Perovskite (structure)
A perovskite is any material with the same type of crystal structure as calcium titanium oxide (CaTiO3), known as the perovskite structure. [2] Perovskites take their name from the mineral, which was first discovered in the Ural mountains of Russia by Gustav Rose in 1839 and is named after Russian mineralogist L. A. Perovski (1792–1856). The general chemical formula for perovskite compounds is ABX3, where 'A' and 'B' are two cations of very different sizes, and X is an anion that bonds to both. The 'A' atoms are larger than the 'B' atoms. The ideal cubic structure has the B cation in 6-fold coordination, surrounded by an octahedron of anions, and the A cation in 12-fold cuboctahedral coordination.
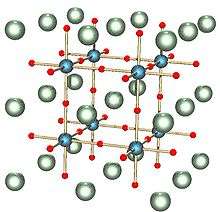

Natural compounds with this structure are perovskite, loparite, and the silicate perovskite bridgmanite.[2][3]
Structure
Perovskite structures are adopted by many oxides that have the chemical formula ABO3. The idealized form is a cubic structure (Pm3m) which is rarely encountered. The orthorhombic (Amm2) and tetragonal (P4mm) phases are the most common non-cubic variants. Although the perovskite structure is named after CaTiO3, this mineral forms a non-idealized form. SrTiO3 and CaRbF3 are examples of cubic perovskites. Barium titanate is an example of a perovskite which can take on the rhombohedral (R3m), orthorhombic, tetragonal and cubic forms depending on temperature.[4]
In the idealized cubic unit cell of such a compound, the type 'A' atom sits at cube corner position (0, 0, 0), the type 'B' atom sits at the body-center position (1/2, 1/2, 1/2) and oxygen atoms sit at face centered positions (1/2, 1/2, 0), (1/2, 0, 1/2) and (0, 1/2, 1/2). The diagram to the right shows edges for an equivalent unit cell with A in the cube corner position, B at the body center, and O at face-centered positions.
Three general categories of cation-pairing are possible: A2+B4+X2−3, or 2:4 perovskites; A3+B3+X2−3, or 3:3 perovskites; and A+B5+X2−3, or 1:5 perovskites.
The relative ion size requirements for stability of the cubic structure are quite stringent, so slight buckling and distortion can produce several lower-symmetry distorted versions, in which the coordination numbers of A cations, B cations or both are reduced. Tilting of the BO6 octahedra reduces the coordination of an undersized A cation from 12 to as low as 8. Conversely, off-centering of an undersized B cation within its octahedron allows it to attain a stable bonding pattern. The resulting electric dipole is responsible for the property of ferroelectricity and shown by perovskites such as BaTiO3 that distort in this fashion.
Complex perovskite structures contain two different B-site cations. This results in the possibility of ordered and disordered variants.
Layered perovskites
Perovskites may be structured in layers, with the ABO
3 structure separated by thin sheets of intrusive material. Different forms of intrusions, based on the chemical makeup of the intrusion, are defined as:[5]
- Aurivillius phase: the intruding layer is composed of a [Bi
2O
2]2+ ion, occurring every n ABO
3 layers, leading to an overall chemical formula of [Bi
2O
2]-A
(n−1)B
2O
7. Their oxide ion-conducting properties were first discovered in the 1970s by Takahashi et al., and they have been used for this purpose ever since.[6] - Dion−Jacobson phase: the intruding layer is composed of an alkali metal (M) every n ABO
3 layers, giving the overall formula as M+
A
(n−1)B
nO
(3n+1) - Ruddlesden-Popper phase: the simplest of the phases, the intruding layer occurs between every one (n = 1) or two (n = 2) layers of the ABO
3 lattice. Ruddlesden−Popper phases have a similar relationship to perovskites in terms of atomic radii of elements with A typically being large (such as La[7] or Sr[8]) with the B ion being much smaller typically a transition metal (such as Mn,[7] Co[9] or Ni[10]).
Thin films

Perovskites can be deposited as epitaxial thin films on top of other perovskites,[11] using techniques such as pulsed laser deposition and molecular-beam epitaxy. These films can be a couple of nanometres thick or as small as a single unit cell.[12] The well-defined and unique structures at the interfaces between the film and substrate can be used for interface engineering, where new types properties can arise.[13] This can happen through several mechanisms, from mismatch strain between the substrate and film, change in the oxygen octahedral rotation, compositional changes, and quantum confinement.[14] An example of this is LaAlO3 grown on SrTiO3, where the interface can exhibit conductivity, even though both LaAlO3 and SrTiO3 is non-conductive.[15]
Common occurrence
The perovskite structure is adopted at high pressure by bridgmanite, a silicate with the formula (Mg,Fe)SiO3, which is the most common mineral in the Earth's mantle. As pressure increases, the SiO44− tetrahedral units in the dominant silica-bearing minerals become unstable compared with SiO68− octahedral units. At the pressure and temperature conditions of the lower mantle, the second most abundant material is likely the rocksalt-structured (Mg,Fe)O oxide, periclase.[2]
At the high pressure conditions of the Earth's lower mantle, the pyroxene enstatite, MgSiO3, transforms into a denser perovskite-structured polymorph; this phase may be the most common mineral in the Earth.[16] This phase has the orthorhombically distorted perovskite structure (GdFeO3-type structure) that is stable at pressures from ~24 GPa to ~110 GPa. However, it cannot be transported from depths of several hundred km to the Earth's surface without transforming back into less dense materials. At higher pressures, MgSiO3 perovskite, commonly known as silicate perovskite, transforms to post-perovskite.
Although the most common perovskite compounds contain oxygen, there are a few perovskite compounds that form without oxygen. Fluoride perovskites such as NaMgF3 are well known. A large family of metallic perovskite compounds can be represented by RT3M (R: rare-earth or other relatively large ion, T: transition metal ion and M: light metalloids). The metalloids occupy the octahedrally coordinated "B" sites in these compounds. RPd3B, RRh3B and CeRu3C are examples. MgCNi3 is a metallic perovskite compound and has received lot of attention because of its superconducting properties. An even more exotic type of perovskite is represented by the mixed oxide-aurides of Cs and Rb, such as Cs3AuO, which contain large alkali cations in the traditional "anion" sites, bonded to O2− and Au− anions.
Materials properties
Perovskite materials exhibit many interesting and intriguing properties from both the theoretical and the application point of view. Colossal magnetoresistance, ferroelectricity, superconductivity, charge ordering, spin dependent transport, high thermopower and the interplay of structural, magnetic and transport properties are commonly observed features in this family. These compounds are used as sensors and catalyst electrodes in certain types of fuel cells[17] and are candidates for memory devices and spintronics applications.[18]
Many superconducting ceramic materials (the high temperature superconductors) have perovskite-like structures, often with 3 or more metals including copper, and some oxygen positions left vacant. One prime example is yttrium barium copper oxide which can be insulating or superconducting depending on the oxygen content.
Chemical engineers are considering a cobalt-based perovskite material as a replacement for platinum in catalytic converters in diesel vehicles.[19]
Applications
Physical properties of interest to materials science among perovskites include superconductivity, magnetoresistance, ionic conductivity, and a multitude of dielectric properties, which are of great importance in microelectronics and telecommunication. They are also some interests for scintillator as they have large light yield for radiation conversion. Because of the flexibility of bond angles inherent in the perovskite structure there are many different types of distortions which can occur from the ideal structure. These include tilting of the octahedra, displacements of the cations out of the centers of their coordination polyhedra, and distortions of the octahedra driven by electronic factors (Jahn-Teller distortions).[20]
Photovoltaics
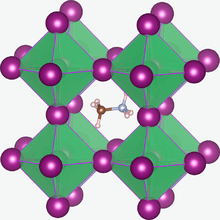
Synthetic perovskites have been identified as possible inexpensive base materials for high-efficiency commercial photovoltaics[22][23] – they showed a conversion efficiency of up to 22.34%[23][24][25] and can be manufactured using the same thin-film manufacturing techniques as that used for thin film silicon solar cells.[26] Methylammonium tin halides and methylammonium lead halides are of interest for use in dye-sensitized solar cells.[27][28] In July 2016, a team of researchers led by Dr. Alexander Weber-Bargioni demonstrated that perovskite PV cells could reach a theoretical peak efficiency of 31%.[29]
Among the methylammonium halides studied so far the most common is the methylammonium lead triiodide (CH
3NH
3PbI
3). It has a high charge carrier mobility and charge carrier lifetime that allow light-generated electrons and holes to move far enough to be extracted as current, instead of losing their energy as heat within the cell. CH
3NH
3PbI
3 effective diffusion lengths are some 100 nm for both electrons and holes.[30]
Methylammonium halides are deposited by low-temperature solution methods (typically spin-coating). Other low-temperature (below 100 °C) solution-processed films tend to have considerably smaller diffusion lengths. Stranks et al. described nanostructured cells using a mixed methylammonium lead halide (CH3NH3PbI3−xClx) and demonstrated one amorphous thin-film solar cell with an 11.4% conversion efficiency, and another that reached 15.4% using vacuum evaporation. The film thickness of about 500 to 600 nm implies that the electron and hole diffusion lengths were at least of this order. They measured values of the diffusion length exceeding 1 μm for the mixed perovskite, an order of magnitude greater than the 100 nm for the pure iodide. They also showed that carrier lifetimes in the mixed perovskite are longer than in the pure iodide.[30] Liu et al. applied Scanning Photo-current Microscopy to show that the electron diffusion length in mixed halide perovskite along (110) plane is in the order of 10 μm.[31]
For CH
3NH
3PbI
3, open-circuit voltage (VOC) typically approaches 1 V, while for CH
3NH
3PbI(I,Cl)
3 with low Cl content, VOC > 1.1 V has been reported. Because the band gaps (Eg) of both are 1.55 eV, VOC-to-Eg ratios are higher than usually observed for similar third-generation cells. With wider bandgap perovskites, VOC up to 1.3 V has been demonstrated.[30]
The technique offers the potential of low cost because of the low temperature solution methods and the absence of rare elements. Cell durability is currently insufficient for commercial use.[30]
Planar heterojunction perovskite solar cells can be manufactured in simplified device architectures (without complex nanostructures) using only vapor deposition. This technique produces 15% solar-to-electrical power conversion as measured under simulated full sunlight.[32]
Lasers
In 2008, researchers demonstrated that perovskite can generate laser light. LaAlO3 doped with neodymium gave laser emission at 1080 nm.[33] In 2014 it was shown that mixed methylammonium lead halide (CH3NH3PbI3−xClx) cells fashioned into optically pumped vertical-cavity surface-emitting lasers (VCSELs) convert visible pump light to near-IR laser light with a 70% efficiency.[34][35]
Light-emitting diodes
Due to their high photoluminescence quantum efficiencies, perovskites may be good candidates for use in light-emitting diodes (LEDs).[36] However, the propensity for radiative recombination has mostly been observed at liquid nitrogen temperatures.
Photoelectrolysis
In September 2014, researchers at EPFL in Lausanne, Switzerland, reported achieving water electrolysis at 12.3% efficiency in a highly efficient and low-cost water-splitting cell using perovskite photovoltaics.[37][38]
Scintillators
In 1997, scintillation properties of Cerium doped Lutetium Aluminum Perovskite (LuAP:Ce) single crystals were reported.[39] The main property of those crystals is a large mass density of 8.4 g/cm3, which gives short X- and gamma-ray absorption length. The scintillation light yield and the decay time with Cs137 radiation source are 11,400 photons/MeV and 17 ns, respectively. Those properties made LUAP:Ce scintillators attractive for commercials and they were used quite often in high energy physics experiments. Until eleven years later, one group in Japan proposed Ruddlesden-Popper solution-based hybrid organic-inorganic perovskite crystals as low-cost scintillators.[40] However, the properties were not so impressive in comparison with LuAP:Ce. Until the next nine years, the solution-based hybrid organic-inorganic perovskite crystals became popular again through a report about their high light yields of more than 100,000 photons/MeV at cryogenic temperatures.[41] Finally, the excellent demonstration of perovskite nanocrystal scintillators for X-ray imaging screen was reported and it is triggering more research efforts for perovskite scintillators.[42]
Examples of perovskites
Simple:
- Strontium titanate
- Calcium titanate
- Lead titanate
- Bismuth ferrite
- Lanthanum ytterbium oxide
- Silicate perovskite
- Lanthanum manganite
- Yttrium aluminum perovskite (YAP)
- Lutetium aluminum perovskite (LuAP)
- Lanthanum strontium manganite
- LSAT (lanthanum aluminate – strontium aluminum tantalate)
- Lead scandium tantalate
- Lead zirconate titanate
- Methylammonium lead halide
See also
References
- A. Navrotsky (1998). "Energetics and Crystal Chemical Systematics among Ilmenite, Lithium Niobate, and Perovskite Structures". Chem. Mater. 10 (10): 2787. doi:10.1021/cm9801901.
- Wenk, Hans-Rudolf; Bulakh, Andrei (2004). Minerals: Their Constitution and Origin. New York, NY: Cambridge University Press. ISBN 978-0-521-52958-7.
- Bridgemanite on Mindat.org
- https://onlinelibrary.wiley.com/doi/full/10.1002/9780470022184.hmm411
- Cava, Robert J. "Cava Lab: Perovskites". Princeton University. Retrieved 13 November 2013.
- Kendall, K. R.; Navas, C.; Thomas, J. K.; Zur Loye, H. C. (1996). "Recent Developments in Oxide Ion Conductors: Aurivillius Phases". Chemistry of Materials. 8 (3): 642–649. doi:10.1021/cm9503083.
- Munnings, C; Skinner, S; Amow, G; Whitfield, P; Davidson, I (15 October 2006). "Structure, stability and electrical properties of the La(2−x)SrxMnO4±δ solid solution series". Solid State Ionics. 177 (19–25): 1849–1853. doi:10.1016/j.ssi.2006.01.009.
- Munnings, Christopher N.; Sayers, Ruth; Stuart, Paul A.; Skinner, Stephen J. (January 2012). "Structural transformation and oxidation of Sr2MnO3.5+x determined by in-situ neutron powder diffraction" (PDF). Solid State Sciences. 14 (1): 48–53. Bibcode:2012SSSci..14...48M. doi:10.1016/j.solidstatesciences.2011.10.015. hdl:10044/1/15437.
- Amow, G.; Whitfield, P. S.; Davidson, I. J.; Hammond, R. P.; Munnings, C. N.; Skinner, S. J. (January 2004). "Structural and sintering characteristics of the La2Ni1−xCoxO4+δ series". Ceramics International. 30 (7): 1635–1639. doi:10.1016/j.ceramint.2003.12.164.
- Amow, G.; Whitfield, P. S.; Davidson, J.; Hammond, R. P.; Munnings, C.; Skinner, S. (11 February 2011). "Structural and Physical Property Trends of the Hyperstoichiometric Series, La2Ni(1−x)CoxO4+δ". MRS Proceedings. 755. doi:10.1557/PROC-755-DD8.10.
- Martin, L.W.; Chu, Y.-H.; Ramesh, R. (May 2010). "Advances in the growth and characterization of magnetic, ferroelectric, and multiferroic oxide thin films". Materials Science and Engineering: R: Reports. 68 (4–6): 89–133. doi:10.1016/j.mser.2010.03.001.
- Yang, G.Z; Lu, H.B; Chen, F; Zhao, T; Chen, Z.H (July 2001). "Laser molecular beam epitaxy and characterization of perovskite oxide thin films". Journal of Crystal Growth. 227-228: 929–935. doi:10.1016/S0022-0248(01)00930-7.
- Mannhart, J.; Schlom, D. G. (25 March 2010). "Oxide Interfaces--An Opportunity for Electronics". Science. 327 (5973): 1607–1611. doi:10.1126/science.1181862. PMID 20339065.
- Chakhalian, J.; Millis, A. J.; Rondinelli, J. (24 January 2012). "Whither the oxide interface". Nature Materials. 11 (2): 92–94. doi:10.1038/nmat3225. PMID 22270815.
- Ohtomo, A.; Hwang, H. Y. (January 2004). "A high-mobility electron gas at the LaAlO3/SrTiO3 heterointerface". Nature. 427 (6973): 423–426. doi:10.1038/nature02308. PMID 14749825.
- John Lloyd; John Mitchinson (2006). "What's the commonest material in the world". QI: The Book of General Ignorance. Faber & Faber. ISBN 978-0-571-23368-7.
- Kulkarni, A; FT Ciacchi; S Giddey; C Munnings; et al. (2012). "Mixed ionic electronic conducting perovskite anode for direct carbon fuel cells". International Journal of Hydrogen Energy. 37 (24): 19092–19102. doi:10.1016/j.ijhydene.2012.09.141.
- J. M. D. Coey; M. Viret; S. von Molnar (1999). "Mixed-valence manganites". Advances in Physics. 48 (2): 167–293. Bibcode:1999AdPhy..48..167C. doi:10.1080/000187399243455.
- Alexandra Witze (2010). "Building a cheaper catalyst". Science News Web Edition.
- Lufaso, Michael W.; Woodward, Patrick M. (2004). "Jahn–Teller distortions, cation ordering and octahedral tilting in perovskites". Acta Crystallographica Section B. 60 (Pt 1): 10–20. doi:10.1107/S0108768103026661. PMID 14734840.
- Eames, Christopher; Frost, Jarvist M.; Barnes, Piers R. F.; o'Regan, Brian C.; Walsh, Aron; Islam, M. Saiful (2015). "Ionic transport in hybrid lead iodide perovskite solar cells". Nature Communications. 6: 7497. Bibcode:2015NatCo...6.7497E. doi:10.1038/ncomms8497. PMC 4491179. PMID 26105623.
- Bullis, Kevin (8 August 2013). "A Material That Could Make Solar Power "Dirt Cheap"". MIT Technology Review. Retrieved 8 August 2013.
- Li, Hangqian. (2016). "A modified sequential deposition method for fabrication of perovskite solar cells". Solar Energy. 126: 243–251. Bibcode:2016SoEn..126..243L. doi:10.1016/j.solener.2015.12.045.
- "Research Cell Efficiency Records". Office of Energy Efficiency & Renewable Energy. 2016.
- Zhu, Rui (2020-02-10). "Inverted devices are catching up". Nature Energy. 5 (2): 123–124. doi:10.1038/s41560-020-0559-z. ISSN 2058-7546.
- Liu, Mingzhen; Johnston, Michael B.; Snaith, Henry J. (2013). "Efficient planar heterojunction perovskite solar cells by vapour deposition". Nature. 501 (7467): 395–398. Bibcode:2013Natur.501..395L. doi:10.1038/nature12509. PMID 24025775.
- Lotsch, B.V. (2014). "New Light on an Old Story: Perovskites Go Solar". Angew. Chem. Int. Ed. 53 (3): 635–637. doi:10.1002/anie.201309368. PMID 24353055.
- Service, R. (2013). "Turning Up the Light". Science. 342 (6160): 794–797. doi:10.1126/science.342.6160.794. PMID 24233703.
- "Nanoscale discovery could push perovskite solar cells to 31% efficency". 2016-07-04.
- Hodes, G. (2013). "Perovskite-Based Solar Cells". Science. 342 (6156): 317–318. Bibcode:2013Sci...342..317H. doi:10.1126/science.1245473. PMID 24136955.
- Liu, Shuhao; Wang, Lili; Lin, Wei-Chun; Sucharitakul, Sukrit; Burda, Clemens; Gao, Xuan P. A. (2016-12-14). "Imaging the Long Transport Lengths of Photo-generated Carriers in Oriented Perovskite Films". Nano Letters. 16 (12): 7925–7929. arXiv:1610.06165. Bibcode:2016NanoL..16.7925L. doi:10.1021/acs.nanolett.6b04235. ISSN 1530-6984. PMID 27960525.
- Liu, M.; Johnston, M. B.; Snaith, H. J. (2013). "Efficient planar heterojunction perovskite solar cells by vapour deposition". Nature. 501 (7467): 395–398. Bibcode:2013Natur.501..395L. doi:10.1038/nature12509. PMID 24025775.
- Dereń, P. J.; Bednarkiewicz, A.; Goldner, Ph.; Guillot-Noël, O. (2008). "Laser action in LaAlO3:Nd3+ single crystal". Journal of Applied Physics. 103 (4): 043102–043102–8. Bibcode:2008JAP...103d3102D. doi:10.1063/1.2842399.
- Wallace, John (28 March 2014) High-efficiency perovskite photovoltaic material also lases. LaserFocusWorld
- "Study: Perovskite solar cells can double as lasers". Rdmag.com. 2014-03-28. Retrieved 2014-08-24.
- Stranks, Samuel D.; Snaith, Henry J. (2015-05-01). "Metal-halide perovskites for photovoltaic and light-emitting devices". Nature Nanotechnology. 10 (5): 391–402. Bibcode:2015NatNa..10..391S. doi:10.1038/nnano.2015.90. ISSN 1748-3387. PMID 25947963.
- Jingshan Luo; et al. (26 September 2014). "Water photolysis at 12.3% efficiency via perovskite photovoltaics and Earth-abundant catalysts". Science. 345 (6204): 1593–1596. Bibcode:2014Sci...345.1593L. doi:10.1126/science.1258307. PMID 25258076.
- "Harvesting hydrogen fuel from the Sun using Earth-abundant materials". Phys.org. Sep 25, 2014. Retrieved 26 September 2014.
- Moszynski, M (11 January 1997). "Properties of the new LuAP:Ce scintillator". Nuclear Inst. And Methods in Physics Research A. 385: 123–131. doi:10.1016/S0168-9002(96)00875-3.
- Kishimoto, S (29 December 2008). "Subnanosecond time-resolved x-ray measurements using an organic-inorganic perovskite scintillator". Appl. Phys. Lett. 93 (26): 261901. Bibcode:2008ApPhL..93z1901K. doi:10.1063/1.3059562.
- Birowosuto, Muhammad Danang (16 November 2016). "X-ray Scintillation in Lead Halide Perovskite Crystals". Sci. Rep. 6: 37254. arXiv:1611.05862. Bibcode:2016NatSR...637254B. doi:10.1038/srep37254. PMC 5111063. PMID 27849019.
- Chen, Quishui (27 August 2018). "All-inorganic perovskite nanocrystal scintillators". Nature. 561 (7721): 88–93. doi:10.1038/s41586-018-0451-1. PMID 30150772.
Further reading
- Tejuca, Luis G (1993). Properties and applications of perovskite-type oxides. New York: Dekker. p. 382. ISBN 978-0-8247-8786-8.
- Mitchell, Roger H (2002). Perovskites modern and ancient. Thunder Bay, Ontario: Almaz Press. p. 318. ISBN 978-0-9689411-0-2.
External links
- "Cubic Perovskite Structure". Center for Computational Materials Science. U.S. Naval Research Laboratory. Archived from the original on 2008-10-08. (includes a Java applet with which the structure can be interactively rotated)