Myeloperoxidase
Myeloperoxidase (MPO) is a peroxidase enzyme that in humans is encoded by the MPO gene on chromosome 17.[5] MPO is most abundantly expressed in neutrophil granulocytes (a subtype of white blood cells), and produces hypohalous acids to carry out their antimicrobial activity.[5][6] It is a lysosomal protein stored in azurophilic granules of the neutrophil and released into the extracellular space during degranulation.[7] Neutrophil myeloperoxidase has a heme pigment, which causes its green color in secretions rich in neutrophils, such as pus and some forms of mucus. The green color contributed to its outdated name verdoperoxidase.
| Myeloperoxidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 1.11.2.2 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Structure
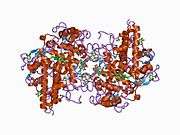
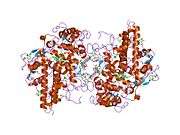
The 150-kDa MPO protein is a cationic homodimer consisting of two 15-kDa light chains and two variable-weight glycosylated heavy chains bound to a prosthetic heme group.[8][9][10] The light chains are glycosylated and contain the modified iron protoporphyrin IX active site. Together, the light and heavy chains form two identical 73-kDa monomers connected by a cystine bridge at Cys153. The protein forms a deep crevice which holds the heme group at the bottom, as well as a hydrophobic pocket at the entrance to the distal heme cavity which carries out its catalytic activity.[10]
Three isoforms have been identified, differing only in the size of the heavy chains.[8]
One of the ligands is the carbonyl group of Asp 96. Calcium-binding is important for structure of the active site because of Asp 96's close proximity to the catalytic His95 side chain.[11]
Function
MPO is a member of the XPO subfamily of peroxidases and produces hypochlorous acid (HOCl) from hydrogen peroxide (H2O2) and chloride anion (Cl−) (or hypobromous acid if Br- is present) during the neutrophil's respiratory burst. It requires heme as a cofactor. Furthermore, it oxidizes tyrosine to tyrosyl radical using hydrogen peroxide as an oxidizing agent.[8][12] Hypochlorous acid and tyrosyl radical are cytotoxic, so they are used by the neutrophil to kill bacteria and other pathogens.[13] However, this hypochlorous acid may also cause oxidative damage in host tissue. Moreover, MPO oxidation of apoA-I reduces HDL-mediated inhibition of apoptosis and inflammation.[14] In addition, MPO mediates protein nitrosylation and the formation of 3-chlorotyrosine and dityrosine crosslinks.[8]
Clinical significance
Myeloperoxidase deficiency is a hereditary deficiency of the enzyme, which predisposes to immune deficiency.[15]
Antibodies against MPO have been implicated in various types of vasculitis, most prominently three clinically and pathologically recognized forms: granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA); and eosinophilic granulomatosis with polyangiitis (EGPA). Antibodies are also known as anti-neutrophil cytoplasmic antibodies (ANCAs), though ANCAs have also been detected in staining of the perinuclear region.[16]
Recent studies have reported an association between elevated myeloperoxidase levels and the severity of coronary artery disease.[17] And Heslop et al. reported that elevated MPO levels more than doubled the risk for cardiovascular mortality over a 13-year period.[18] It has also been suggested that myeloperoxidase plays a significant role in the development of the atherosclerotic lesion and rendering plaques unstable.[19][20]
Medical uses
An initial 2003 study suggested that MPO could serve as a sensitive predictor for myocardial infarction in patients presenting with chest pain.[21] Since then, there have been over 100 published studies documenting the utility of MPO testing. The 2010 Heslop et al. study reported that measuring both MPO and CRP (C-reactive protein; a general and cardiac-related marker of inflammation) provided added benefit for risk prediction than just measuring CRP alone.[18]
Immunohistochemical staining for myeloperoxidase used to be administered in the diagnosis of acute myeloid leukemia to demonstrate that the leukemic cells were derived from the myeloid lineage. Myeloperoxidase staining is still important in the diagnosis of myeloid sarcoma, contrasting with the negative staining of lymphomas, which can otherwise have a similar appearance.[22] In the case of screening patients for vasculitis, flow cytometric assays have demonstrated comparable sensitivity to immunofluorescence tests, with the additional benefit of simultaneous detection of multiple autoantibodies relevant to vasculitis. Nonetheless, this method still requires further testing.[23]
Myeloperoxidase is the first and so far only human enzyme known to break down carbon nanotubes, allaying a concern among clinicians that using nanotubes for targeted delivery of medicines would lead to an unhealthy buildup of nanotubes in tissues.[24]
Inhibitors of MPO
Azide has been used traditionally as an MPO inhibitor, but 4-aminobenzoic acid hydrazide (4-ABH) is a more specific inhibitor of MPO.[25]
See also
References
- GRCh38: Ensembl release 89: ENSG00000005381 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000009350 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Entrez Gene: Myeloperoxidase".
- Klebanoff SJ (May 2005). "Myeloperoxidase: friend and foe". Journal of Leukocyte Biology. 77 (5): 598–625. doi:10.1189/jlb.1204697. PMID 15689384.
- Kinkade JM, Pember SO, Barnes KC, Shapira R, Spitznagel JK, Martin LE (Jul 1983). "Differential distribution of distinct forms of myeloperoxidase in different azurophilic granule subpopulations from human neutrophils". Biochemical and Biophysical Research Communications. 114 (1): 296–303. doi:10.1016/0006-291x(83)91627-3. PMID 6192815.
- "Mouse MPO EasyTestTM ELISA Kit" (PDF). Archived from the original (PDF) on 2016-03-03. Retrieved 2015-08-06.
- Mathy-Hartert M, Bourgeois E, Grülke S, Deby-Dupont G, Caudron I, Deby C, Lamy M, Serteyn D (Apr 1998). "Purification of myeloperoxidase from equine polymorphonuclear leucocytes". Canadian Journal of Veterinary Research. 62 (2): 127–32. PMC 1189459. PMID 9553712.
- Davies MJ (Jan 2011). "Myeloperoxidase-derived oxidation: mechanisms of biological damage and its prevention". Journal of Clinical Biochemistry and Nutrition. 48 (1): 8–19. doi:10.3164/jcbn.11-006FR. PMC 3022070. PMID 21297906.
- Shin K, Hayasawa H, Lönnerdal B (Mar 2001). "Mutations affecting the calcium-binding site of myeloperoxidase and lactoperoxidase". Biochemical and Biophysical Research Communications. 281 (4): 1024–9. doi:10.1006/bbrc.2001.4448. PMID 11237766.
- Heinecke JW, Li W, Francis GA, Goldstein JA (Jun 1993). "Tyrosyl radical generated by myeloperoxidase catalyzes the oxidative cross-linking of proteins". The Journal of Clinical Investigation. 91 (6): 2866–72. doi:10.1172/JCI116531. PMC 443356. PMID 8390491.
- Hampton MB, Kettle AJ, Winterbourn CC (Nov 1998). "Inside the neutrophil phagosome: oxidants, myeloperoxidase, and bacterial killing". Blood. 92 (9): 3007–17. doi:10.1182/blood.V92.9.3007. PMID 9787133.
- Shao B, Oda MN, Oram JF, Heinecke JW (Mar 2010). "Myeloperoxidase: an oxidative pathway for generating dysfunctional high-density lipoprotein". Chemical Research in Toxicology. 23 (3): 447–54. doi:10.1021/tx9003775. PMC 2838938. PMID 20043647.
- Kutter D, Devaquet P, Vanderstocken G, Paulus JM, Marchal V, Gothot A (2000). "Consequences of total and subtotal myeloperoxidase deficiency: risk or benefit ?". Acta Haematologica. 104 (1): 10–5. doi:10.1159/000041062. PMID 11111115.
- Flint SM, McKinney EF, Smith KG (Mar 2015). "Emerging concepts in the pathogenesis of antineutrophil cytoplasmic antibody-associated vasculitis". Current Opinion in Rheumatology. 27 (2): 197–203. doi:10.1097/BOR.0000000000000145. PMID 25629443.
- Zhang R, Brennan ML, Fu X, Aviles RJ, Pearce GL, Penn MS, Topol EJ, Sprecher DL, Hazen SL (Nov 2001). "Association between myeloperoxidase levels and risk of coronary artery disease". JAMA. 286 (17): 2136–42. doi:10.1001/jama.286.17.2136. PMID 11694155.
- Heslop CL, Frohlich JJ, Hill JS (Mar 2010). "Myeloperoxidase and C-reactive protein have combined utility for long-term prediction of cardiovascular mortality after coronary angiography". Journal of the American College of Cardiology. 55 (11): 1102–9. doi:10.1016/j.jacc.2009.11.050. PMID 20223364.
- Nicholls SJ, Hazen SL (Jun 2005). "Myeloperoxidase and cardiovascular disease". Arteriosclerosis, Thrombosis, and Vascular Biology. 25 (6): 1102–11. doi:10.1161/01.ATV.0000163262.83456.6d. PMID 15790935.
- Lau D, Baldus S (Jul 2006). "Myeloperoxidase and its contributory role in inflammatory vascular disease". Pharmacology & Therapeutics. 111 (1): 16–26. doi:10.1016/j.pharmthera.2005.06.023. PMID 16476484.
- Brennan ML, Penn MS, Van Lente F, Nambi V, Shishehbor MH, Aviles RJ, Goormastic M, Pepoy ML, McErlean ES, Topol EJ, Nissen SE, Hazen SL (Oct 2003). "Prognostic value of myeloperoxidase in patients with chest pain". The New England Journal of Medicine. 349 (17): 1595–604. doi:10.1056/NEJMoa035003. PMID 14573731.
- Leong A S-Y, Cooper K, Leong, FJ W-M (2003). Manual of Diagnostic Antibodies for Immunohistology. London: Greenwich Medical Media. pp. 325–326. ISBN 1-84110-100-1.CS1 maint: multiple names: authors list (link)
- Csernok E, Moosig F (Aug 2014). "Current and emerging techniques for ANCA detection in vasculitis". Nature Reviews. Rheumatology. 10 (8): 494–501. doi:10.1038/nrrheum.2014.78. PMID 24890776.
- Kagan VE, Konduru NV, Feng W, Allen BL, Conroy J, Volkov Y, Vlasova II, Belikova NA, Yanamala N, Kapralov A, Tyurina YY, Shi J, Kisin ER, Murray AR, Franks J, Stolz D, Gou P, Klein-Seetharaman J, Fadeel B, Star A, Shvedova AA (May 2010). "Carbon nanotubes degraded by neutrophil myeloperoxidase induce less pulmonary inflammation". Nature Nanotechnology. 5 (5): 354–9. doi:10.1038/nnano.2010.44. PMC 6714564. PMID 20364135. Lay summary – popsci.com.
- Kettle AJ, Gedye CA, Winterbourn CC (Jan 1997). "Mechanism of inactivation of myeloperoxidase by 4-aminobenzoic acid hydrazide". The Biochemical Journal. 321. 321 (2): 503–8. doi:10.1042/bj3210503. PMC 1218097. PMID 9020887.
External links
- Overview of all the structural information available in the PDB for UniProt: P05164 (Myeloperoxidase) at the PDBe-KB.