Technetium(IV) chloride
Technetium(IV) chloride is the chemical compound composed of technetium and chlorine with the formula TcCl4. It was discovered in 1957 as the first binary halide of technetium. It is the highest oxidation binary chloride of technetium that has been isolated in the solid-state. It is volatile at elevated temperatures and its volatility has been used for separating technetium from other metal chlorides.[2] Colloidal solutions of technetium(IV) chloride are oxidized to form Tc(VII) ions when exposed to gamma rays. [3]
 | |
| Names | |
|---|---|
| IUPAC name
Technetium(IV) chloride | |
| Other names
Technetium tetrachloride, Technetium chloride | |
| Identifiers | |
| Properties | |
| TcCl4 | |
| Molar mass | 239.718 g/mol |
| Boiling point | 300 °C (572 °F; 573 K)[1] |
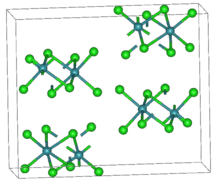
| Structure | |
| Orthorhombic, oP40 | |
| Pbca, No. 61 | |
a = 0.603 nm, b = 1.165 nm, c = 1.406 nm α = 90°, β = 90°, γ = 90° | |
| Related compounds | |
Other anions |
Technetium(VI) fluoride |
Other cations |
Manganese(II) chloride Rhenium(V) chloride Ruthenium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Technetium tetrachloride can be synthesized from the reaction of Cl2(g) with technetium metal at elevated temperatures between 300-500 °C:[4]
Tc + 2 Cl2 → TcCl4
Technetium tetrachloride has also been prepared from the reaction of ditechnetium heptoxide with carbon tetrachloride in a bomb reaction vessel at elevated temperature and pressure:
Tc2O7 + 7 CCl4 → 2 TcCl4 + 7 COCl2 + 3 Cl2
At 450 °C under vacuum, TcCl4 decomposes to TcCl3 and TcCl2.
Technetium trichloride
Technetium trichloride TcCl3 was reported in 2010. It was prepared from ditechnetium(III) tetraacetate dichloride and HCl(g) at 300 °C as a black solid. Its structure consists of triangular Tc3Cl9 units with C3V symmetry, with each Tc atom coordinated to two Tc neighbors and five chloride ligands (Tc-Tc bond length 2.44 angstrom). The Tc-Tc distances are indicative of double bonded Tc atoms. Tc3Cl9 is isostructural to its rhenium homologue.[5]
References
- Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- Schwochau, Klaus (2000). Technetium. Wiley-VCH. p. 67. ISBN 978-3-527-29496-1.
- Fattahi, M.; Vichot, L.; Poineau, F.; Houée-Levin, C.; Grambow, B. (2005). "Speciation of technetium(IV) chloride under gamma irradiation". Radiochimica Acta. 93 (7): 409–413. doi:10.1524/ract.2005.93.7.409.
- Johnstone, Erik V.; Poineau, Frederic; Forster, Paul M.; Ma, Longzou; Hartmann, Thomas; Cornelius, Andrew; Antonio, Daniel; Sattelberger, Alfred P.; Czerwinski, Kenneth R. (2012-07-09). "Technetium Tetrachloride Revisited: A Precursor to Lower-Valent Binary Technetium Chlorides". Inorganic Chemistry. 51 (15): 8462–8467. doi:10.1021/ic301011c. PMID 22775538.
- Poineau, Frederic; Johnstone, Erik V.; Weck, Philippe F.; Kim, Eunja; Forster, Paul M.; Scott, Brian L.; Sattelberger, Alfred P.; Czerwinski, Kenneth R. (2010). "Synthesis and Structure of Technetium Trichloride". Journal of the American Chemical Society. 132 (45): 15864–5. doi:10.1021/ja105730e. PMID 20977207.