Chloroacetic acid
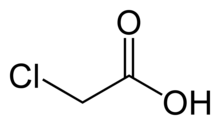
Chloroacetic acid, industrially known as monochloroacetic acid (MCA), is the organochlorine compound with the formula ClCH2CO2H. This carboxylic acid is a useful building-block in organic synthesis. It is a colorless solid. Related compounds are dichloroacetic acid and trichloroacetic acid.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Chloroacetic acid | |
| Systematic IUPAC name
Chloroethanoic acid | |
| Other names
2-Chloroacetic acid 2-Chloroethanoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.072 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H3ClO2 | |
| Molar mass | 94.49 g·mol−1 |
| Appearance | Colorless or white crystals |
| Density | 1.58 g·cm−3 |
| Melting point | 63 °C (145 °F; 336 K) |
| Boiling point | 189.3 °C (372.7 °F; 462.4 K) |
| 85.8 g/100mL (25 °C) | |
| Solubility | Soluble in methanol, acetone, diethyl ether, benzene, chloroform, ethanol |
| log P | 0.22 |
| Vapor pressure | 0.22 hPa |
| Acidity (pKa) | 2.86[1] |
| -48.1·10−6 cm3/mol | |
Refractive index (nD) |
1.4351 (55 °C) |
| Structure | |
| Monoclinic | |
| Thermochemistry | |
Heat capacity (C) |
144.02 J/K mol |
Std enthalpy of formation (ΔfH⦵298) |
-490.1 kJ/mol |
| Hazards | |
| Main hazards | alkylating agent |
| Safety data sheet | External MSDS |
| GHS pictograms |    |
| GHS Signal word | Danger |
GHS hazard statements |
H301, H311, H314, H331, H400 |
| P260, P261, P264, P270, P271, P273, P280, P301+310, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P311, P312, P321, P322, P330, P361, P363, P391, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 126 °C (259 °F; 399 K) |
| 470 °C (878 °F; 743 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
165 mg/kg (mouse, oral) |
| Related compounds | |
Related compounds |
2-Chloropropionic acid Sodium chloroacetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Production
Chloroacetic acid was first prepared (in impure form) by the French chemist Félix LeBlanc (1813–1886) in 1843 by chlorinating acetic acid in the presence of sunlight,[2] and in 1857 (in pure form) by the German chemist Reinhold Hoffmann (1831–1919) by refluxing glacial acetic acid in the presence of chlorine and sunlight,[3] and then by the French chemist Charles Adolphe Wurtz by hydrolysis of chloroacetyl chloride (ClCH2COCl), also in 1857.[4]
Chloroacetic acid is prepared industrially via two routes. The predominant method involves chlorination of acetic acid, with acetic anhydride as a catalyst. This route suffers from the production of dichloroacetic acid and trichloroacetic acid as impurities, which are difficult to separate by distillation.
The second method entails hydrolysis of trichloroethylene:
- ClHC=CCl
2 + 2 H
2O → ClH
2CCO
2H + 2 HCl
The hydrolysis is conducted at 130–140 °C in a concentrated (75%+) solution of sulfuric acid. This method produces a highly pure product, unlike the halogenation route. However, the significant quantities of HCl released have led to the increased popularity of the halogenation route. Approximately, 420,000,000 kg/y are produced globally.[5]
Uses and reactions
Most reactions take advantage of the high reactivity of the C–Cl bond.
In its largest-scale application, chloroacetic acid is used to prepare the thickening agent carboxymethyl cellulose and carboxymethyl starch.
Chloroacetic acid is also used in the production of phenoxy herbicides by etherification with chlorophenols. In this way 2-methyl-4-chlorophenoxyacetic acid (MCPA), 2,4-Dichlorophenoxyacetic acid, and [[2,4,5-Trichlorophenoxyacetic acid]|2,4,5-trichlorophenoxyacetic acid]] (2,4,5-T) are produced. It is the precursor to the herbicide glyphosate and dimethoate. Chloroacetic acid is converted to chloroacetyl chloride, a precursor to adrenaline (epinephrine). Displacement of chloride by sulfide gives thioglycolic acid, which is used as a stabilizer in PVC and a component in some cosmetics.[5]
Illustrative of its usefulness in organic chemistry is the O-alkylation of salicylaldehyde with chloroacetic acid, followed by decarboxylation of the resulting ether, producing benzofuran.[6][7]
Safety
Like other chloroacetic acids and related halocarbons, chloroacetic acid is a hazardous alkylating agent. The LD50 for rats is 76 mg/kg.[5]
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.[8]
See also
References
- Dippy, J. F. J.; Hughes, S. R. C.; Rozanski, A. (1959). "498. The dissociation constants of some symmetrically disubstituted succinic acids". Journal of the Chemical Society. 1959: 2492–2498. doi:10.1039/JR9590002492.
- LeBlanc, Félix (1844) "Recherches sur les produits dérivés de l'éther acétique par l'action du chlore, et en particulier sur l'éther acétique perchloruré," Annales de Chimie et de Physique, 3rd series, 10 : 197–221 ; see especially p. 212
- Hoffmann, Reinhold (1857) "Ueber Monochloressigsäure" (On mono-chloroacetic acid), Annalen der Chemie und Pharmacie, 102 (1) : 1–20.
- Wurtz, Adolphe (1857) "Note sur l'aldéhyde et sur le chlorure d'acétyle" (Note on aldehyde and on acetyl chloride), Annales de chimie et de physique, 3rd series, 49 : 58–62, see p. 61.
- Koenig, G.; Lohmar, E.; Rupprich, N. (2005). "Chloroacetic Acids". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a06_537.CS1 maint: multiple names: authors list (link)
- Burgstahler, A. W.; Worden, L. R. (1966). "Coumarone". Organic Syntheses. 46: 28. doi:10.15227/orgsyn.046.0028.CS1 maint: multiple names: authors list (link); Collective Volume, 5, p. 251
- Inglis, J. K. H. (1928). "Ethyl Cyanoacetate". Organic Syntheses. 8: 74. doi:10.15227/orgsyn.008.0074.
- "40 C.F.R.: Appendix A to Part 355—The List of Extremely Hazardous Substances and Their Threshold Planning Quantities" (PDF) (1 July 2008 ed.). Government Printing Office. Archived from the original (PDF) on 25 February 2012. Retrieved 29 October 2011. Cite journal requires
|journal=(help)
External links
- "Monochloroacetic Acid". CABB. Archived from the original on 6 February 2015. Retrieved 6 February 2015.
- "Monochloroacetic Acid". IPCS Inchem. Retrieved 20 May 2007.
