.4407. PMC 146274. PMID 8948631.
- Roberts R, Timchenko NA, Miller JW, et al. (1998). "Altered phosphorylation and intracellular distribution of a (CUG)n triplet repeat RNA-binding protein in patients with myotonic dystrophy and in myotonin protein kinase knockout mice". Proc. Natl. Acad. Sci. U.S.A. 94 (24): 13221–6. doi:10.1073/pnas.94.24.13221. PMC 24290. PMID 9371827.
- Michalowski S, Miller JW, Urbinati CR, et al. (1999). "Visualization of double-stranded RNAs from the myotonic dystrophy protein kinase gene and interactions with CUG-binding protein". Nucleic Acids Res. 27 (17): 3534–42. doi:10.1093/nar/27.17.3534. PMC 148598. PMID 10446244.
- Timchenko NA, Welm AL, Lu X, Timchenko LT (1999). "CUG repeat binding protein (CUGBP1) interacts with the 5' region of C/EBPbeta mRNA and regulates translation of C/EBPbeta isoforms". Nucleic Acids Res. 27 (22): 4517–25. doi:10.1093/nar/27.22.4517. PMC 148737. PMID 10536163.
- Good PJ, Chen Q, Warner SJ, Herring DC (2000). "A family of human RNA-binding proteins related to the Drosophila Bruno translational regulator". J. Biol. Chem. 275 (37): 28583–92. doi:10.1074/jbc.M003083200. PMID 10893231.
- Timchenko NA, Cai ZJ, Welm AL, et al. (2001). "RNA CUG repeats sequester CUGBP1 and alter protein levels and activity of CUGBP1". J. Biol. Chem. 276 (11): 7820–6. doi:10.1074/jbc.M005960200. PMID 11124939.
- Ladd AN, Charlet N, Cooper TA (2001). "The CELF Family of RNA Binding Proteins Is Implicated in Cell-Specific and Developmentally Regulated Alternative Splicing". Mol. Cell. Biol. 21 (4): 1285–96. doi:10.1128/MCB.21.4.1285-1296.2001. PMC 99581. PMID 11158314.
- Timchenko NA, Iakova P, Cai ZJ, et al. (2001). "Molecular Basis for Impaired Muscle Differentiation in Myotonic Dystrophy". Mol. Cell. Biol. 21 (20): 6927–38. doi:10.1128/MCB.21.20.6927-6938.2001. PMC 99869. PMID 11564876.
- Takahashi N, Sasagawa N, Usuki F, et al. (2002). "Coexpression of the CUG-binding protein reduces DM protein kinase expression in COS cells". J. Biochem. 130 (5): 581–7. doi:10.1093/oxfordjournals.jbchem.a003022. PMID 11686919.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Paillard L, Legagneux V, Beverley Osborne H (2003). "A functional deadenylation assay identifies human CUG-BP as a deadenylation factor". Biol. Cell. 95 (2): 107–13. doi:10.1016/S0248-4900(03)00010-8. PMID 12799066.
- Ebralidze A, Wang Y, Petkova V, et al. (2004). "RNA leaching of transcription factors disrupts transcription in myotonic dystrophy". Science. 303 (5656): 383–7. doi:10.1126/science.1088679. PMID 14657503.
- Ota T, Suzuki Y, Nishikawa T, et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
- Hillman RT, Green RE, Brenner SE (2005). "An unappreciated role for RNA surveillance". Genome Biol. 5 (2): R8. doi:10.1186/gb-2004-5-2-r8. PMC 395752. PMID 14759258.
- Baldwin BR, Timchenko NA, Zahnow CA (2004). "Epidermal Growth Factor Receptor Stimulation Activates the RNA Binding Protein CUG-BP1 and Increases Expression of C/EBPβ-LIP in Mammary Epithelial Cells". Mol. Cell. Biol. 24 (9): 3682–91. doi:10.1128/MCB.24.9.3682-3691.2004. PMC 387752. PMID 15082764.
- Watanabe T, Takagi A, Sasagawa N, et al. (2004). "Altered expression of CUG binding protein 1 mRNA in myotonic dystrophy 1: possible RNA-RNA interaction". Neurosci. Res. 49 (1): 47–54. doi:10.1016/j.neures.2004.01.008. PMID 15099703.
- Gerhard DS, Wagner L, Feingold EA, et al. (2004). "The Status, Quality, and Expansion of the NIH Full-Length cDNA Project: The Mammalian Gene Collection (MGC)". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928. PMID 15489334.
- Dansithong W, Paul S, Comai L, Reddy S (2005). "MBNL1 is the primary determinant of focus formation and aberrant insulin receptor splicing in DM1". J. Biol. Chem. 280 (7): 5773–80. doi:10.1074/jbc.M410781200. PMID 15546872.