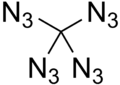
Tetraazidomethane
Tetraazidomethane is a colorless, highly explosive liquid. Its chemical structure consists of a carbon atom substituted with four azide functional groups.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Tetraazidomethane | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CN12 | |||
| Molar mass | 180.09 g/mol | ||
| Boiling point | ~165 °C (estimate) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Synthesis
It was first prepared by Klaus Banert in 2006 by reaction of trichloroacetonitrile with sodium azide.[1]
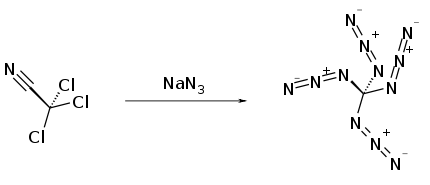
Uses
As with other polyazides, tetraazidomethane has interest as a high-energy-density material with potential uses in explosives, propellants, or fireworks.[2] Silicon tetraazide is also a known compound.
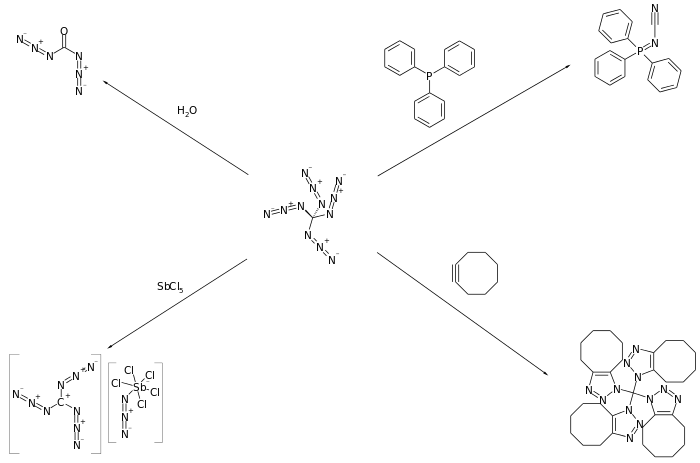
Reactions
Banert has reported that tetraazidomethane participates in a number of surprising reactions including hydrolysis, cycloaddition reactions with alkenes and alkynes, and reaction with phosphines to form phosphazenes.[1]
gollark: 50% of the KNOWN bugs.
gollark: <@341618941317349376> Steal osmarks dot tk cascade style sheeys
gollark: <@319753218592866315> utilise base65536
gollark: Prove it empirically.
gollark: Prove it. Mathematically.
References
- "The Exciting Chemistry of Tetraazidomethane", Klaus Banert, Young-Hyuk Joo, Tobias Ruffer, Bernhard Walfort, and Heinrich Lang, Angew. Chem. Int. Ed. 2007, 46, 1168–1171. doi:10.1002/anie.200603960
- "Tetraazidomethane: Chemistry with a Bang", Chemical & Engineering News, Dec. 18, 2006, 46.
Salts and covalent derivatives of the azide ion | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HN3 | He | ||||||||||||||||||
| LiN3 | Be(N3)2 | B(N3)3 | CH3N3, C(N3)4 |
N(N3)3,H2N—N3 | O | FN3 | Ne | ||||||||||||
| NaN3 | Mg(N3)2 | Al(N3)3 | Si(N3)4 | P | SO2(N3)2 | ClN3 | Ar | ||||||||||||
| KN3 | Ca(N3)2 | Sc(N3)3 | Ti(N3)4 | VO(N3)3 | Cr(N3)3, CrO2(N3)2 |
Mn(N3)2 | Fe(N3)2, Fe(N3)3 |
Co(N3)2, Co(N3)3 |
Ni(N3)2 | CuN3, Cu(N3)2 |
Zn(N3)2 | Ga(N3)3 | Ge | As | Se(N3)4 | BrN3 | Kr | ||
| RbN3 | Sr(N3)2 | Y | Zr(N3)4 | Nb | Mo | Tc | Ru(N3)63− | Rh(N3)63− | Pd(N3)2 | AgN3 | Cd(N3)2 | In | Sn | Sb | Te | IN3 | Xe(N3)2 | ||
| CsN3 | Ba(N3)2 | Hf | Ta | W | Re | Os | Ir(N3)63− | Pt(N3)62− | Au(N3)4− | Hg2(N3)2, Hg(N3)2 |
TlN3 | Pb(N3)2 | Bi(N3)3 |
Po | At | Rn | |||
| Fr | Ra(N3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| ↓ | |||||||||||||||||||
| La | Ce(N3)3, Ce(N3)4 |
Pr | Nd | Pm | Sm | Eu | Gd(N3)3 | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||||
| Ac | Th | Pa | UO2(N3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.