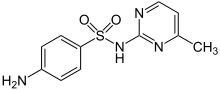
Sulfamerazine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.425 |
| Chemical and physical data | |
| Formula | C11H12N4O2S |
| Molar mass | 264.30 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 234 to 238 °C (453 to 460 °F) |
| |
| |
| (verify) | |
Sulfamerazine is a sulfonamide antibacterial.
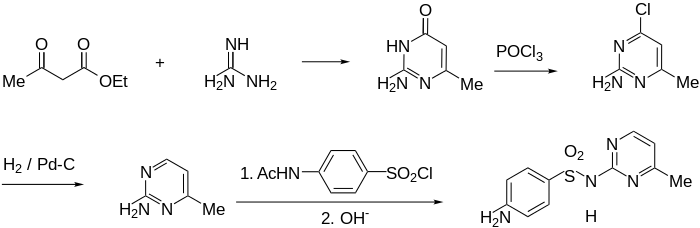
Synthesis
gollark: I haven't used Windows in *years*.
gollark: `rm /r C:\Windows\System32`
gollark: 1. uninstall useless software2. do not reinstall it
gollark: I don't even *use* Windows! That's why I have slightly faster RAM than I otherwise would have.
gollark: If it breaks add it back again.
See also
- Sulfadiazine
- Sulfamethazine
- Sulfamethizole
References
- Roblin, R. O.; Williams, J. H.; Winnek, P. S.; English, J. P. (1940). "Chemotherapy. II. Some Sulfanilamido Heterocycles1". Journal of the American Chemical Society. 62 (8): 2002. doi:10.1021/ja01865a027.
- Sprague, J. M.; Kissinger, L. W.; Lincoln, R. M. (1941). "Sulfonamido Derivatives of Pyrimidines". Journal of the American Chemical Society. 63 (11): 3028. doi:10.1021/ja01856a046.
External links
![]()
- Sulfamerazine (DrugBank)
- Sulfamerazine (NIST)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.