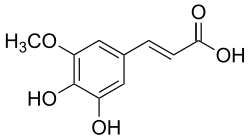
5-Hydroxyferulic acid
It is a precursor in the biosynthesis of sinapic acid. Phenylalanine is first converted to cinnamic acid by the action of the enzyme phenylalanine ammonia-lyase (PAL). A series of enzymatic hydroxylations and methylations leads to coumaric acid, caffeic acid, ferulic acid, 5-hydroxyferulic acid and sinapic acid.
 | |
| Names | |
|---|---|
| IUPAC name
(E)-3-(3,4-dihydroxy-5-methoxyphenyl)prop-2-enoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.230.072 |
PubChem CID |
|
| |
| |
| Properties | |
| C10H10O5 | |
| Molar mass | 210.18 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
5-Hydroxyferulic acid is a hydroxycinnamic acid.
Thus 5-hydroxyferulic acid is formed from ferulic acid by the action of the specific enzyme ferulate 5-hydroxylase (F5H).
References
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.