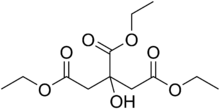
Triethyl citrate
Triethyl citrate is an ester of citric acid. It is a colorless, odorless liquid used as a food additive (E number E1505) to stabilize foams, especially as whipping aid for egg white.[4] It is also used in pharmaceutical coatings and plastics.[5]
 | |
| Names | |
|---|---|
| IUPAC name
Triethyl citrate[2] | |
| Preferred IUPAC name
Triethyl 2-hydroxypropane-1,2,3-tricarboxylate | |
| Other names
Ethyl citrate E1505 Citric acid ethyl ester | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.974 |
| EC Number |
|
| E number | E1505 (additional chemicals) |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H20O7 | |
| Molar mass | 276.283 g/mol |
| Appearance | Oily liquid |
| Density | 1.137 g/mL at 25 °C |
| Melting point | −55 °C (−67 °F; 218 K)[3] |
| Boiling point | 294 °C (561 °F; 567 K) at 1 atm 235 °C at 150 mmHg |
| 65 g/L[3] | |
| -161.9·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Triethyl citrate is also used as a plasticizer for polyvinyl chloride (PVC) and similar plastics.[6]
Triethyl citrate has been used as a pseudo-emulsifier in e-cigarette juices. It functions essentially like lecithin used in food products, but with the possibility of vaporization which lecithin does not have.
References
- Triethyl citrate at Sigma-Aldrich
- 'citric acid' is listed as a valid, expert-verified IUPAC name for 2-hydroxypropane-1,2,3-tricarboxylic acid on ChemSpider.
- Record of Triethyl citrate in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- William J. Stadelman; Owen J. Cotterill (1995). Egg Science and Technology. Haworth Press. ISBN 1-56022-855-5.
- Pharmaceutical Coatings Bulletin 102-4, morflex.com
- Hwan-Man Park; Manjusri Misra; Lawrence T. Drzal & Amar K. Mohanty (2004). ""Green" Nanocomposites from Cellulose Acetate Bioplastic and Clay: Effect of Eco-Friendly Triethyl Citrate Plasticizer". Biomacromolecules. 5 (6): 2281–2288. doi:10.1021/bm049690f. PMID 15530043.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.