Triosmium dodecacarbonyl
Triosmium dodecacarbonyl is a chemical compound with the formula Os3(CO)12. This yellow-colored metal carbonyl cluster is an important precursor to organo-osmium compounds. Many of the advances in cluster chemistry have arisen from studies on derivatives of Os3(CO)12 and its lighter analogue Ru3(CO)12.
 | |
| Names | |
|---|---|
| IUPAC name
cyclo-tris(tetracarbonylosmium) | |
| Other names
Osmium carbonyl | |
| Identifiers | |
| ECHA InfoCard | 100.036.157 |
CompTox Dashboard (EPA) |
|
| Properties | |
| C12O12Os3 | |
| Molar mass | 906.81 g/mol |
| Appearance | yellow solid |
| Density | 3.48 g/cm3 |
| Melting point | 224 °C (435 °F; 497 K) |
| Boiling point | sublimes in vacuum |
| insoluble | |
| Solubility in other solvents | slightly in organic solvents |
| Structure | |
| 0 D (0 C·m) | |
| Hazards | |
| Main hazards | CO source |
| R-phrases (outdated) | 22-36/37/38 |
| S-phrases (outdated) | 22-26-36/37/39 |
| Related compounds | |
Related compounds |
Fe3(CO)12 Ru3(CO)12 Decacarbonyldihydridotriosmium |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure and synthesis
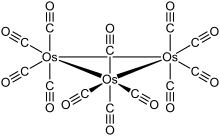
The cluster has D3h symmetry, consisting of an equilateral triangle of Os atoms, each of which bears two axial and two equatorial CO ligands. The Os–Os bond distance is 2.88 Â (288 pm).[1] Ru3(CO)12 has the same structure, whereas Fe3(CO)12 is different, with two bridging CO ligands resulting in C2v symmetry.
Os3(CO)12 is prepared by the direct reaction of OsO4 with carbon monoxide at 175 °C:[2]
- 3 OsO4 + 24 CO → Os3(CO)12 + 12 CO2
The yield is nearly quantitative.
Reactions
The chemical properties of Os3(CO)12 have been thoroughly examined. Direct reactions of ligands with the cluster often lead to complex product distributions because the inert Os–CO bonds require high temperatures to break, and at such high temperatures the initially formed adducts react further. More successfully, Os3(CO)12 is converted to more labile derivatives such as Os3(CO)11(MeCN) and Os3(CO)10(MeCN)2 using Me3NO as a decarbonylating agent.[3] Os3(CO)11(MeCN) reacts with a variety of even weakly basic ligands to form adducts such as Os3(CO)11(ethylene) and Os3(CO)11(pyridine). The direct reaction of Os3(CO)12 with ethylene and pyridine results in degradation of these organic ligands to give the vinyl hydride HOs3(CO)10(η1,η2-C2H3) and the pyridyl-hydride HOs3(CO)10(NC5H4). These products illustrate the stability of Os–H and Os–C bonds.
Os3(CO)12 is a platform to examine the ways that hydrocarbons can interact with ensembles of metals. For example, the molecule CH3(H)Os3(CO)10 provided one of the first clear-cut examples of agostic bonding.[4]
From the perspective of bonding, the molecule H2Os3(CO)10 is noteworthy. In this compound, the two hydride ligands bridge one Os-Os edge. The molecule displays reactivity reminiscent of diborane.[5]
References
- Corey, E. R.; Dahl, L. F. “The Molecular and Crystal Structure of Os3(CO)12” Inorganic Chemistry 1962, volume 1, pages 521–526; doi:10.1021/ic50003a016.
- Drake, S. R.; Loveday, P. A. “Dodecarbonyltriosmium” Inorganic Syntheses, 1990, volume 28, pages 230–231. ISBN 0-471-52619-3.
- Nicholls, J. N.; Vargas, M. D. “Some Useful Derivatives of Dodecarbonyltriosmium” Inorganic Syntheses, 1990, volume 28, pages 232–235. ISBN 0-471-52619-3.
- Calvert, R. B.; Shapley, J. R. “Decacarbonyl(methyl)hydrotriosmium: NMR evidence for a Carbon..Hydrogen..Osmium Interaction” Journal of the American Chemical Society 1978, volume 100, pages 7726–7727; doi:10.1021/ja00492a047.
- Keister, J. B.; Shapley, J. R. “Solution Structures and Dynamics of complexes of Decacarbonyldihydrotriosmium with Lewis Bases” Inorganic Chemistry 1982, volume 21, pages 3304–3310; doi:10.1021/ic00139a011.