Thioketone
Thioketones (also known as thiones[1] or thiocarbonyls) are organosulfur compounds related to conventional ketones in which the oxygen has been replaced by a sulfur.[2] Instead of a structure of R2C=O, thioketones have the structure R2C=S, which is reflected by the prefix "thio-" in the name of the functional group. Unhindered alkylthioketones typically tend to form polymers or rings.[3]
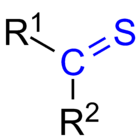
Structure and bonding
The C=S bond length of thiobenzophenone is 1.63 Å, which is comparable to 1.64 Å, the C=S bond length of thioformaldehyde, measured in the gas phase. Due to steric interactions, the phenyl groups are not coplanar and the dihedral angle SC-CC is 36°.[4]
Consistent with the double bond rule, most alkyl thioketones are unstable with respect to dimerization.[5] The energy difference between the p orbitals of sulfur and carbon is greater than that between oxygen and carbon in ketones.[6] The relative difference in energy and diffusity of the atomic orbitals of sulfur compared to carbon results in poor overlap of the orbitals and the energy gap between the HOMO and LUMO is thus reduced for C=S relative to C=O.[4] The striking blue appearance of thiobenzophenone is due to π→ π* transitions upon the absorption of light with a wavelength of 314.5 nm.[6]
Preparative methods
Thiones are usually prepared from ketones using reagents that exchange S and O atoms. A common reagent is phosphorus pentasulfide[7] and the related reagent Lawesson's reagent. Other methods uses a mixture of hydrogen chloride combined with hydrogen sulfide. Bis(trimethylsilyl)sulfide has also been employed.[3][8]
Thiobenzophenone [(C6H5)2CS] is a stable deep blue compound that dissolves readily in organic solvents. It photooxidizes in air to benzophenone and sulfur. Since its discovery, a variety of related thiones have been prepared.[9]
Thiosulfines
Thiosulfines, also called thiocarbonyl S-sulfides, are compounds with the formula R2CSS. Although superficially appearing to be cumulenes, with the linkage R2C=S=S, they are more usefully classified as 1,3-dipoles and indeed participate in 1,3-dipolar cycloadditions. Thiosulfines are proposed to exist in equilibrium with dithiiranes, three-membered CS2 rings. Thiosulfines are often invoked as intermediates in mechanistic discussions of the chemistry of thiones. For example, thiobenzophenone decomposes upon oxidation to the 1,2,4-trithiolane (Ph2C)2S3, which arises via the cycloaddition of Ph2CSS to its parent Ph2CS.[10]
See also
- Thial, for a description of thioaldehydes.
- Thioketene
- Thioacetone
References
- Campainge, E. (1 August 1946). "Thiones and Thials". Chemical Reviews. 39 (1): 1–77. doi:10.1021/cr60122a001.
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "Thioketones". doi:10.1351/goldbook.T06356
- Kuhn, N.; Verani, G. (2007). "Chalcogenone C=E compounds". Handbook of Chalcogen Chemistry: New Perspectives in Sulfur, Selenium and Tellurium. Royal Society of Chemistry. doi:10.1039/9781847557575-00107.
- Sustmann, R.; Sicking, W.; Huisgen, R. "A Computational Study of the Cycloaddition of Thiobenzophenone S-Methylide to Thiobenzophenone". J. Am. Chem. Soc. 2003, 125, 14425-14434. doi:10.1021/ja0377551
- Organosulfur Chemistry I: Topics in Current Chemistry, 1999, Volume 204/1999, 127-181, doi:10.1007/3-540-48956-8_2
- Fisera, L.; Huisgen, R.; Kalwinsch, I.; Langhals,E.; Li, X.; Mloston, G.; Polborn, K.; Rapp, J.; Sicking, W.; Sustmann, R. "New Thione Chemistry". Pure Appl. Chem., 1996, 68, 789-798. doi:10.1351/pac199668040789
- Polshettiwar, Vivek; Kaushik, M. P. (2004). "A new, efficient and simple method for the thionation of ketones to thioketones using P4S10/Al2O3". Tetrahedron Letters. 45: 6255–6257. doi:10.1016/j.tetlet.2004.06.091.
- Mcgregor, W. M.; Sherrington, D. C. (1993). "Some recent synthetic routes to thioketones and thioaldehydes". Chemical Society Reviews. 22 (3): 199. doi:10.1039/CS9932200199.
- Okazaki, R.; Tokitoh, N. (2000). "Heavy ketones, the heavier element congeners of a ketone". Accounts of Chemical Research. 33 (9): 625–630. doi:10.1021/ar980073b. PMID 10995200.
- Rolf Huisgen; J. Rapp (1997). "1,3-Dipolar Cycloadditions. 98. The Chemistry of Thiocarbonyl S-Sulfides". Tetrahedron. 53 (3): 939–960. doi:10.1016/S0040-4020(96)01068-X.
External links and further reading
- Definition of Selones in the IUPAC Gold Book
- Kroto, H.; Landsberg, B. M.; Suffolk, R. J.; Vodden, A. (1974). "The photoelectron and microwave spectra of the unstable species thioacetaldehyde, CH3CHS, and thioacetone, (CH3)2CS". Chemical Physics Letters. 29 (2): 265–269. Bibcode:1974CPL....29..265K. doi:10.1016/0009-2614(74)85029-3.