Tungsten dichloride dioxide
Tungsten dichloride dioxide is the chemical compound with the formula WO2Cl2. It is a yellow-colored solid. It is used as a precursor to other tungsten compounds. Like other tungsten halides, WO2Cl2 is sensitive to moisture, undergoing hydrolysis.
 | |
| Names | |
|---|---|
| Other names
tungsten(VI) dioxydichloride | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.033.496 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| WO2Cl2 | |
| Molar mass | 286.749 g/mol |
| Appearance | yellow-red crystals |
| Density | 4.67 g/cm3, solid |
| Melting point | 265 °C (509 °F; 538 K) |
| Boiling point | sublimes > 350 °C in vacuo |
| decomposes | |
| Solubility | slightly soluble in ethanol |
| Structure | |
| orthorhombic | |
| Hazards | |
EU classification (DSD) (outdated) |
not listed |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
WO2Cl2 is prepared by ligand redistribution reaction from tungsten trioxide and tungsten hexachloride:
- 2 WO3 + WCl6 → 3 WO2Cl2
Using a two-zone tube furnace, a vacuum-sealed tube containing these solids is heated to 350 °C. The yellow product sublimes to the cooler end of the reaction tube. No redox occurs in this process.[1] An alternative route highlights the oxophilicity of tungsten:[2]
- WCl6 + 2 O(Si(CH3)3)2 → 3 WO2Cl2 + 4 ClSi(CH3)3
This reaction, like the preceding one, proceeds via the intermediacy of WOCl4.
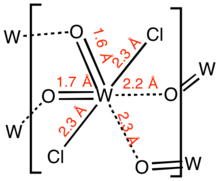
Structure
The compound is a polymer consisting of distorted octahedral W centres. The monomer is characterized by two short W-O distances, typical for a multiple W-O bond, and two long W-O distances more typical of a single or dative W-O bond.[3]
Related oxy halides
Tungsten forms a number of oxyhalides including WOCl4, WOCl3, WOCl2. The corresponding bromides (WOBr4, WOBr3, WOBr2) are also known as is WO2I2.[4]
.png)
Reactions
WO2Cl2 is a Lewis acid, forming soluble adducts of the type WO2Cl2L2, where L is a donor ligand such as bipyridine and dimethoxyethane. Such complexes often cannot be prepared by depolymerization of the inorganic solid, but are generated in situ from WOCl4.[5]
References
- Tillack, J. (1973). "Tungsten Oxyhalides". Inorg. Synth. 14: 109–122. doi:10.1002/9780470132456.ch22.
- Gibson, V. C.; Kee, T. P.; Shaw, A. (1988). "New, improved synthesis of the group 6 oxyhalides, W(O)Cl4, W(O)2Cl2 and Mo(O)2Cl2". Polyhedron. 7 (7): 579–80. doi:10.1016/S0277-5387(00)86336-6.
- Jarchow, O.; Schröder, F.; Schulz, H. "Kristallstruktur und Polytypie von WO2Cl2" Zeitschrift für anorganische und allgemeine Chemie 1968, vol. 363, p. 345ff. doi:10.1002/zaac.19683630108
- Holleman, A. F.; Wiberg, E. Inorganic Chemistry Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- K. Dreisch, C. Andersson, C. Stalhandske "Synthesis and structure of dimethoxyethane-dichlorodioxo-tungsten(VI)—a highly soluble derivative of tungsten dioxodichloride" Polyhedron 1991, volume 10, p. 2417. doi:10.1016/S0277-5387(00)86203-8