Dicyclohexylamine
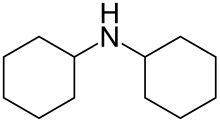
Dicyclohexylamine is a secondary amine with the chemical formula HN(C6H11)2. It is a colorless liquid, although commercial samples can appear yellow. It has a fishy odor, typical for amines. It is sparingly soluble in water. As an amine, it is an organic base and useful precursor to other chemicals.[1]
 | |
| Names | |
|---|---|
| IUPAC name
N,N-Dicyclohexylamine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.002.710 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H23N | |
| Molar mass | 181.323 g·mol−1 |
| Appearance | Pale yellow liquid |
| Density | 0.912 g/cm3 |
| Melting point | −0.1 °C (31.8 °F; 273.0 K) |
| Boiling point | 255.8 °C (492.4 °F; 529.0 K) |
| 0.8 g/L | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
373 mg/kg (oral) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis
Dicyclohexylamine, as a mixture with cyclohexylamine, is prepared by the catalytic hydrogenation of aniline (phenylamine), with a catalyst of ruthenium and/or palladium. This method produces mainly cyclohexylamine with little dicyclohexylamine. Better results have been reported when the catalyst is applied to a support of niobic acid and/or tantalic acid.[2] It is also obtained by reductive amination of cyclohexanone with ammonia or cyclohexylamine.[1]
Dicyclohexylamine may also be prepared by pressure hydrogenation of diphenylamine using a ruthenium catalyst, or by the reaction of cyclohexanone with cyclohexylamine in the presence of a palladium/carbon catalyst under a hydrogen pressure of about 4 mm Hg.[2]
Applications
Dicyclohexylamine has applications that are similar to those of cyclohexylamine, namely the production of:
- antioxidants in rubber and plastics
- vulcanization accelerators for rubber
- corrosion inhibitors in steam pipes and boilers
- agrochemicals
- textile chemicals
- catalysts for flexible polyurethane foams
References
- Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic". In Ullmann's Encyclopedia of Industrial Chemistry, 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a02_001
- U.S. Patent 5322965, "Process for the preparation of a mixture of cyclohexylamine and dicyclohexylamine using a supported noble metal catalyst", Bayer AG, 21 June 1994