3,4-Dihydroxyphenylacetaldehyde
3,4-Dihydroxyphenylacetaldehyde (DOPAL) is an important metabolite of the major brain neurotransmitter dopamine. All of the enzymatic metabolism of dopamine in neurons passes through DOPAL. According to the "catecholaldehyde hypothesis," DOPAL plays a role in the pathogenesis of Parkinson's disease.[2] DOPAL has been chemically synthesized.[3] DOPAL is detoxified mainly by aldehyde dehydrogenase.
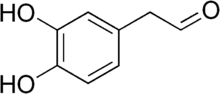 | |
| Names | |
|---|---|
| Preferred IUPAC name
(3,4-Dihydroxyphenyl)acetaldehyde | |
| Other names
2-(3,4-Dihydroxyphenyl)acetaldehyde[1] Dopaldehyde | |
| Identifiers | |
3D model (JSmol) |
|
| 3DMet | |
| Abbreviations | DOPAL |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.237.172 |
| KEGG | |
| MeSH | 3,4-dihydroxyphenylacetaldehyde |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H8O3 | |
| Molar mass | 152.149 g·mol−1 |
| Density | 1.306 g/mL |
| Boiling point | 351 °C (664 °F; 624 K) |
| Related compounds | |
Related 2-phenyl aldehydes |
Phenylacetaldehyde |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
References
- "3,4-dihydroxyphenylacetaldehyde - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 24 June 2005. Identification and Related Records. Retrieved 13 October 2011.
- Goldstein DS, Sullivan P, Holmes C, Miller GW, Alter S, Strong R, Mash DC, Kopin IJ, Sharabi Y., "Determinants of buildup of the toxic dopamine metabolite DOPAL in Parkinson's disease," J. Neurochem. 2013 Sep;126(5):591-603.
- Li, W., Spaziano, V.T., Burke, WJ., "Synthesis of a biochemically important aldehyde - 3,4-dihydroxyphenylacetaldehyde", Bio-Organic Chemistry, 26, 45-50, 1998.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.