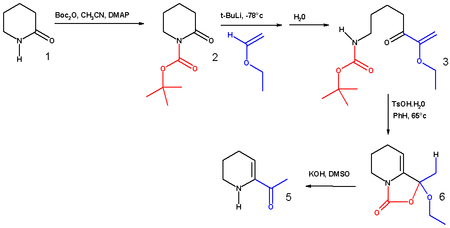
Di-tert-butyl dicarbonate
Di-tert-butyl dicarbonate is a reagent widely used in organic synthesis.[1] Since this compound can be regarded formally as the acid anhydride derived from a tert-butoxycarbonyl (Boc) group, it is commonly referred to as Boc anhydride. This pyrocarbonate reacts with amines to give N-tert-butoxycarbonyl or so-called Boc derivatives. These carbamate derivatives do not behave as amines, which allows certain subsequent transformations to occur that would be incompatible with the amine functional group. The Boc group can later be removed from the amine using moderately strong acids (e.g., trifluoroacetic acid). Thus, Boc serves as a protective group, for instance in solid phase peptide synthesis. Boc-protected amines are unreactive to most bases and nucleophiles, allowing for the use of the fluorenylmethyloxycarbonyl group (Fmoc) as an orthogonal protecting group.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Di-tert-butyl dicarbonate | |
| Other names
Di-t-butyl pyrocarbonate Boc anhydride Boc2O | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.042.021 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H18O5 | |
| Molar mass | 218.249 g·mol−1 |
| Appearance | Colorless solid or oil |
| Density | 0.95 g·cm−3 |
| Melting point | 22 to 24 °C (72 to 75 °F; 295 to 297 K) |
| Boiling point | 56 to 57 °C (133 to 135 °F; 329 to 330 K) (0.5 mmHg) |
| Insol | |
| Solubility in other solvents | Soluble in most organic solvents |
| Hazards | |
| Main hazards | Very toxic on inhalation T+, LC50 = 100 mg/m3 (4 hr, rat) |
| Related compounds | |
Related compounds |
Ethyl chloroformate Phosgene Diethyl pyrocarbonate Dimethyl dicarbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
Di-tert-butyl dicarbonate is inexpensive, so it is usually purchased. Classically, this compound is prepared from tert-butanol, carbon dioxide, and phosgene, using DABCO as a base:[2]
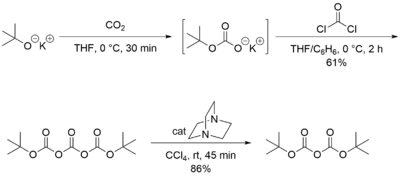
This route is currently employed commercially by manufacturers in China and India. European and Japanese companies use the reaction of sodium tert-butoxide with carbon dioxide, catalysed by p-toluenesulfonic acid or methanesulfonic acid. This process involves a distillation of the crude material yielding a very pure grade.
Boc anhydride is also available as a 70% solution in toluene or THF. As boc anhydride may melt at ambient temperatures, its storage and handling is sometimes simplified by using a solution.
Protection and deprotection of amines
The Boc group can be added to the amine under aqueous conditions using di-tert-butyl dicarbonate in the presence of a base such as sodium bicarbonate. Protection of the amine can also be accomplished in acetonitrile solution using 4-dimethylaminopyridine (DMAP) as the base.[3]
Removal of the Boc in amino acids can be accomplished with strong acids such as trifluoroacetic acid neat or in dichloromethane or with HCl in methanol.[4][5][6] A complication may be the tendency of the t-butyl cation intermediate to alkylate other nucleophiles; scavengers such as anisole or thioanisole may be used.[7][8] Selective cleavage of the N-Boc group in the presence of other protecting groups is possible when using AlCl3.
Reaction with trimethylsilyl iodide in acetonitrile followed by methanol is a mild and versatile method of deprotecting Boc-protected amines.[9][10][11][12]
The use of triethylsilane as a carbocation scavenger in the presence of trifluoroacetic acid in dichloromethane has been shown to lead to increased yields, decreased reaction times, simple work-up and improved selectivity for the deprotection of t-butyl ester and t-butoxycarbonyl sites in protected amino-acids and peptides in the presence of other acid-sensitive protecting groups such as the benzyloxycarbonyl, 9-fluorenylmethoxycarbonyl, O- and S-benzyl and t-butylthio groups.[13]
Other uses
The synthesis of 6-acetyl-1,2,3,4-tetrahydropyridine, an important bread aroma compound, starting from 2-piperidone was accomplished using t-boc anhydride.[14] (See Maillard reaction). The first step in this reaction sequence is the formation of the carbamate from the reaction of the amide nitrogen with boc anhydride in acetonitrile using DMAP as a catalyst.
Di-tert-butyl dicarbonate also finds applications as a polymer blowing agent due to its decomposition into gaseous products upon heating.[15][16]
Hazards
Bottles of di-tert-butyl dicarbonate buildup of internal pressure in sealed containers caused by its slow decomposition to di-tert-butyl carbonate and ultimately tert-butanol and CO2 in the presence of moisture. For this reason, it is usually sold and stored in plastic bottles rather than glass ones.
The main hazard of the reagent is its inhalational toxicity. Its median lethal concentration of 100 mg/m3 over 4 hours in rats[17] is comparable to that of phosgene[18] (49 mg/m3 over 50 min in rats).
References
- M. Wakselman, "Di-t-butyl Dicarbonate" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289.
- Pope BM, Yamamoto Y, Tarbell DS (1977). "DI-tert-BUTYL DICARBONATE". Organic Syntheses. 57: 45. doi:10.15227/orgsyn.057.0045. ISSN 0078-6209.
- Yochai Basel; Alfred Hassner (2000). "Di-tert-butyl Dicarbonate and 4-(Dimethylamino)pyridine Revisited. Their Reactions with Amines and Alcohols". J. Org. Chem. 65: 6368–6380. doi:10.1021/jo000257f.
- Williams RM, Sinclair PJ, DeMong DE, Chen D, Zhai D (2003). "ASYMMETRIC SYNTHESIS OF N-tert-BUTOXYCARBONYL a-AMINO ACIDS. SYNTHESIS OF (5S,6R)-4-tert-BUTOXYCARBONYL-5,6-DIPHENYLMORPHOLIN-2-ONE". Organic Syntheses. 80: 18. doi:10.15227/orgsyn.080.0018. ISSN 0078-6209.
- E. A. Englund; H. N. Gopi; D. H. Appella (2004). "An Efficient Synthesis of a Probe for Protein Function: 2,3-Diaminopropionic Acid with Orthogonal Protecting Groups". Org. Lett. 6 (2): 213–215. doi:10.1021/ol0361599. PMID 14723531.
- D. M. Shendage; R. Fröhlich; G. Haufe (2004). "Highly Efficient Stereoconservative Amidation and Deamidation of α-Amino Acids". Org. Lett. 6 (21): 3675–3678. doi:10.1021/ol048771l. PMID 15469321.
- Lundt, Behrend F.; Johansen, Nils L.; Vølund, Aage; Markussen, Jan (1978). "Removal of t-Butyl and t-Butoxycarbonyl Protecting Groups with Trifluoroacetic acid". International Journal of Peptide and Protein Research. 12 (5): 258–268. doi:10.1111/j.1399-3011.1978.tb02896.x. PMID 744685.
- Andrew B. Hughes. "1. Protection Reactions". In Vommina V. Sureshbabu; Narasimhamurthy Narendra (eds.). Amino Acids, Peptides and Proteins in Organic Chemistry: Protection Reactions, Medicinal Chemistry, Combinatorial Synthesis. 4. doi:10.1002/9783527631827.ch1.
- Michael E. Jung; Mark A. Lyster (1978). "Conversion of alkyl carbamates into amines via treatment with trimethylsilyl iodide". J. Chem. Soc., Chem. Commun. (7): 315–316. doi:10.1039/C39780000315.
- Richard S. Lott; Virander S. Chauhan; Charles H. Stammer (1979). "Trimethylsilyl iodide as a peptide deblocking agent". J. Chem. Soc., Chem. Commun. (11): 495–496. doi:10.1039/C39790000495.
- Olah, G; Narang, S. C. (1982). "Iodotrimethylsilane—a versatile synthetic reagent". Tetrahedron. 38 (15): 2225. doi:10.1016/0040-4020(82)87002-6.
- Zhijian Liu; Nobuyoshi Yasuda; Michael Simeone; Robert A. Reamer (2014). "N-Boc Deprotection and Isolation Method for Water-Soluble Zwitterionic Compounds". J. Org. Chem. 79: 11792–11796. doi:10.1021/jo502319z. PMID 25376704.
- Mehta, Anita; Jaouhari, Rabih; Benson, Timothy J.; Douglas, Kenneth T. (1992). "Improved efficiency and selectivity in peptide synthesis: Use of triethylsilane as a carbocation scavenger in deprotection of t-butyl esters and t-butoxycarbonyl-protected sites". Tetrahedron Letters. 33 (37): 5441–5444. doi:10.1016/S0040-4039(00)79116-7. ISSN 0040-4039.
- T. J. Harrison; G. R. Dake (2005). "An Expeditious, High-Yielding Construction of the Food Aroma Compounds 6-Acetyl-1,2,3,4-tetrahydropyridine and 2-Acetyl-1-pyrroline". J. Org. Chem. 70 (26): 10872–10874. doi:10.1021/jo051940a. PMID 16356012.
- Wirth, D. (8 April 2020). "Highly Expandable Foam for Lithographic 3D Printing". ACS Appl. Mater. Interfaces. 12 (16): 19033–19043. doi:10.1021/acsami.0c02683. PMID 32267677.
- Wypych, George (20 February 2017). Handbook of Foaming and Blowing Agents - 1st Edition (1st ed.). ChemTec Publishing. p. 258. ISBN 978-1-895198-99-7. Retrieved 4 May 2020.
- "Material Safety Data Sheet" (PDF). CHEM-IMPEX INTERNATIONAL INC. Retrieved 2016-09-10.
- "Phosgene". Wireless Information System for Emergency Responders. US National Library of Medicine. Retrieved 2016-09-10.
External links
- Protection or deprotection conditions - Synthetic protocols from organic-reaction.com
- Basel, Yochai; Hassner, Alfred (2001). "Imidazole and Trifluoroethanol as Efficient and Mild Reagents for Destruction of Excess Di-tert-butyl Dicarbonate [(BOC)2O]". Synthesis. 2001 (4): 0550. doi:10.1055/s-2001-12350.