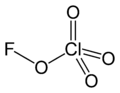
Fluorine perchlorate
Fluorine perchlorate, also called perchloryl hypofluorite is the rarely encountered chemical compound of fluorine, chlorine, and oxygen with the chemical formula ClO
4F or FOClO
3. It is an extremely unstable gas that explodes spontaneously[2] and has a penetrating odor.[3]
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Perchloryl hypofluorite | |||
| Other names
Fluorine perchlorate | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| FClO 4 | |||
| Melting point | −167.3 °C (−269.1 °F; 105.8 K) | ||
| Boiling point | −16 °C (3 °F; 257 K) | ||
| Thermochemistry | |||
Std enthalpy of formation (ΔfH⦵298) |
9 kcal/mol[1] | ||
| Hazards | |||
| Main hazards | Highly explosive gas | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Synthesis
One synthesis uses fluorine and perchloric acid,[4] though the action of ClF5 on water is another method.
Another method of synthesis involves the thermal decomposition of tetrafluoroammonium perchlorate, NF
4ClO
4, which yields very pure FClO
4 that may be manipulated and frozen without explosions.[5]
Structure
Fluorine perchlorate is not analogous to perchloric acid because the fluorine atom does not exist as a positive ion. It contains an oxygen atom in a rare oxidation state of 0 due to the electronegativity of oxygen, which is higher than that of chlorine but lower than that of fluorine.
Safety
FClO4 has a very dangerous and unpredictable series of reactions associated with it, as a covalent perchlorate (chlorine in the +7 oxidation state) and a compound featuring a very sensitive O-F single bond. Small amounts of reducing agent, such as organic compounds, can trigger explosive detonation. Products of these decomposition reactions could include oxygen halides, interhalogen compounds, and other hazardous substances.
Accidental synthesis is possible if precursors are carelessly mixed. Like similar covalent fluorides and perchlorates, it needs to be handled with extreme caution.
Reaction
FClO4 is a strong oxidant and it reacts with iodide ion:
FClO4 can also react with tetrafluoroethylene:[6]
It may be a radical addition reaction.[7]
References
- Breazeale, J. D.; MacLaren, R. O.. Thermochemistry of oxygen-fluorine bonding, United Technology Center, Sunnyvale, CA, 1963. Accession Number: AD0402889. Retrieved online from on 2009-05-21.
- Pradyot Patnaik. A comprehensive guide to the hazardous properties of chemical substances, 3rd ed., Wiley-Interscience, 2007. ISBN 0-471-71458-5
- Robert Alan Lewis. Lewis' dictionary of toxicology, CRC Press, 1998, p. 508. ISBN 1-56670-223-2
- Rohrback, G. H.; Cady, G. H. (1947). "The Preparation of Fluorine Perchlorate from Fluorine and Perchloric Acid". Journal of the American Chemical Society. 69 (3): 677–678. doi:10.1021/ja01195a063.
- Schack, C. J.; Christe, K. O. (1979). "Reactions of fluorine perchlorate with fluorocarbons and the polarity of the oxygen-fluorine bond in covalent hypofluorites". Inorganic Chemistry. 18 (9): 2619–2620. doi:10.1021/ic50199a056.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Schack, Carl J.; Christe, Karl O. (1979). "Reactions of fluorine perchlorate with fluorocarbons and the polarity of the oxygen-fluorine bond in covalent hypofluorites". Inorganic Chemistry. 18 (9): 2619. doi:10.1021/ic50199a056.
External links
- Chemist Derek Lowe's experiences with perchlorates
| HClO4 | He | ||||||||||||||||
| LiClO4 | Be(ClO4)2 | B(ClO 4)− 4 B(ClO4)3 |
ROClO3 | N(ClO4)3 NH4ClO4 NOClO4 |
O | FClO4 | Ne | ||||||||||
| NaClO4 | Mg(ClO4)2 | Al(ClO4)3 | Si | P | S | ClO− 4 ClOClO3 Cl2O7 |
Ar | ||||||||||
| KClO4 | Ca(ClO4)2 | Sc(ClO4)3 | Ti(ClO4)4 | VO(ClO4)3 VO2(ClO4) |
Cr(ClO4)3 | Mn(ClO4)2 | Fe(ClO4)3 | Co(ClO4)2, Co(ClO4)3 |
Ni(ClO4)2 | Cu(ClO4)2 | Zn(ClO4)2 | Ga(ClO4)3 | Ge | As | Se | Br | Kr |
| RbClO4 | Sr(ClO4)2 | Y(ClO4)3 | Zr(ClO4)4 | Nb(ClO4)5 | Mo | Tc | Ru | Rh(ClO4)3 | Pd(ClO4)2 | AgClO4 | Cd(ClO4)2 | In(ClO4)3 | Sn(ClO4)4 | Sb | TeO(ClO4)2 | I | Xe |
| CsClO4 | Ba(ClO4)2 | Hf(ClO4)4 | Ta(ClO4)5 | W | Re | Os | Ir | Pt | Au | Hg2(ClO4)2, Hg(ClO4)2 |
Tl(ClO4), Tl(ClO4)3 |
Pb(ClO4)2 | Bi(ClO4)3 | Po | At | Rn | |
| FrClO4 | Ra | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce(ClO4)x | Pr | Nd | Pm | Sm(ClO4)3 | Eu(ClO4)3 | Gd(ClO4)3 | Tb(ClO4)3 | Dy(ClO4)3 | Ho(ClO4)3 | Er(ClO4)3 | Tm(ClO4)3 | Yb(ClO4)3 | Lu(ClO4)3 | |||
| Ac | Th(ClO4)4 | Pa | UO2(ClO4)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||