Mesolimbic pathway
The mesolimbic pathway, sometimes referred to as the reward pathway, is a dopaminergic pathway in the brain.[1] The pathway connects the ventral tegmental area in the midbrain to the ventral striatum of the basal ganglia in the forebrain. The ventral striatum includes the nucleus accumbens and the olfactory tubercle.[2]
The release of dopamine from the mesolimbic pathway into the nucleus accumbens regulates incentive salience (e.g. motivation and desire for rewarding stimuli) and facilitates reinforcement and reward-related motor function learning;[3][4][5] it may also play a role in the subjective perception of pleasure.[3][5] The dysregulation of the mesolimbic pathway and its output neurons in the nucleus accumbens plays a significant role in the development and maintenance of an addiction.[1][6][7][8]
Anatomy
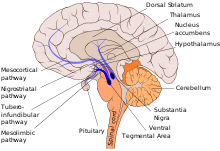
The mesolimbic pathway is a collection of dopaminergic (i.e., dopamine-releasing) neurons that project from the ventral tegmental area (VTA) to the ventral striatum, which includes the nucleus accumbens (NAcc) and olfactory tubercle.[9] It is one of the component pathways of the medial forebrain bundle, which is a set of neural pathways that mediate brain stimulation reward.[10]
The VTA is located in the midbrain and consists of dopaminergic, GABAergic, and glutamatergic neurons.[11] The dopaminergic neurons in this region receive stimuli from both cholinergic neurons in the pedunculopontine nucleus and the laterodorsal tegmental nucleus as well as glutamatergic neurons in other regions such as the prefrontal cortex. The nucleus accumbens and olfactory tubercle are located in the ventral striatum and are primarily composed of medium spiny neurons.[9][12][13] The nucleus accumbens is subdivided into limbic and motor subregions known as the NAcc shell and NAcc core.[11] The medium spiny neurons in the nucleus accumbens receive input from both the dopaminergic neurons of the VTA and the glutamatergic neurons of the hippocampus, amygdala, and medial prefrontal cortex. When they are activated by these inputs, the medium spiny neurons' projections release GABA onto the ventral pallidum.[11]
Function
The mesolimbic pathway regulates incentive salience, motivation, reinforcement learning, and fear, among other cognitive processes.[14][15][16]
The mesolimbic pathway is involved in motivation cognition. Depletion of dopamine in this pathway, or lesions at its site of origin, decrease the extent to which an animal is willing to go to obtain a reward (e.g. the number of lever presses for intravenous nicotine delivery in rats or time spent searching for food). Dopaminergic drugs are also able to increase the extent an animal is willing to go to obtain a reward. Moreover, the firing rate of neurons in the mesolimbic pathway increases during anticipation of reward, which may explain craving.[17] Mesolimbic dopamine release was once thought to be the primary mediator of pleasure, but is now believed to have only a minor or secondary role in pleasure perception.[18][19]
Clinical significance
Mechanisms of addiction
The mesolimbic pathway and a specific set of the pathway's output neurons (e.g. D1-type medium spiny neurons within the nucleus accumbens) play a central role in the neurobiology of addiction.[6][7][8] Drug addiction is an illness caused by habitual substance abuse that induces chemical changes in the brain's circuitry.[20] Commonly abused substances such as cocaine, alcohol, and nicotine have been shown to increase extracellular levels of dopamine within the mesolimbic pathway, preferentially within the nucleus accumbens. The mechanisms by which these drugs do so vary depending on the drug prototype. For example, cocaine precludes the re-uptake of synaptic dopamine through blocking the presynaptic dopamine transporter. Another stimulant, amphetamine, promotes increased dopamine from the synaptic vesicles. Non-stimulant drugs typically bind with ligand-gated channels or G protein-coupled receptors. Such drugs include alcohol, nicotine, and tetrahydrocannabinol (THC).[21]
These dopaminergic activations of the mesolimbic pathway are accompanied by the perception of reward. This stimulus-reward association shows a resistance to extinction and creates an increased motivation to repeat that same behavior that caused it.[22]
In relation, a 2017 study found that abusive (emotional, physical, and sexual) and adverse life events were associated with a heightened limbic response to cocaine. In other words, individuals who had previously suffered abuse were more likely to have a brain pathway primed for cocaine or drug use.[23]
Relation to neurological and psychological disorders
The mesolimbic pathway is implicated in schizophrenia, depression,[24][25][26] and Parkinson's disease.[27][28] It is also theorized to be implicated in overuse of digital media.[29] Each involves distinct structural changes within the mesolimbic pathway.[24]
Other dopamine pathways
References
- Dreyer JL (2010). "New insights into the roles of microRNAs in drug addiction and neuroplasticity". Genome Med. 2 (12): 92. doi:10.1186/gm213. PMC 3025434. PMID 21205279.
- Ikemoto S (2010). "Brain reward circuitry beyond the mesolimbic dopamine system: a neurobiological theory". Neurosci Biobehav Rev. 35 (2): 129–50. doi:10.1016/j.neubiorev.2010.02.001. PMC 2894302. PMID 20149820.
Recent studies on intracranial self-administration of neurochemicals (drugs) found that rats learn to self-administer various drugs into the mesolimbic dopamine structures–the posterior ventral tegmental area, medial shell nucleus accumbens and medial olfactory tubercle. ... In the 1970s it was recognized that the olfactory tubercle contains a striatal component, which is filled with GABAergic medium spiny neurons receiving glutamatergic inputs form cortical regions and dopaminergic inputs from the VTA and projecting to the ventral pallidum just like the nucleus accumbens
Figure 3: The ventral striatum and self-administration of amphetamine - Malenka RC, Nestler EJ, Hyman SE (2009). Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 147–148, 367, 376. ISBN 978-0-07-148127-4.
VTA DA neurons play a critical role in motivation, reward-related behavior (Chapter 15), attention, and multiple forms of memory. This organization of the DA system, wide projection from a limited number of cell bodies, permits coordinated responses to potent new rewards. Thus, acting in diverse terminal fields, dopamine confers motivational salience (“wanting”) on the reward itself or associated cues (nucleus accumbens shell region), updates the value placed on different goals in light of this new experience (orbital prefrontal cortex), helps consolidate multiple forms of memory (amygdala and hippocampus), and encodes new motor programs that will facilitate obtaining this reward in the future (nucleus accumbens core region and dorsal striatum). In this example, dopamine modulates the processing of sensorimotor information in diverse neural circuits to maximize the ability of the organism to obtain future rewards. ...
The brain reward circuitry that is targeted by addictive drugs normally mediates the pleasure and strengthening of behaviors associated with natural reinforcers, such as food, water, and sexual contact. Dopamine neurons in the VTA are activated by food and water, and dopamine release in the NAc is stimulated by the presence of natural reinforcers, such as food, water, or a sexual partner. ...
The NAc and VTA are central components of the circuitry underlying reward and memory of reward. As previously mentioned, the activity of dopaminergic neurons in the VTA appears to be linked to reward prediction. The NAc is involved in learning associated with reinforcement and the modulation of motoric responses to stimuli that satisfy internal homeostatic needs. The shell of the NAc appears to be particularly important to initial drug actions within reward circuitry; addictive drugs appear to have a greater effect on dopamine release in the shell than in the core of the NAc. - Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. p. 266. ISBN 978-0-07-148127-4.
Dopamine acts in the nucleus accumbens to attach motivational significance to stimuli associated with reward.
- Berridge KC, Kringelbach ML (May 2015). "Pleasure systems in the brain". Neuron. 86 (3): 646–664. doi:10.1016/j.neuron.2015.02.018. PMC 4425246. PMID 25950633.
To summarize: the emerging realization that many diverse pleasures share overlapping brain substrates; better neuroimaging maps for encoding human pleasure in orbitofrontal cortex; identification of hotspots and separable brain mechanisms for generating ‘liking’ and ‘wanting’ for the same reward; identification of larger keyboard patterns of generators for desire and dread within NAc, with multiple modes of function; and the realization that dopamine and most ‘pleasure electrode’ candidates for brain hedonic generators probably did not cause much pleasure after all.
- Robison AJ, Nestler EJ (November 2011). "Transcriptional and epigenetic mechanisms of addiction". Nat. Rev. Neurosci. 12 (11): 623–637. doi:10.1038/nrn3111. PMC 3272277. PMID 21989194.
ΔFosB has been linked directly to several addiction-related behaviors ... Importantly, genetic or viral overexpression of ΔJunD, a dominant negative mutant of JunD which antagonizes ΔFosB- and other AP-1-mediated transcriptional activity, in the NAc or OFC blocks these key effects of drug exposure14,22–24. This indicates that ΔFosB is both necessary and sufficient for many of the changes wrought in the brain by chronic drug exposure. ΔFosB is also induced in D1-type NAc MSNs by chronic consumption of several natural rewards, including sucrose, high fat food, sex, wheel running, where it promotes that consumption14,26–30. This implicates ΔFosB in the regulation of natural rewards under normal conditions and perhaps during pathological addictive-like states.
- Blum K, Werner T, Carnes S, Carnes P, Bowirrat A, Giordano J, Oscar-Berman M, Gold M (2012). "Sex, drugs, and rock 'n' roll: hypothesizing common mesolimbic activation as a function of reward gene polymorphisms". Journal of Psychoactive Drugs. 44 (1): 38–55. doi:10.1080/02791072.2012.662112. PMC 4040958. PMID 22641964.
It has been found that deltaFosB gene in the NAc is critical for reinforcing effects of sexual reward. Pitchers and colleagues (2010) reported that sexual experience was shown to cause DeltaFosB accumulation in several limbic brain regions including the NAc, medial pre-frontal cortex, VTA, caudate, and putamen, but not the medial preoptic nucleus. Next, the induction of c-Fos, a downstream (repressed) target of DeltaFosB, was measured in sexually experienced and naive animals. The number of mating-induced c-Fos-IR cells was significantly decreased in sexually experienced animals compared to sexually naive controls. Finally, DeltaFosB levels and its activity in the NAc were manipulated using viral-mediated gene transfer to study its potential role in mediating sexual experience and experience-induced facilitation of sexual performance. Animals with DeltaFosB overexpression displayed enhanced facilitation of sexual performance with sexual experience relative to controls. In contrast, the expression of DeltaJunD, a dominant-negative binding partner of DeltaFosB, attenuated sexual experience-induced facilitation of sexual performance, and stunted long-term maintenance of facilitation compared to DeltaFosB overexpressing group. Together, these findings support a critical role for DeltaFosB expression in the NAc in the reinforcing effects of sexual behavior and sexual experience-induced facilitation of sexual performance. ... both drug addiction and sexual addiction represent pathological forms of neuroplasticity along with the emergence of aberrant behaviors involving a cascade of neurochemical changes mainly in the brain's rewarding circuitry.
- Olsen CM (December 2011). "Natural rewards, neuroplasticity, and non-drug addictions". Neuropharmacology. 61 (7): 1109–22. doi:10.1016/j.neuropharm.2011.03.010. PMC 3139704. PMID 21459101.
- Ikemoto S (2010). "Brain reward circuitry beyond the mesolimbic dopamine system: a neurobiological theory". Neurosci Biobehav Rev. 35 (2): 129–50. doi:10.1016/j.neubiorev.2010.02.001. PMC 2894302. PMID 20149820.
Recent studies on intracranial self-administration of neurochemicals (drugs) found that rats learn to self-administer various drugs into the mesolimbic dopamine structures–the posterior ventral tegmental area, medial shell nucleus accumbens and medial olfactory tubercle. ... In the 1970s it was recognized that the olfactory tubercle contains a striatal component, which is filled with GABAergic medium spiny neurons receiving glutamatergic inputs form cortical regions and dopaminergic inputs from the VTA and projecting to the ventral pallidum just like the nucleus accumbens
Figure 3: The ventral striatum and self-administration of amphetamine - You ZB, Chen YQ, Wise RA (2001). "Dopamine and glutamate release in the nucleus accumbens and ventral tegmental area of rat following lateral hypothalamic self-stimulation". Neuroscience. 107 (4): 629–39. doi:10.1016/s0306-4522(01)00379-7. PMID 11720786.
- Pierce RC, Kumaresan V (2006). "The mesolimbic dopamine system: The final common pathway for the reinforcing effect of drugs of abuse?". Neuroscience and Biobehavioral Reviews. 30 (2): 215–38. doi:10.1016/j.neubiorev.2005.04.016. PMID 16099045.
- Zhang TA, Maldve RE, Morrisett RA (2006). "Coincident signaling in mesolimbic structures underlying alcohol reinforcement". Biochemical Pharmacology. 72 (8): 919–27. doi:10.1016/j.bcp.2006.04.022. PMID 16764827.
- Purves D et al. 2008. Neuroscience. Sinauer 4ed. 754-56
- Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 6: Widely Projecting Systems: Monoamines, Acetylcholine, and Orexin". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 147–148, 154–157. ISBN 9780071481274.
Neurons from the SNc densely innervate the dorsal striatum where they play a critical role in the learning and execution of motor programs. Neurons from the VTA innervate the ventral striatum (nucleus accumbens), olfactory bulb, amygdala, hippocampus, orbital and medial prefrontal cortex, and cingulate cortex. VTA DA neurons play a critical role in motivation, reward-related behavior, attention, and multiple forms of memory. ... Thus, acting in diverse terminal fields, dopamine confers motivational salience ("wanting") on the reward itself or associated cues (nucleus accumbens shell region), updates the value placed on different goals in light of this new experience (orbital prefrontal cortex), helps consolidate multiple forms of memory (amygdala and hippocampus), and encodes new motor programs that will facilitate obtaining this reward in the future (nucleus accumbens core region and dorsal striatum). ... DA has multiple actions in the prefrontal cortex. It promotes the "cognitive control" of behavior: the selection and successful monitoring of behavior to facilitate attainment of chosen goals. Aspects of cognitive control in which DA plays a role include working memory, the ability to hold information "on line" in order to guide actions, suppression of prepotent behaviors that compete with goal-directed actions, and control of attention and thus the ability to overcome distractions. ... Noradrenergic projections from the LC thus interact with dopaminergic projections from the VTA to regulate cognitive control.
- Engert, Veronika; Pruessner, Jens C (9 January 2017). "Dopaminergic and Noradrenergic Contributions to Functionality in ADHD: The Role of Methylphenidate". Current Neuropharmacology. 6 (4): 322–328. doi:10.2174/157015908787386069. ISSN 1570-159X. PMC 2701285. PMID 19587853.
- Pezze, Marie A.; Feldon, Joram (1 December 2004). "Mesolimbic dopaminergic pathways in fear conditioning". Progress in Neurobiology. 74 (5): 301–320. doi:10.1016/j.pneurobio.2004.09.004. ISSN 0301-0082. PMID 15582224.
- Salamone, John D.; Correa, Mercè (2012). "The Mysterious Motivational Functions of Mesolimbic Dopamine". Neuron. 76 (3): 470–485. doi:10.1016/j.neuron.2012.10.021. PMC 4450094. PMID 23141060.
- Berridge KC, Kringelbach ML (May 2015). "Pleasure systems in the brain". Neuron. 86 (3): 646–664. doi:10.1016/j.neuron.2015.02.018. PMC 4425246. PMID 25950633.
To summarize: the emerging realization that many diverse pleasures share overlapping brain substrates; better neuroimaging maps for encoding human pleasure in orbitofrontal cortex; identification of hotspots and separable brain mechanisms for generating ‘liking’ and ‘wanting’ for the same reward; identification of larger keyboard patterns of generators for desire and dread within NAc, with multiple modes of function; and the realization that dopamine and most ‘pleasure electrode’ candidates for brain hedonic generators probably did not cause much pleasure after all.
- Berridge, Kent C; Kringelbach, Morten L (1 June 2013). "Neuroscience of affect: brain mechanisms of pleasure and displeasure". Current Opinion in Neurobiology. 23 (3): 294–303. doi:10.1016/j.conb.2013.01.017. PMC 3644539. PMID 23375169.
- Administration (US), Substance Abuse and Mental Health Services; General (US), Office of the Surgeon (November 2016). THE NEUROBIOLOGY OF SUBSTANCE USE, MISUSE, AND ADDICTION. US Department of Health and Human Services.
- Adinoff, Bryon (2004). "Neurobiologic Processes in Drug Reward and Addiction". Harvard Review of Psychiatry. 12 (6): 305–320. doi:10.1080/10673220490910844. ISSN 1067-3229. PMC 1920543. PMID 15764467.
- Di Chiara, Gaetano; Bassareo, Valentina (1 February 2007). "Reward system and addiction: what dopamine does and doesn't do". Current Opinion in Pharmacology. Neurosciences. 7 (1): 69–76. doi:10.1016/j.coph.2006.11.003. ISSN 1471-4892. PMID 17174602.
- Regier PS, Monge ZA, Franklin TR, Wetherill RR, Teitelman AM, Jagannathan K, et al. Emotional, physical and sexual abuse are associated with a heightened limbic response to cocaine cues. Addiction Biology. 2017 Nov;22(6):1768-177. doi: 10.1111/adb.12445
- Van, den Heuval DMA, Pasterkamp RJ (2008). "Getting connected in the dopamine system". Progress in Neurobiology. 85 (1): 75–93. doi:10.1016/j.pneurobio.2008.01.003. PMID 18304718.CS1 maint: multiple names: authors list (link)
- Laviolette SR (2007). "Dopamine modulation of emotional processing in cortical and subcortical neural circuits: evidence for a final common pathway in schizophrenia?". Schizophrenia Bulletin. 33 (4): 971–981. doi:10.1093/schbul/sbm048. PMC 2632330. PMID 17519393.
- Diaz J. 1996. How Drugs Influence Behavior: A Neurobehavorial Approach. Prentice Hall
- Nyberg, Eric M.; Tanabe, Jody; Honce, Justin M.; Krmpotich, Theodore; Shelton, Erika; Hedeman, Jessica; Berman, Brian D. (1 May 2015). "Morphologic changes in the mesolimbic pathway in Parkinson's disease motor subtypes". Parkinsonism & Related Disorders. 21 (5): 536–540. doi:10.1016/j.parkreldis.2015.03.008. ISSN 1353-8020. PMC 4424152. PMID 25817514.
- Caminiti, Silvia Paola; Presotto, Luca; Baroncini, Damiano; Garibotto, Valentina; Moresco, Rosa Maria; Gianolli, Luigi; Volonté, Maria Antonietta; Antonini, Angelo; Perani, Daniela (1 January 2017). "Axonal damage and loss of connectivity in nigrostriatal and mesolimbic dopamine pathways in early Parkinson's disease". NeuroImage: Clinical. 14: 734–740. doi:10.1016/j.nicl.2017.03.011. ISSN 2213-1582. PMC 5379906. PMID 28409113.
- "Dopamine, Smartphones & You: A battle for your time". Science in the News. 1 May 2018. Retrieved 10 May 2019.