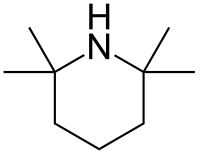
2,2,6,6-Tetramethylpiperidine
2,2,6,6-Tetramethylpiperidine, abbreviated TMP, HTMP, or TMPH, is an organic compound of the amine class. In appearance, it is a colorless liquid and has a "fishy", amine-like odor. This amine is used in chemistry as a hindered base (hindered amine). Although TMP finds limited use per se, its derivatives are a mainstay of hindered amine light stabilizers.
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2,2,6,6-Tetramethylpiperidine | |||
| Other names
Norpempidine Tetramethylpiperidine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| Abbreviations | TMP | ||
| ChemSpider | |||
| ECHA InfoCard | 100.011.090 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C9H19N | |||
| Molar mass | 141.254 g/mol | ||
| Appearance | Clear liquid | ||
| Density | 0.83 g/mL | ||
| Melting point | −59 °C (−74 °F; 214 K) | ||
| Boiling point | 152 °C (306 °F; 425 K) | ||
| Hazards | |||
| R-phrases (outdated) | R10 R22 R36/37/38 | ||
| S-phrases (outdated) | S16 S26 S37/39 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
TMP is the starting material for an even stronger base lithium tetramethylpiperidide and the radical species TEMPO. Another non-nucleophilic base is N,N-diisopropylethylamine.
Preparation
Many routes for the synthesis of TMP have been reported. One method[1] starts with a conjugate addition reaction of ammonia to phorone. The intermediate triacetone amine is then reduced in a Wolff-Kishner reaction.
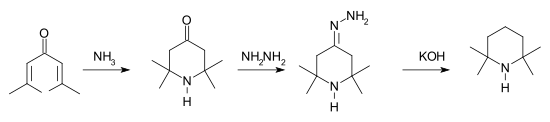
TMP synthesis
gollark: --magic py return 4
gollark: Oh right.
gollark: ++help magic
gollark: ++magic py return 4
gollark: ++magic py return 4
See also
- 2,6-Dimethylpiperidine
- Pempidine
- TEMPO ((2,2,6,6-Tetramethylpiperidin-1-yl)oxyl)
References
- Detlef Kampmann; Georg Stuhlmüller; Roger Simon; Fabrice Cottet; Frédéric Leroux; Manfred Schlosser (2005). "A Large-Scale Low-Cost Access to the Lithium 2,2,6,6-Tetramethylpiperidide Precursor". Synthesis. 2005 (06): 1028–1029. doi:10.1055/s-2004-834856.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.