Cyclopropene
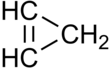
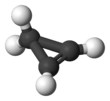
Cyclopropene is an organic compound with the formula C3H4. It is the simplest cycloalkene. Because the ring is highly strained, cyclopropene is difficult to prepare and highly reactive. This colorless gas has been the subject for many fundamental studies of bonding and reactivity.[2] It does not occur naturally, but derivatives are known in some fatty acids. Derivatives of cyclopropene are used commercially to control ripening of some fruit.
 | |||
| |||
| Names | |||
|---|---|---|---|
| Systematic IUPAC name
Cyclopropene[1] | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| MeSH | cyclopropene | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C3H4 | |||
| Molar mass | 40.065 g·mol−1 | ||
| Boiling point | −36 °C (−33 °F; 237 K) | ||
| Thermochemistry | |||
Heat capacity (C) |
51.9-53.9 J K−1 mol−1 | ||
Std enthalpy of combustion (ΔcH⦵298) |
-2032--2026 kJ mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Structure and bonding
The molecule has a triangular structure. The reduced length of the double bond compared to a single bond causes the angle opposite the double bond to narrow to about 51° from the 60° angle found in cyclopropane.[3] As with cyclopropane, the carbon–carbon bonding in the ring has increased p character: the alkene carbon atoms use sp2.68 hybridization for the ring.[4]
Synthesis of cyclopropene and derivatives
Early syntheses
The first confirmed synthesis of cyclopropene, carried out by Dem'yanov and Doyarenko, involved the thermal decomposition of trimethylcyclopropylammonium hydroxide over platinized clay at 320–330 °C under a CO2 atmosphere. This reaction produces mainly trimethylamine and dimethylcyclopropyl amine, together with about 5% of cyclopropene. Cyclopropene can also be obtained in about 1% yield by thermolysis of the adduct of cycloheptatriene and dimethyl acetylenedicarboxylate.
Modern syntheses from allyl chlorides
Allyl chloride undergoes dehydrohalogenation upon treatment with the base sodium amide at 80 °C to produce cyclopropene in about 10% yield.[5]
- CH2=CHCH2Cl + NaNH2 → C3H4 (cyclopropene) + NaCl + NH3
The major byproduct of the reaction is allylamine. Adding allyl chloride to sodium bis(trimethylsilyl)amide in boiling toluene over a period of 45–60 minutes produces the targeted compound in about 40% yield with an improvement in purity:[6]
- CH2=CHCH2Cl + NaN(TMS)2 → C3H4 (cyclopropene) + NaCl + NH(TMS)2
1-Methylcyclopropene is synthesized similarly but at room temperature from methallylchloride using phenyllithium as the base:[7]
- CH2=C(CH3)CH2Cl + LiC6H5 → CH3C3H3 (1-methylcyclopropene) + LiCl + C6H6
Syntheses of derivatives
Treatment of nitrocyclopropanes with sodium methoxide eliminates the nitrite, giving the respective cyclopropene derivative. The synthesis of purely aliphatic cyclopropenes was first illustrated by the copper-catalyzed additions of carbenes to alkynes. In the presence of a copper catalyst, ethyl diazoacetate reacts with acetylenes to give cyclopropenes. 1,2-Dimethylcyclopropene-3-carboxylate arises via this method from 2-butyne. Copper, as copper sulfate and copper dust, are among the more popular forms of copper used to promote such reactions. Rhodium acetate has also been used. The addition of dichlorocarbene to tetrachloroethylene gives tetrachlorocyclopropene.
Reactions of cyclopropene
Studies on cyclopropene mainly focus on the consequences of its high ring strain. At 425 °C, cyclopropene isomerizes to methylacetylene (propyne).
- C3H4 → H3CC≡CH
Attempted fractional distillation of cyclopropene at –36 °C (its predicted boiling point) results in polymerization. The mechanism is assumed to be a free-radical chain reaction, and the product, based on NMR spectra, is thought to be polycyclopropane.
Cyclopropene undergoes the Diels–Alder reaction with cyclopentadiene to give endo-tricyclo[3.2.1.02,4]oct-6-ene. This reaction is commonly used to check for the presence of cyclopropene, following its synthesis.[6]
Related compounds
- Malvalic acid is a toxic cyclopropene fatty acid that occurs in cottonseed oil.
- 1-Methylcyclopropene (1-MCP) is used to slow the ripening in fruits.[8][9]
- Borirenes, phosphirenes, and silirenes are boron-, phosphorus-, and silicon-substituted cyclopropenes, with the formula RBC2R'2, RPC2R'2, and R2SiC2R'2.
- Cyclopropene fatty acids a class of naturally occurring cyclopropenes.
References
- "cyclopropene - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 27 March 2005. Identification and Related Records. Retrieved 9 October 2011.
- Carter, F. L.; Frampton, V. L. (1964). "Review of the Chemistry of Cyclopropene Compounds". Chemical Reviews. 64: 497–525. doi:10.1021/cr60231a001.
- Staley, S. W.; Norden, T. D.; Su, C.-F.; Rall, M.; Harmony, M. D. (1987). "Structure of 3-cyanocyclopropene by microwave spectroscopy and ab initio molecular orbital calculations. Evidence for substituent-ring double bond interactions". J. Am. Chem. Soc. 109 (10): 2880–2884. doi:10.1021/ja00244a004.
- Allen, F. H. (1982). "The geometry of small rings: Molecular geometry of cyclopropene and its derivatives". Tetrahedron. 38 (5): 645–655. doi:10.1016/0040-4020(82)80206-8.
- Closs, G.L.; Krantz, K.D. (1966). "A Simple Synthesis of Cyclopropene". Journal of Organic Chemistry. 31: 638. doi:10.1021/jo01340a534.
- Binger, P.; Wedermann, P.; Brinker, U. H. (2000). "Cyclopropene: A New Simple Synthesis and Its Diels-Alder reaction with Cyclopentadiene". Organic Syntheses. 77: 254.; Collective Volume, 10, p. 231
- Clarke, T. C.; Duncan, C. D.; Magid, R. M. (1971). "An Efficient and Convenient Synthesis of 1-Methylcyclopropene". J. Org. Chem. 36: 1320. doi:10.1021/jo00808a041.
- Beaudry, R.; Watkins, C. (2001). "Use of 1-MCP on Apples". Perishable Handling Quarterly. University of California (108): 12.
- Trinchero, G. D.; Sozzi, G. O.; Covatta, F.; Fraschina, A. A. (May 2004). "Inhibition of ethylene action by 1-methylcyclopropene extends postharvest life of "Bartlett" pears". Postharvest Biology and Technology. 32 (2): 193–204. doi:10.1016/j.postharvbio.2003.11.009.