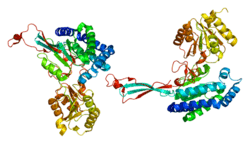Aldehyde dehydrogenase 18 family, member A1
Delta-1-pyrroline-5-carboxylate synthetase (P5CS) is an enzyme that in humans is encoded by the ALDH18A1 gene.[5][6] This gene is a member of the aldehyde dehydrogenase family and encodes a bifunctional ATP- and NADPH-dependent mitochondrial enzyme with both gamma-glutamyl kinase and gamma-glutamyl phosphate reductase activities. The encoded protein catalyzes the reduction of glutamate to delta1-pyrroline-5-carboxylate, a critical step in the de novo biosynthesis of proline, ornithine and arginine. Mutations in this gene lead to hyperammonemia, hypoornithinemia, hypocitrullinemia, hypoargininemia and hypoprolinemia and may be associated with neurodegeneration, cataracts and connective tissue diseases. Alternatively spliced transcript variants, encoding different isoforms, have been described for this gene.[6]
Structure
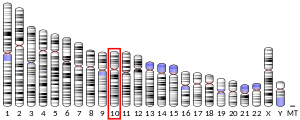
P5CS consists of two domains: gamma-glutamyl kinase and gamma-glutamyl phosphate reductase, each of which are used to complete the two steps to create ornithine and proline. The gamma-glutamyl kinase domain employs a 367-residue chain that folds into an N-terminal amino acid kinase domain, responsible for catalysis and proline inhibition, and a C-terminal PUA RNA binding domain. This enzyme is also a tetramer formed by two dimers, and the monomers and dimers are assembled in a manner that allows the amino acid kinase active pockets to be alternatively oriented in the tetramer.[7] The short version (P5CS.short) and the long version (P5CS.long) of PC5S are two isoforms of this enzyme which differs by the addition of two amino acids in the long form and with an extra 6-bp insert following bp+711. This slight difference creates a dramatic difference in how they are affected by the inhibition of ornithine.[8] The ALDH18A1 gene spans 15 kb, is mapped on 10q24.3, and has an exon count of 18.[6][9]
Function
P5CS catalyzes the phosphorylation- and reduction-conversion of glutamate to Delta-1-pyrroline-5-carboxylate (P5C). This occurs through a process in which glutamate is converted into gamma-glutamyl phosphate in the gamma-glutamyl kinase domain and then the gamma-glutamyl phosphate is the made into gamma-glutamic semi-aldehyde in the gamma-glutamyl phosphate reductase domain. The gamma-glutamic semi-aldehyde is in tautomeric equilibrium with P5C and it is the obligatory intermediate in the interconversions of proline, ornithine, and glutamate.[9] The two isoforms (PC5S.short and PC5S.long) are both involved in different activities as well. The short version has high activity in the gut and is a main participant in the biosynthesis of arginine. The long version of PC5S is expressed in various tissues and is significant for its ability to synthesize proline from glutamate. Also, the short version is inhibited by ornithine, whereas the long version is insensitive to the amino acid.[10]
Clinical significance
Ornithine and/or arginine are key intermediates for the synthesis of urea, creatine, nitric oxide, polyamines, and protein; while proline is a major component of the connective tissue proteins, collagen and elastin. Because all three of these amino acids are a part of very significant processes, the presence of P5CS becomes an important regulator which makes sure that none of these three become deficient.[11] Therefore, a lack of P5CS, due to mutations in the ALDH18A1 gene, often leads to neurodegeneration, joint laxity, skin hyperelasticity, bilateral sub capsular cataracts, and a plethora of other complications associated with impaired proline and ornithine synthesis.[10]
Interactions
P5CS has been seen to interact with:
- glutamate
- gamma-glutamyl phosphate
- ornithine
References
- GRCh38: Ensembl release 89: ENSG00000059573 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000025007 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Liu G, Maunoury C, Kamoun P, Aral B (Oct 1996). "Assignment of the human gene encoding the delta 1-pyrroline-5-carboxylate synthetase (P5CS) to 10q24.3 by in situ hybridization". Genomics. 37 (1): 145–6. doi:10.1006/geno.1996.0535. PMID 8921385.
- "Entrez Gene: ALDH18A1 aldehyde dehydrogenase 18 family, member A1".
- Pérez-Arellano I, Carmona-Álvarez F, Gallego J, Cervera J (Dec 2010). "Molecular mechanisms modulating glutamate kinase activity. Identification of the proline feedback inhibitor binding site". Journal of Molecular Biology. 404 (5): 890–901. doi:10.1016/j.jmb.2010.10.019. PMID 20970428.
- Hu CA, Lin WW, Obie C, Valle D (Mar 1999). "Molecular enzymology of mammalian Delta1-pyrroline-5-carboxylate synthase. Alternative splice donor utilization generates isoforms with different sensitivity to ornithine inhibition". The Journal of Biological Chemistry. 274 (10): 6754–62. doi:10.1074/jbc.274.10.6754. PMID 10037775.
- Hu CA, Khalil S, Zhaorigetu S, Liu Z, Tyler M, Wan G, Valle D (Nov 2008). "Human Delta1-pyrroline-5-carboxylate synthase: function and regulation". Amino Acids. 35 (4): 665–72. doi:10.1007/s00726-008-0075-0. PMC 2707934. PMID 18401542.
- Baumgartner MR, Hu CA, Almashanu S, Steel G, Obie C, Aral B, Rabier D, Kamoun P, Saudubray JM, Valle D (Nov 2000). "Hyperammonemia with reduced ornithine, citrulline, arginine and proline: a new inborn error caused by a mutation in the gene encoding delta(1)-pyrroline-5-carboxylate synthase". Human Molecular Genetics. 9 (19): 2853–8. doi:10.1093/hmg/9.19.2853. PMID 11092761.
- Martinelli D, Häberle J, Rubio V, Giunta C, Hausser I, Carrozzo R, Gougeard N, Marco-Marín C, Goffredo BM, Meschini MC, Bevivino E, Boenzi S, Colafati GS, Brancati F, Baumgartner MR, Dionisi-Vici C (Sep 2012). "Understanding pyrroline-5-carboxylate synthetase deficiency: clinical, molecular, functional, and expression studies, structure-based analysis, and novel therapy with arginine". Journal of Inherited Metabolic Disease. 35 (5): 761–76. doi:10.1007/s10545-011-9411-8. PMID 22170564.
External links
- Human ALDH18A1 genome location and ALDH18A1 gene details page in the UCSC Genome Browser.
Further reading
- Maruyama K, Sugano S (Jan 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Aral B, Schlenzig JS, Liu G, Kamoun P (Mar 1996). "Database cloning human delta 1-pyrroline-5-carboxylate synthetase (P5CS) cDNA: a bifunctional enzyme catalyzing the first 2 steps in proline biosynthesis". Comptes Rendus de l'Académie des Sciences, Série III. 319 (3): 171–8. PMID 8761662.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (Oct 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Hu CA, Lin WW, Obie C, Valle D (Mar 1999). "Molecular enzymology of mammalian Delta1-pyrroline-5-carboxylate synthase. Alternative splice donor utilization generates isoforms with different sensitivity to ornithine inhibition". The Journal of Biological Chemistry. 274 (10): 6754–62. doi:10.1074/jbc.274.10.6754. PMID 10037775.
- Baumgartner MR, Hu CA, Almashanu S, Steel G, Obie C, Aral B, Rabier D, Kamoun P, Saudubray JM, Valle D (Nov 2000). "Hyperammonemia with reduced ornithine, citrulline, arginine and proline: a new inborn error caused by a mutation in the gene encoding delta(1)-pyrroline-5-carboxylate synthase". Human Molecular Genetics. 9 (19): 2853–8. doi:10.1093/hmg/9.19.2853. PMID 11092761.
- Baumgartner MR, Rabier D, Nassogne MC, Dufier JL, Padovani JP, Kamoun P, Valle D, Saudubray JM (Jan 2005). "Delta1-pyrroline-5-carboxylate synthase deficiency: neurodegeneration, cataracts and connective tissue manifestations combined with hyperammonaemia and reduced ornithine, citrulline, arginine and proline". European Journal of Pediatrics. 164 (1): 31–6. doi:10.1007/s00431-004-1545-3. PMID 15517380.