Myoviridae
Myoviridae is a family of bacteriophages in the order Caudovirales. Bacteria and archaea serve as natural hosts. There are currently 434 species in this family, divided among five subfamilies and 168 genera.[1][2]
| Myoviridae | |
|---|---|
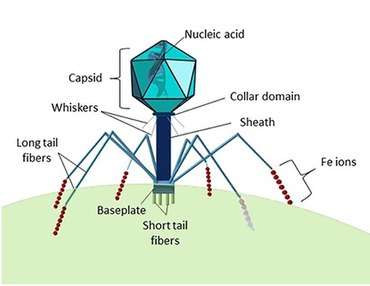 | |
| Typical structure of a myovirus | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Duplodnaviria |
| Kingdom: | Heunggongvirae |
| Phylum: | Uroviricota |
| Class: | Caudoviricetes |
| Order: | Caudovirales |
| Family: | Myoviridae |
| Subfamilies and genera | |
|
see text | |
Subdivisions
The subfamily Tevenvirinae (synonym: Tequatrovirinae) is named after its type species Enterobacteria phage T4. Members of this subfamily are morphologically indistinguishable and have moderately elongated heads of about 110 nanometers (nm) in length, 114 nm long tails with a collar, base plates with short spikes and six long kinked tail fibers. The genera within this subfamily are divided on the basis of head morphology with the genus Tequatrovirus (Provisional name: T4virus) having a head length of 137 nm and those in the genus Schizot4virus being 111 nm in length. Within the genera on the basis of protein homology the species have been divided into a number of groups.
The subfamily Peduovirinae have virions with heads of 60 nm in diameter and tails of 135 × 18 nm. These phages are easily identified because contracted sheaths tend to slide off the tail core. The P" phage is the type species.
The subfamily Spounavirinae are all virulent, broad-host range phages that infect members of the Firmicutes. They possess isometric heads of 87-94 nm in diameter and conspicuous capsomers, striated 140-219 nm long tails and a double base plate. At the tail tip are globular structures now known to be the base plate spikes and short kinked tail fibers with six-fold symmetry. Members of this group usually possess large (127–142 kb) nonpermuted genomes with 3.1–20 kb terminal redundancies. The name for this subfamily is derived from SPO plus una (Latin for one).
The haloviruses HF1 and HF2 belong to the same genus but since they infect archaea rather than bacteria are likely to be placed in a separate genus once their classification has been settled.[3]
A dwarf group has been proposed on morphological and genomic grounds. This group includes the phages Aeromonas salmonicida phage 56, Vibrio cholerae phages 138 and CP-T1, Bdellovibrio phage φ1422 and Pectobacterium carotovorum phage ZF40.[4] Their shared characteristics include an identical virion morphology, characterized by usually short contractile tails and all have genome sizes of approximately 45 kilobases. The gene order in the structural unit of the genome is in the order: terminase—portal—head—tail—base plate—tail fibers.
Virology
Viruses in Myoviridae are non-enveloped, with head-tail (with a neck) geometries. Genomes are linear, double-stranded DNA, around 33-244kb in length. The genome codes for 40 to 415 proteins.[1] It has terminally redundant sequences. The GC-content is ~35%. The genome encodes 200-300 proteins that are transcribed in operons. 5-Hydroxymethylcytosine may be present in the genome (instead of thymidine).
The tubular tail has helical symmetry and is 16-20 nm in diameter. It consists of a central tube, a contractile sheath, a collar, a base plate, six tail pins and six long fibers. It is similar to Tectiviridae, but differs in the fact that a myovirus' tail is permanent.
Contractions of the tail require ATP. On contraction of the sheath, sheath subunits slide over each other and the tail shortens to 10–15 nm in length.
Life cycle
On attaching to a host cell, the virus uses its contractile sheath like a syringe, piercing the cell wall with its central tube and injecting the genetic material into the host. The injected DNA takes over the host cell's mechanisms for transcription and translation and begins to manufacture new viruses. Replication follows the replicative transposition model. DNA-templated transcription is the method of transcription. Translation takes place by -1 ribosomal frameshifting. The virus exits the host cell by lysis, and holin/endolysin/spanin proteins. Bacteria and archaea serve as the natural host. Transmission route is passive diffusion.[1]
Although Myoviruses are in general lytic, lacking the genes required to become lysogenic, a number of temperate species are known.
Applications
Because most Myoviridae are lytic, rather than temperate, phages, some researchers have investigated their use as a therapy for bacterial diseases in humans and other animals.[5]
Taxonomy
The following five subfamilies are recognized:[2]
- Eucampyvirinae, containing two genera and nine species
- Ounavirinae, containing four genera and 25 species
- Peduovirinae, containing 26 genera and 47 species
- Tevenvirinae, containing 11 genera and 77 species
- Vequintavirinae, containing four genera and 18 species
Additionally, the following genera are unassigned to a subfamily:[2]
- Abouovirus
- Acionnavirus
- Agricanvirus
- Ahtivirus
- Alcyoneusvirus
- Alexandravirus
- Anamdongvirus
- Anaposvirus
- Aokuangvirus
- Aphroditevirus
- Asteriusvirus
- Atlauavirus
- Aurunvirus
- Baikalvirus
- Barbavirus
- Bcepmuvirus
- Bellamyvirus
- Bendigovirus
- Biquartavirus
- Bixzunavirus
- Brigitvirus
- Brizovirus
- Brunovirus
- Busanvirus
- Carpasinavirus
- Chakrabartyvirus
- Charybdisvirus
- Chiangmaivirus
- Cymopoleiavirus
- Derbicusvirus
- Elvirus
- Emdodecavirus
- Eneladusvirus
- Eponavirus
- Erskinevirus
- Eurybiavirus
- Ficleduovirus
- Flaumdravirus
- Gofduovirus
- Goslarvirus
- Hapunavirus
- Heilongjiangvirus
- Iapetusvirus
- Iodovirus
- Ionavirus
- Jedunavirus
- Jilinvirus
- Jimmervirus
- Kanaloavirus
- Kleczkowskavirus
- Lagaffevirus
- Leucotheavirus
- Libanvirus
- Llyrvirus
- Loughboroughvirus
- Lubbockvirus
- Machinavirus
- Marthavirus
- Mazuvirus
- Metrivirus
- Mieseafarmvirus
- Mimasvirus
- Mushuvirus
- Muvirus
- Myohalovirus
- Naesvirus
- Namakavirus
- Nankokuvirus
- Neptunevirus
- Nereusvirus
- Nerrivikvirus
- Nodensvirus
- Noxifervirus
- Obolenskvirus
- Otagovirus
- Pakpunavirus
- Palaemonvirus
- Pbunavirus
- Peatvirus
- Petsuvirus
- Phabquatrovirus
- Phapecoctavirus
- Phikzvirus
- Plaisancevirus
- Polybotosvirus
- Pontusvirus
- Popoffvirus
- Punavirus
- Qingdaovirus
- Radnorvirus
- Ripduovirus
- Risingsunvirus
- Rosemountvirus
- Saclayvirus
- Salacisavirus
- Salmondvirus
- Sasquatchvirus
- Schmittlotzvirus
- Seoulvirus
- Shalavirus
- Svunavirus
- Tabernariusvirus
- Tamkungvirus
- Taranisvirus
- Tefnutvirus
- Tegunavirus
- Thaumasvirus
- Thetisvirus
- Thornevirus
- Tidunavirus
- Tijeunavirus
- Toutatisvirus
- Tulanevirus
- Vellamovirus
- Vhmlvirus
- Viunavirus
- Wellingtonvirus
- Wifcevirus
- Winklervirus
- Yokohamavirus
- Yoloswagvirus
Lastly, the following three species are unassigned to a genus and subfamily:[2]
- Bacillus virus G
- Bacillus virus PBS1
- Microcystis virus Ma-LMM01
Proposed Genera
The following genera have been proposed but are not currently ratified by the International Committee on Taxonomy of Viruses:[3][6][7][8][9][10]
- Genus Bxzunalikevirus; type species: Mycobacterium phage Bxz1
- Species
- Mycobacterium phage Bxz1
- Mycobacterium phage Cali
- Mycobacterium phage Catera
- Mycobacterium phage Myrna
- Mycobacterium phage Rizal
- Mycobacterium phage ScottMcG
- Mycobacterium phage Spud
- Species
- Genus Cbasmlikevirus; type species: Cellulophaga phage phiSM
- Species
- Cellulophaga phage phiSM
- Cellulophaga phage phi3:1
- Cellulophaga phage phi3ST:2
- Cellulophaga phage phi38:2
- Cellulophaga phage phi47:1
- Species
- Genus cyanomyoviruses (informal); type species: Synechococcus phage S-PM2
- Species
- Synechococcus phage S-PM2
- Synechococcus Syn9
- Prochlorococcus phage P-SSM2
- Prochlorococcus phage P-SSM4
- Species
- Genus Ellikevirus; type species: Pseudomonas phage EL
- Species
- Pseudomonas phage EL
- Pseudomonas phage OBP
- Salmonella phage SPN3US
- Species
- Genus Hfunalikevirus; type species: Halovirus HF1
- Species
- Halovirus HF1
- Halovirus HF2
- Species
- Genus Plpelikevirus (synonym: dwarf myoviruses); type species: Iodobacter phage phiPLPE
- Species
- Aeromonas phage 56
- Aggregatibacter phage Aaphi23
- Bdellovibrio phage phi1402
- Bdellovibrio phage phi1422
- Iodobacter phage phiPLPE
- Pectobacterium phage ZF40
- Vibrio phage 138
- Vibrio phage CP-T1
- Yersinia phage PY100
- Species
- Genus Rv5likevirus; type species: Escherichia phage rV5
- Species
- Enterobacteria phage phi92
- Escherichia phage rV5
- Escherichia phage vB_EcoM_FV3
- Salmonella phage PVP-SE1
- Species
- Genus Sfv and relatives (informal); type species: Shigella phage SfV
- Species
- Shigella phage SfV
- Escherichia phage P27
- Species
References
- "Viral Zone". ExPASy. Retrieved 1 July 2015.
- "Virus Taxonomy: 2019 Release". talk.ictvonline.org. International Committee on Taxonomy of Viruses. Retrieved 3 May 2020.
- Tang, SL; Nuttall, S; Dyall-Smith, M (2004). "Haloviruses HF1 and HF2: evidence for a recent and large recombination event". J Bacteriol. 186 (9): 2810–7. doi:10.1128/JB.186.9.2810-2817.2004. PMC 387818. PMID 15090523.
- Comeau, AM; Tremblay, D; Moineau, S; Rattei, T; Kushkina, AI; Tovkach, FI; Krisch, HM; Ackermann, HW (2012). "Phage morphology recapitulates phylogeny: the comparative genomics of a new group of myoviruses". PLOS ONE. 7 (7): e40102. doi:10.1371/journal.pone.0040102. PMC 3391216. PMID 22792219.
- Capparelli, Rosanna; et al. (August 2007). "Experimental phage therapy against Staphylococcus aureus in mice". Antimicrobial Agents and Chemotherapy. 51 (8): 2765–73. doi:10.1128/AAC.01513-06. PMC 1932491. PMID 17517843.
- Santos, S. B.; Kropinski, A. M.; Ceyssens, P. -J.; Ackermann, H. - W.; Villegas, A.; Lavigne, R.; Krylov, V. N.; Carvalho, C. M.; Ferreira, E. C.; Azeredo, J. (2011). "Genomic and Proteomic Characterization of the Broad-Host-Range Salmonella Phage PVP-SE1: Creation of a New Phage Genus". Journal of Virology. 85 (21): 11265–73. doi:10.1128/JVI.01769-10. PMC 3194984. PMID 21865376.
- Truncaite, L.; Šimoliūnas, E.; Zajančkauskaite, A.; Kaliniene, L.; Mankevičiūte, R.; Staniulis, J.; Klausa, V.; Meškys, R. (2012). "Bacteriophage vB_EcoM_FV3: A new member of "rV5-like viruses"". Archives of Virology. 157 (12): 2431–5. doi:10.1007/s00705-012-1449-x. PMID 22907825.
- Cornelissen, A.; Hardies, S. C.; Shaburova, O. V.; Krylov, V. N.; Mattheus, W.; Kropinski, A. M.; Lavigne, R. (2011). "Complete Genome Sequence of the Giant Virus OBP and Comparative Genome Analysis of the Diverse KZ-Related Phages". Journal of Virology. 86 (3): 1844–52. doi:10.1128/JVI.06330-11. PMC 3264338. PMID 22130535.
- Mizuno, C. M.; Rodriguez-Valera, F.; Kimes, N. E.; Ghai, R. (2013). "Expanding the Marine Virosphere Using Metagenomics". PLoS Genetics. 9 (12): e1003987. doi:10.1371/journal.pgen.1003987. PMC 3861242. PMID 24348267.
- Holmfeldt, K.; Solonenko, N.; Shah, M.; Corrier, K.; Riemann, L.; Verberkmoes, N. C.; Sullivan, M. B. (2013). "Twelve previously unknown phage genera are ubiquitous in global oceans". Proceedings of the National Academy of Sciences. 110 (31): 12798–803. doi:10.1073/pnas.1305956110. PMC 3732932. PMID 23858439.