Beta-peptide
β-peptides consist of β amino acids, which have their amino group bonded to the β carbon rather than the α carbon as in the 20 standard biological amino acids. The only common naturally occurring β amino acid is β-alanine; although it is used as a component of larger bioactive molecules, β-peptides in general do not appear in nature. For this reason β-peptide-based antibiotics are being explored as ways of evading antibiotic resistance. Early studies in this field were published in 1996 by the group of Dieter Seebach[1] and that of Samuel Gellman.[2]
Chemical structure and synthesis
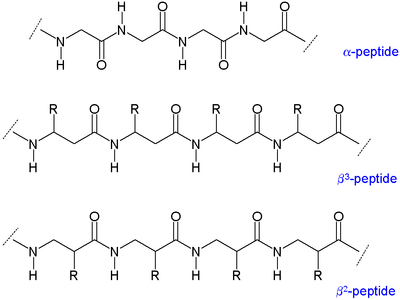
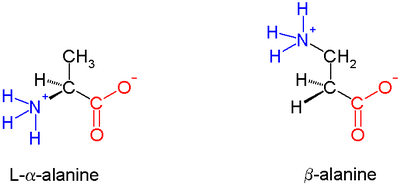
In α amino acids (molecule at left), both the carboxylic acid group (red) and the amino group (blue) are bonded to the same carbon center, termed the α carbon () because it is one atom away from the carboxylate group. In β amino acids, the amino group is bonded to the β carbon (), which is found in most of the 20 standard amino acids. Only glycine lacks a β carbon, which means that β-glycine is not possible.
The chemical synthesis of β amino acids can be challenging, especially given the diversity of functional groups bonded to the β carbon and the necessity of maintaining chirality. In the alanine molecule shown, the β carbon is achiral; however, most larger amino acids have a chiral atom. A number of synthesis mechanisms have been introduced to efficiently form β amino acids and their derivatives[3][4] notably those based on the Arndt-Eistert synthesis.
Two main types of β-peptides exist: those with the organic residue (R) next to the amine are called β3-peptides and those with position next to the carbonyl group are called β2-peptides.[5]
Secondary structure
Because the backbones of β-peptides are longer than those of peptides that consist of α-amino acids, β-peptides form different secondary structures. The alkyl substituents at both the α and β positions in a β amino acid favor a gauche conformation about the bond between the α-carbon and β-carbon. This also affects the thermodynamic stability of the structure.
Many types of helix structures consisting of β-peptides have been reported. These conformation types are distinguished by the number of atoms in the hydrogen-bonded ring that is formed in solution; 8-helix, 10-helix, 12-helix, 14-helix, and 10/12-helix have been reported. Generally speaking, β-peptides form a more stable helix than α-peptides.[6]
Clinical potential
β-peptides are stable against proteolytic degradation in vitro and in vivo, an important advantage over natural peptides in the preparation of peptide-based drugs.[7] β-Peptides have been used to mimic natural peptide-based antibiotics such as magainins, which are highly potent but difficult to use as drugs because they are degraded by proteolytic enzymes in the body.[8]
List
8 beta forms exist: alanine, leucine, lysine, arginine, glutamate, glutamine, phenylalanine, tyrosine forms.[9] Aspartic acid is regarded as both beta and alpha forms, although the beta form is found in microcystins.[9]
See also
References
- Seebach D, Overhand M, Kühnle FNM, Martinoni B, Oberer L, Hommel U, Widmer H (June 1996). "β-Peptides: Synthesis by Arndt-Eistert homologation with concomitant peptide coupling. Structure determination by NMR and CD spectroscopy and by X-ray crystallography. Helical secondary structure of a -hexapeptide in solution and its stability towards pepsin". Helvetica Chimica Acta. 79 (4): 913–941. doi:10.1002/hlca.19960790402.
- Appella DH, Christianson LA, Karle IL, Powell DR, Gellman SH (1996). "β-Peptide Foldamers: Robust Helix Formation in a New Family of -Amino Acid Oligomers". J. Am. Chem. Soc. 118 (51): 13071–2. doi:10.1021/ja963290l.
- Basler B, Schuster O, Bach T (November 2005). "Conformationally constrained β-amino acid derivatives by intramolecular [2 + 2]-photocycloaddition of a tetronic acid amide and subsequent lactone ring opening". J. Org. Chem. 70 (24): 9798–808. doi:10.1021/jo0515226. PMID 16292808.
- Murray JK, Farooqi B, Sadowsky JD, et al. (September 2005). "Efficient synthesis of a β-peptide combinatorial library with microwave irradiation". J. Am. Chem. Soc. 127 (38): 13271–80. doi:10.1021/ja052733v. PMID 16173757.
- Seebach D, Matthews JL (1997). "β-Peptides: a surprise at every turn". Chem. Commun. (21): 2015–22. doi:10.1039/a704933a.
- Gademann K, Hintermann T, Schreiber JV (October 1999). "Beta-peptides: twisting and turning". Curr. Med. Chem. 6 (10): 905–25. PMID 10519905.
- Beke T, Somlai C, Perczel A (January 2006). "Toward a rational design of β-peptide structures". J Comput Chem. 27 (1): 20–38. doi:10.1002/jcc.20299. PMID 16247761.
- Porter EA, Weisblum B, Gellman SH (2002). "Mimicry of host-defense peptides by unnatural oligomers: antimicrobial β-peptides". J. Am. Chem. Soc. 124 (25): 7324–30. doi:10.1021/ja0260871. PMID 12071741.
- Enantioselective Synthesis of Beta-Amino Acids Sec 2.2, Eusebio Juaristi, Vadim A. Soloshonok