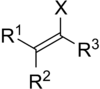
Vinyl halide
In organic chemistry, a vinyl halide is a compound with the formula CH2=CHX (X = halide). The term vinyl is often used to describe any alkenyl group. For this reason, alkenyl halides with the formula RCH=CHX are sometimes called vinyl halides. From the perspective of applications, the dominant member of this class of compounds is vinyl chloride, which is produced on the scale of millions of tons per year as a precursor to polyvinyl chloride.[1] Polyvinyl fluoride is another commercial product. Related compounds include vinylidene chloride and vinylidene fluoride.
Synthesis
Vinyl chloride is produced by dehydrochlorination of 1,2-dichloroethane.[1]
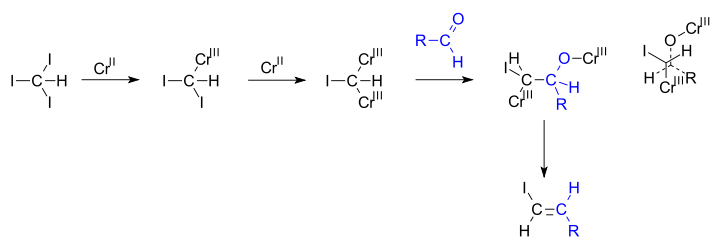
Due to their high utility, many approaches to vinyl halides have been developed, such as:
- reactions of vinyl organometallic species with halogens
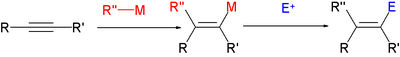
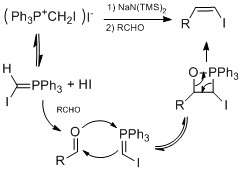
- Stork-Zhao olefination - a modification of the Wittig reaction
Reactions
Vinyl bromide and related alkenyl halides form the Grignard reagent and related organolithium reagents. Alkenyl halides undergo base elimination to give the corresponding alkyne. Most important is their use in cross-coupling reactions (e.g. Suzuki-Miyaura coupling, Stille coupling, Heck coupling, etc.).
See also
References
- E.-L. Dreher; T. R. Torkelson; K. K. Beutel (2011). "Chlorethanes and Chloroethylenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.o06_o01. ISBN 978-3527306732.
- Koh, Ming Joo; Nguyen, Thach T.; Zhang, Hanmo; Schrock, Richard R.; Hoveyda, Amir H. (2016). "Direct synthesis of Z-alkenyl halides through catalytic cross-metathesis". Nature. 531 (7595): 459–465. Bibcode:2016Natur.531..459K. doi:10.1038/nature17396. PMC 4858352. PMID 27008965.
- Nguyen, Thach T.; Koh, Ming Joo; Shen, Xiao; Romiti, Filippo; Schrock, Richard R.; Hoveyda, Amir H. (2016-04-29). "Kinetically controlled E-selective catalytic olefin metathesis". Science. 352 (6285): 569–575. Bibcode:2016Sci...352..569N. doi:10.1126/science.aaf4622. PMC 5748243. PMID 27126041.