Flavonoid biosynthesis
Flavonoids are synthesized by the phenylpropanoid metabolic pathway in which the amino acid phenylalanine is used to produce 4-coumaroyl-CoA.[1] This can be combined with malonyl-CoA to yield the true backbone of flavonoids, a group of compounds called chalcones, which contain two phenyl rings. Conjugate ring-closure of chalcones results in the familiar form of flavonoids, the three-ringed structure of a flavone. The metabolic pathway continues through a series of enzymatic modifications to yield flavanones → dihydroflavonols → anthocyanins. Along this pathway, many products can be formed, including the flavonols, flavan-3-ols, proanthocyanidins (tannins) and a host of other various polyphenolics.
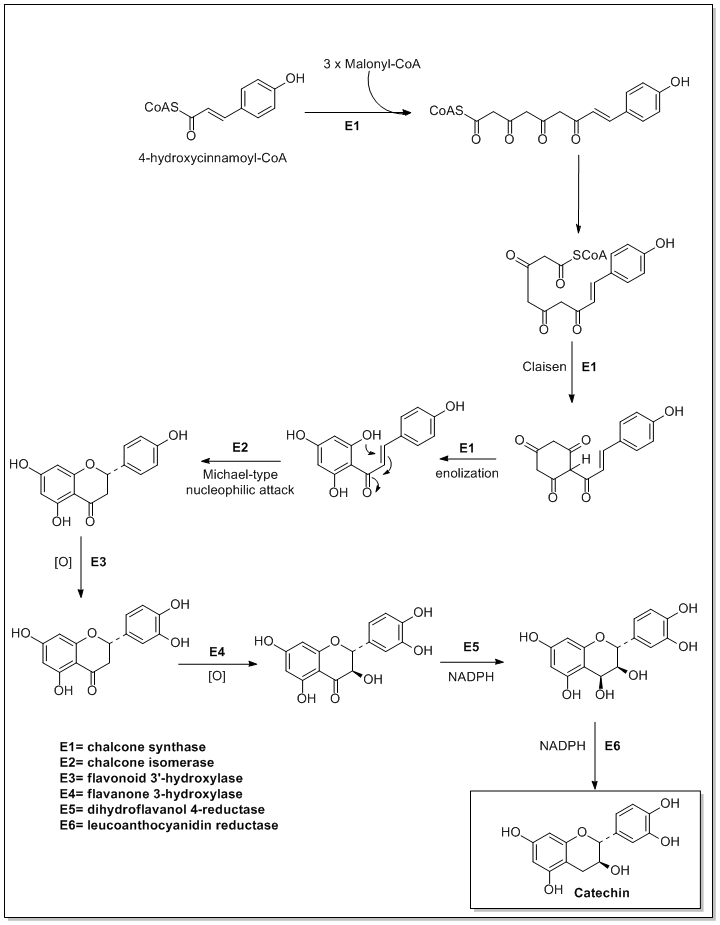
Flavanoids can possess chiral carbons. Methods of analysis should take this element into account[2] especially regarding bioactivity or enzyme stereospecificity.[3]
Enzymes
The biosynthesis of flavonoids involves several enzymes.
- Anthocyanidin reductase
- Chalcone isomerase
- Dihydrokaempferol 4-reductase
- Flavone synthase
- Flavonoid 3'-monooxygenase
- Flavonol synthase
- Flavanone 3-dioxygenase
- Flavanone 4-reductase
- Leucoanthocyanidin reductase
- Leucocyanidin oxygenase
- Naringenin-chalcone synthase
Methylation
Glycosylation
Further acetylations
References
- Ververidis Filippos, F; Trantas Emmanouil; Douglas Carl; Vollmer Guenter; Kretzschmar Georg; Panopoulos Nickolas (October 2007). "Biotechnology of flavonoids and other phenylpropanoid-derived natural products. Part I: Chemical diversity, impacts on plant biology and human health". Biotechnology Journal. 2 (10): 1214–34. doi:10.1002/biot.200700084. PMID 17935117.
- Methods of analysis and separation of chiral flavonoids. Jaime A. Yáñeza, Preston K. Andrewsb and Neal M. Journal of Chromatography B, Volume 848, Issue 2, 1 April 2007, Pages 159-181
- A theoretical study of the conformational behavior and electronic structure of taxifolin correlated with the free radical-scavenging activity. Patrick Trouillas, Catherine Fagnère, Roberto Lazzaroni, Claude Calliste, Abdelghafour Marfak and Jean-Luc Duroux, Food Chemistry, Volume 88, Issue 4, December 2004, Pages 571-582